1. Introduction
Organic cultural heritage objects account for 28.5% of national treasure and 44.1% of treasure in Korea, and the importance of its preservation is recognised. In comparison with mineral cultural heritage objects composed of metal, stone, or earthenware, the organic cultural heritage articles, made of fabric, paper, and wood, are intrinsically more apt to be harmed by microorganisms and insects. These damages to the organic cultural heritage are a very serious problem, because they not only sully the aesthetic value of cultural heritage objects by contaminating the appearance or blemishing prints or patterns but also influence the structure by destroying the constituting material.
Recently, the need to conserve wooden cultural heritage objects has raised public concern. Most of historic wooden architectures in Korea are consist of pine (Pinus densiflora) trees, and then many species are zelkova (Zelkova serrata) trees.1) Wooden architecture would be exposed to outdoor environment, and no sufficient data showed effective preservation methods against dangerous factors. Wooden cultural heritage articles are easy to destroy, and the damage progresses because insufficient data about effective preservation methods against dangerous factors, especially fungi.
Wood-decay fungi pose a serious danger to wooden cultural heritage objects. To prevent the damage, chemicals have been employed as preservatives, e.g. the gas mixture of ethylene oxide and methyl bromide is widely used to avert germination.2) However, when this gas mixture is used in preserving wooden cultural heritage objects, it may cause discoloration and peeling of pigments and dyes.3) Furthermore, it is a serious health and environmental hazard. Therefore, the demands for preservatives based on natural substances are increasing. Numerