1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Recovery of lignin from black liquor
2.3 Measurements
3. Results and Discussion
3.1 Effect of acid addition sequence and pH conditions
3.2 Effect of black liquor dilution concentration
4. Conclusions
1. Introduction
Black liquor, a byproduct generated in large quantities during the pulp and paper manufacturing process, is a highly alkaline solution composed of lignin, inorganic salts, organic acids, and residual carbohydrates remaining after the dissolution of cellulose and hemicellulose during pulping [1,2,3]. Globally, more than 50 million tons of lignin are produced annually in the form of black liquor; however, lignin-derived products account for only about 1–2% of total lignin production. Most black liquor is utilized as boiler fuel for energy recovery and for regenerating pulping chemicals such as NaOH and Na2S [4].
Recently, there has been growing interest in shifting from this conventional energy recovery approach toward the valorization of lignin as a high-value industrial material [5]. Lignin, a natural aromatic polymer, exhibits outstanding thermal stability, ultraviolet (UV) resistance, antioxidant capacity, and adhesive properties, making it a suitable high-performance precursor for various applications, including carbon fibers, polyurethane resins, adhesives, and polymer composites [6].
The recovery of lignin from kraft black liquor is generally carried out through acidification. According to previous studies, factors such as precipitation pH, type of acid, temperature, salt concentration of the black liquor, and the structural characteristics of lignin are known to significantly influence the recovery efficiency and molecular structure of lignin [7]. Sun et al. [8] demonstrated in experiments using black liquor from oil palm empty fruit bunch that, across various pH levels (4.8, 4.0, 3.0, 2.0, and 1.5), decreasing pH led to increased lignin yield and purity. Similarly, Sewring et al. [9] reported that when sulfuric acid was added to kraft black liquor to precipitate lignin, acidification conditions within the pH range of 3.9–8.9 had a direct effect on particle size, and the highest recovery yield was achieved under strong acidic conditions below pH 3. As such, lignin precipitation conditions are known to affect not only yield and purity but also molecular weight characteristics. Watumlawar and Park [10] reported that as pH increased, lignin recovery yield gradually decreased, while the average molecular weight (Mw) of the precipitated lignin increased from 4,240 g/mol at pH 3 to 4,510 g/mol at pH 9. Additionally, Ryu et al. [11] found that, when sulfuric acid and hydrochloric acid were used to precipitate lignin from organosolv pulp-based oak black liquor, sulfuric acid produced a higher recovery yield and slightly higher Mw than hydrochloric acid. Regardless of the acid type, however, lower precipitation pH resulted in higher recovery yield and purity, along with a decrease in molecular weight. Meanwhile, some studies [12] have confirmed that CO2 injection into highly alkaline black liquor can cause slight pH reductions and changes in lignin solubility, which may induce limited aggregation under certain conditions. However, most previous studies have primarily focused on chemical factors, such as acid type and precipitation pH, while systematic investigations into the effects of process parameters—such as the order of acid addition and black liquor dilution ratio—on lignin recovery efficiency and purification characteristics remain limited.
Therefore, in this study, hardwood kraft black liquor generated from domestic pulp and paper manufacturing processes was used as the experimental material. The variables were set as follows: acid addition sequence (black liquor → sulfuric acid solution and sulfuric acid solution → black liquor), precipitation pH conditions (3, 9, and 9→3), and black liquor dilution concentrations (10, 20, and 30%). By comparing the effects of these conditions on lignin recovery yield, ash content, purity, and molecular weight distribution, this study aimed to determine the optimal conditions for efficient lignin recovery and high-quality fractionation.
2. Materials and Methods
2.1 Materials
Black liquor derived from hardwood kraft pulping, with a solid content of approximately 36 ± 2%, was supplied by Moorim P&P (Ulsan, Korea) and used in this study. For lignin recovery, 95.0% sulfuric acid (Daejung Chemical & Materials Co., Ltd., Korea) was employed.
2.2 Recovery of lignin from black liquor
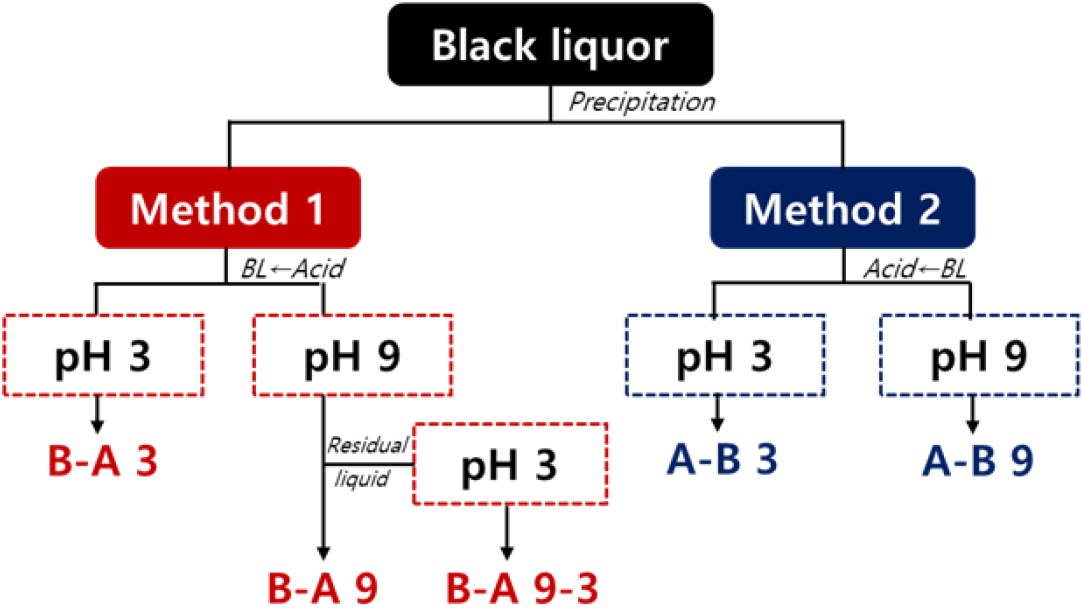
Lignin was fractionated from kraft black liquor under various precipitation conditions, and the detailed procedures are summarized in Fig. 1 and Table 1. For Method 1, sulfuric acid was added gradually to 1 L of black liquor at room temperature until the target pH was reached, followed by stirring at 70°C for 1 hr. The formed precipitate was collected by vacuum filtration, washed with distilled water until the filtrate became neutral, and then dried at 50°C. In addition, to compare the effect of black liquor dilution, the black liquor concentration was adjusted to 10% and 20%, and the same precipitation procedures of Method 1 (B-A 9 and B-A 9-3) were conducted. The resulting lignin was then compared with that obtained from the undiluted black liquor (approximately 30% solids).
Table 1.
Experimental conditions for lignin recovery according to the acid addition method and target pH
2.3 Measurements
The lignin recovery yield according to the fractionation method was calculated using Eq. (1). The purity of lignin was evaluated by determining the acid-insoluble lignin (Klason lignin) and acid-soluble lignin (ASL) contents according to the standard methods of TAPPI T 222 and TAPPI UM 250, respectively. Additionally, the ash content of the recovered lignin was measured according to TAPPI T 211. For molecular weight determination, the lignin samples were acetylated as follows. Dried lignin (100 mg) was dissolved in 5 mL of pyridine, followed by the addition of 5 mL of acetic anhydride. The mixture was then reacted for 12 hr. After the reaction was completed, the product was slowly added to 500 mL of distilled water maintained at 0–10°C to precipitate the acetylated lignin. The precipitate was then separated by centrifugation at 20,000 rpm, washed repeatedly with distilled water, and dried at 60°C for more than three days. The dried acetylated lignin (0.01 g) was dissolved in 100 mL of THF and analyzed using gel permeation chromatography (GPC; Alliance e2695, Waters, USA). The analysis was performed with Waters Styragel HR3, HR4, and HR5E columns and a refractive index (RI) detector, at 35°C with a flow rate of 1 mL/min. Calibration was conducted using polystyrene standards with molecular weights ranging from 1,000 to 4,000,000 g/mol.
3. Results and Discussion
3.1 Effect of acid addition sequence and pH conditions
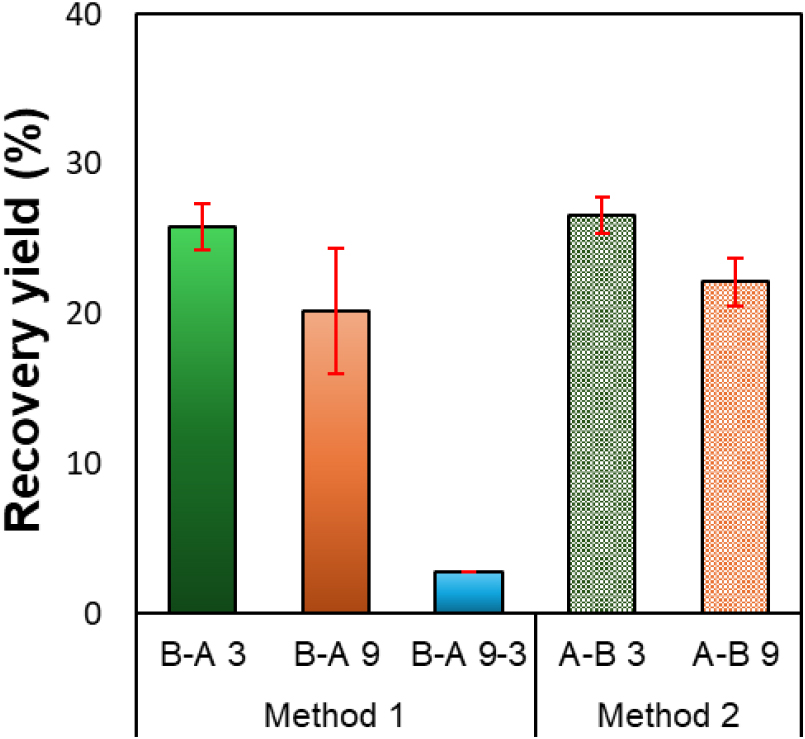
The lignin recovery yields according to the sulfuric acid addition sequence and pH conditions are presented in Fig. 2. The recovery yields of samples acidified and precipitated at pH 3, B-A 3 and A-B 3, were 25.8% and 26.6%, respectively, showing relatively high values. In contrast, samples precipitated at pH 9 (B-A 9: 20.2% and A-B 9: 22.1%) exhibited lower recovery yields. These results were consistent with the findings of Watumlawar and Park [10].
In general, lignin acquires negative charges at high pH (alkaline conditions) due to the ionization of phenolic hydroxyl (–OH) and carboxyl (–COOH) groups, resulting in electrostatic repulsion that maintains colloidal stability. Conversely, as the pH decreases, hydrogen ions (H+) neutralize the negative surface charges of lignin, resulting in a sharp reduction in electrostatic repulsion. Consequently, hydrophobic interactions and hydrogen bonding between lignin molecules are strengthened, promoting aggregation and precipitation [13,14].
Meanwhile, the two-step acidification sample (B-A 9-3) exhibited a very low recovery yield of less than 3%. This result can be attributed to the fact that most lignin was initially coagulated and precipitated at pH 9, while the remaining dissolved lignin in the filtrate was only partially precipitated under the subsequent pH 3 condition. Therefore, the total recovery yield from B-A 9 and B-A 9-3 (23%) was approximately 3% lower than that obtained under the single pH 3 precipitation condition, indicating that a single-step low-pH acidification process is more efficient than stepwise acidification in terms of yield. In addition, the method in which black liquor was added dropwise to the acid solution (Method 2) showed 1–2% higher recovery yields compared with the method where acid was added to the black liquor (Method 1), confirming that the acid addition sequence also has a noticeable effect on lignin precipitation efficiency. The slightly higher recovery and purity observed for Method 2 may be related to differences in the acid–black liquor mixing behavior. When black liquor is added to the acid solution, localized acidification occurs more rapidly and uniformly, which can promote more efficient precipitation.
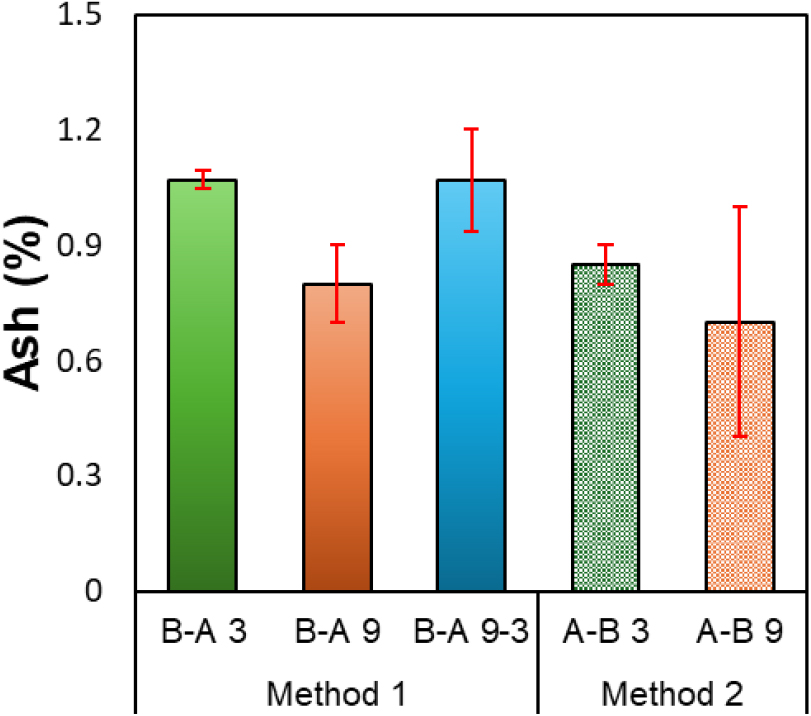
The ash content of lignin recovered under different precipitation conditions is shown in Fig. 3; all samples exhibited very low levels, below 1.1%. This is attributed to the presence of sodium and sulfur compounds originating from the pulping process, as well as trace amounts of natural inorganic elements (K, Ca) that are inherently present in the biomass and may remain within or on the surface of the lignin structure [15]. These inorganic residues can influence the purity and the thermal and chemical stability of lignin. In general, previous studies on wood-based industrial kraft lignin have reported ash contents in the range of approximately 0.5–2% [16,17]. The low ash content (~1%) observed in this study indicates that most inorganic impurities were effectively removed during the acidification and washing processes.
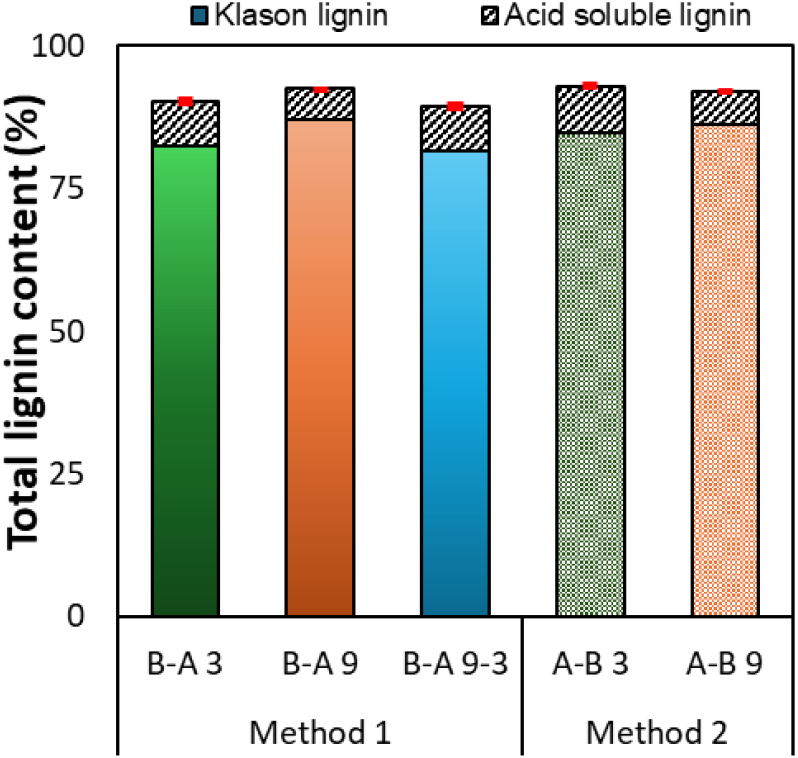
Klason lignin and ASL are key indicators for evaluating lignin purity [16]. The total lignin content of the recovered samples ranged from approximately 90–94%, indicating high purity under all conditions (Fig. 4). Even under the same pH 3 condition, the method in which black liquor was added to the acid solution (Method 2) showed about 3% higher total lignin content than the method in which acid was added to black liquor (Method 1), suggesting that Method 2 achieved higher purity. The ratio of Klason lignin to ASL varied slightly depending on the precipitation conditions. The ASL content of the samples precipitated at pH 9 was approximately 5–6%, whereas at pH 3, it increased to around 8%, indicating that a larger proportion of low-molecular-weight or water-soluble lignin fractions was present under stronger acidic conditions. These differences in purity and composition are likely to be reflected in the structural characteristics of the lignin, which were further examined through analysis of the molecular weight distribution of each sample (Table 2).
Table 2.
Molecular weight distribution of recovered lignin under different acidification methods and pH conditions.
| Sample | GPC | |||
| Mn | Mw | PDI | ||
| Commercial lignin | 2,722 | 4,894 | 1.798 | |
| Method 1 | B-A 3 | 2,290 | 3,597 | 1.570 |
| B-A 9 | 2,463 | 4,026 | 1.635 | |
| B-A 9-3 | 2,089 | 2,954 | 1.414 | |
| Method 2 | A-B 3 | 2,307 | 3,632 | 1.574 |
| A-B 9 | 2,360 | 3,767 | 1.596 | |
The number-average molecular weight (Mn) of the lignin recovered in this study ranged from 2,089 to 2,463 g/mol, and the weight-average molecular weight (Mw) ranged from 2,954 to 4,026 g/mol, which were lower than those of the commercial lignin (Moorim technical lignin) in all samples. For Method 1, the sample precipitated at pH 3 (B-A 3) showed an Mw of 3,597 g/mol, approximately 11% lower than that of the pH 9 sample (B-A 9). In particular, the two-step acidification sample (B-A 9-3) exhibited the lowest Mw (2,954 g/mol). This result can be interpreted as molecular weight-based fractionation, where relatively high-molecular-weight lignin was first precipitated at pH 9, and the remaining low-molecular-weight lignin in the filtrate subsequently precipitated at pH 3. The B-A 9-3 sample exhibited the lowest polydispersity index (PDI) (1.414), indicating that lignin with a more homogeneous molecular weight distribution was recovered.
For Method 2, the Mw values at pH 3 and pH 9 were 3,632 g/mol and 3,767 g/mol, respectively. While the pH 3 condition showed a similar Mw to that of Method 1, the pH 9 condition exhibited about 10% lower Mw. This can be attributed to a locally rapid decrease in pH when the black liquor was added to the acid solution, which caused partial cleavage of β–O–4 ether linkages under strongly acidic conditions (below pH 3), leading to lignin depolymerization [18,19]. In addition, the PDI values at pH 3 (1.41–1.57) were lower than those at pH 9 (1.60–1.64), indicating that lignin recovered under alkaline conditions possessed a relatively heterogeneous molecular weight distribution.
3.2 Effect of black liquor dilution concentration
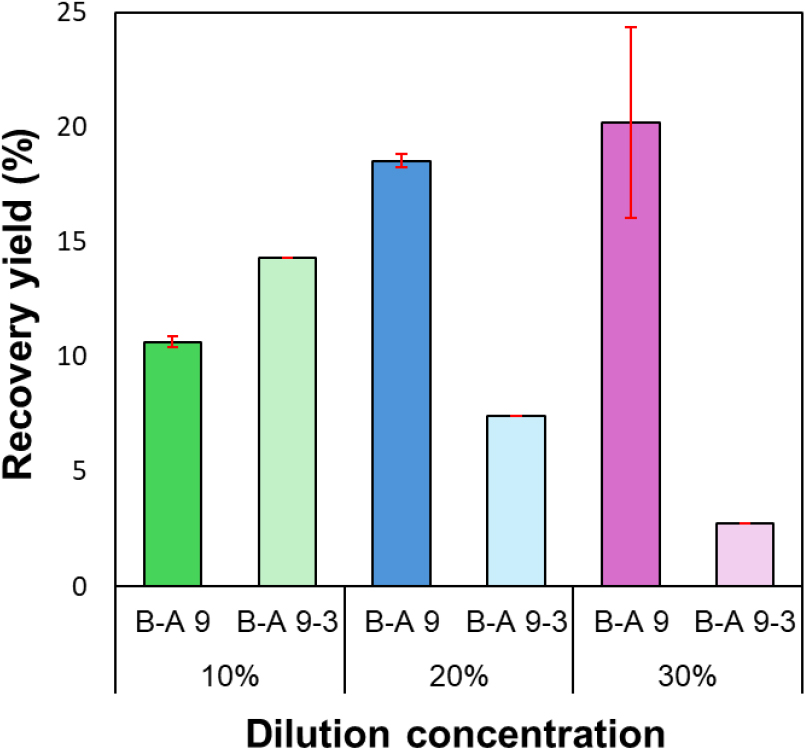
In the lignin precipitation process, the dilution concentration of black liquor is a key variable that directly influences the reaction rate of acidification, ionic strength, and the aggregation behavior of lignin particles [20]. Analysis of lignin recovery yield, ash content, and purity as a function of black liquor dilution ratio (10–30%) revealed distinct variations in precipitation efficiency and purity. As the dilution concentration increased (10 → 30%), the lignin recovery yield changed noticeably (Fig. 5). Under the 10% dilution condition, the recovery yield of the B-A 9 sample was relatively low at approximately 10.6%, whereas at higher black liquor concentrations of 20% and 30% under the same precipitation condition (pH 9), the yields increased to 18.5% and 20.2%, respectively. This trend aligns with previous studies [20], which have reported that higher ionic strength in concentrated solutions reduces the repulsive range between lignin molecules, allowing attractive forces to dominate and promoting aggregation.
In other words, excessive dilution increases the distance between lignin particles, thereby lowering the collision frequency and reducing precipitation efficiency. Conversely, at moderate concentrations (20–30%), high-molecular-weight lignin can efficiently aggregate and precipitate. Meanwhile, the two-step precipitation sample (B-A 9-3) exhibited lower recovery yields of 7% and 2% at 20% and 30% concentrations, respectively. This can be attributed to the fact that most lignin had already precipitated during the first pH 9 stage, and only a small portion of the remaining low-molecular-weight lignin further precipitated in the second pH 3 stage. In contrast, under the 10% low-concentration condition, the recovery yield was approximately 35% higher than that at pH 9. This can be explained by the rapid acidification to pH 3, which increased the ionic strength of the solution and effectively neutralized the negative charges on the lignin surface, promoting aggregation. Thus, although dilution increases the distance between lignin particles, in a strongly acidic environment (pH 3), the abundant H+ ions rapidly protonate phenolic –OH and –COOH groups of lignin, leading to effective precipitation of low-molecular-weight lignin that remained dissolved under pH 9 conditions [11].
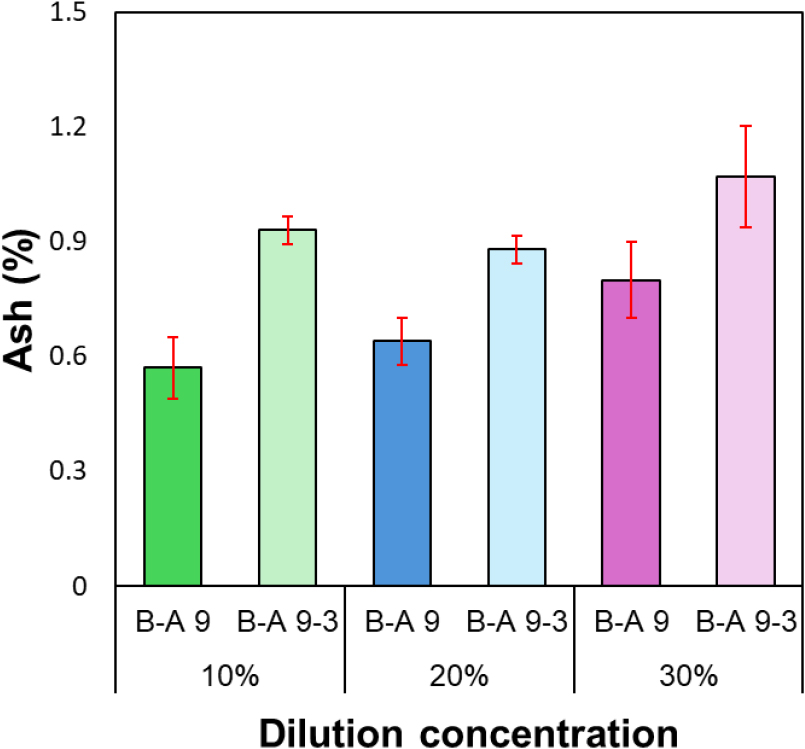
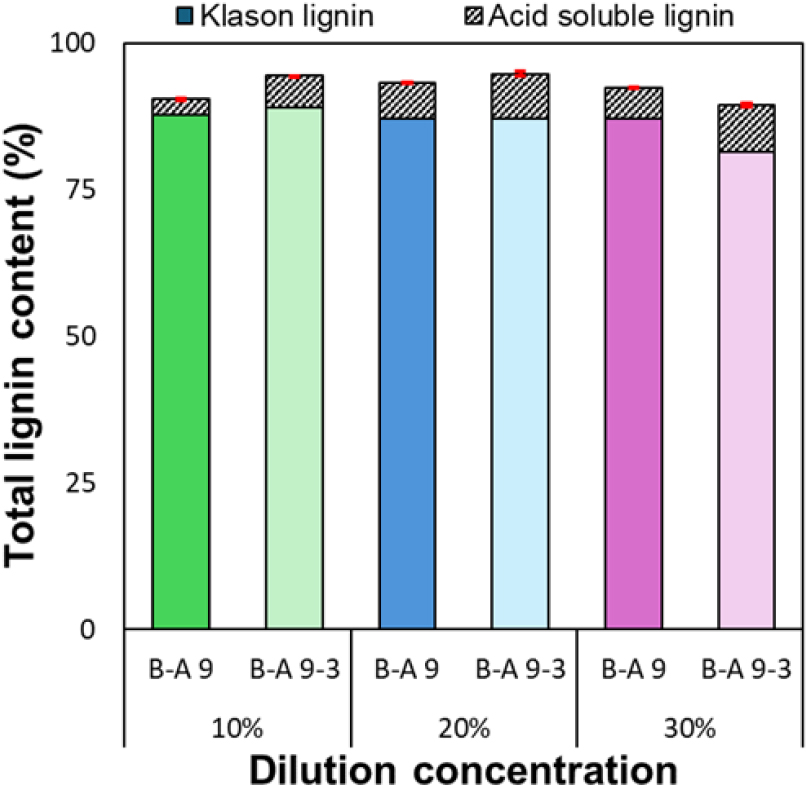
The ash content of lignin recovered under different dilution concentrations (Fig. 6) remained consistently low, ranging from 0.6% to 1.1%. Similar to the results presented in Section 3.1, this indicates that inorganic impurities were almost completely removed under all conditions, confirming that high-purity lignin could be effectively recovered. The total lignin content obtained under different dilution concentrations (10–30%) (Fig. 7) ranged from approximately 89.4% to 94.7%, maintaining an overall high level of purity. Lignin recovered at a 20% dilution concentration exhibited the highest purity among all conditions, with 93.2% (B-A 9) and 94.7% (B-A 9-3), respectively. This result suggests that at an intermediate concentration—neither excessively low (10%) nor overly high (30%)—a balance between ionic strength and viscosity during the acidification process allows lignin aggregation to occur most stably.
Analysis of the molecular weight distribution of lignin at different dilution concentrations (Table 3) showed that lignin recovered under the 10% dilution condition (B-A 9) exhibited the highest Mw (4,894 g mol-1) and PDI (1.77), indicating heterogeneous precipitation behavior in which both high- and low-molecular-weight lignin fractions coexisted during aggregation. In contrast, the 30% B-A 9 sample exhibited lower Mw and PDI values of 4,026 g mol-1 and 1.64, respectively. This can be attributed to the reduction in solution viscosity and decreased collision frequency between lignin particles caused by dilution, which suppressed the aggregation of low-molecular-weight lignin.
Table 3.
Molecular weight distribution of lignin recovered under different dilution concentrations
Meanwhile, the two-step acidification samples (B-A 9-3) under the same conditions exhibited Mw values of 2,954 g mol-1 and 3,586 g mol-1, with PDI values ranging from 1.41 to 1.50, indicating that lignin recovered under lower pH conditions consisted mainly of low-molecular-weight fractions and showed a relatively uniform molecular weight distribution regardless of dilution concentration.
These results suggest that the molecular weight and homogeneity of lignin can be controlled by precipitation pH, acid addition sequence, and black liquor dilution ratio, implying that low-molecular-weight lignin (for chemical modification, adhesives, or resin additives) or high-molecular-weight lignin (for electrospun fibers or composite materials) can be selectively isolated depending on the intended application.
4. Conclusions
In this study, lignin was recovered from hardwood kraft black liquor, and the effects of acid addition sequence, precipitation pH, and dilution concentration on recovery efficiency and lignin properties were systematically analyzed. Regardless of the recovery conditions, all lignin samples exhibited ash contents below 1%, indicating that most inorganic impurities were removed and that high-purity lignin (approximately 90%) was successfully obtained.
Lignin recovery yield was higher under strong acidic conditions (pH 3) compared to alkaline conditions (pH 9), and Method 2 (dropwise addition of black liquor into the acid solution) achieved approximately 1–2% higher yield and purity than Method 1. Additionally, molecular weight analysis showed that Mw decreased and PDI values were lower under pH 3 conditions, suggesting that precipitation pH and acid addition sequence influence lignin aggregation behavior and structural characteristics.
Among the dilution tests, the 20% concentration condition produced the highest purity (up to 94.7%) and the most uniform molecular weight distribution. These findings provide fundamental information for optimizing lignin recovery and support the potential use of the recovered lignin in functional materials such as biopolymer precursors, adhesives, and composite fillers. In addition, further experimental studies are needed to clarify how selective molecular-weight fractionation influences the structural characteristics of lignin.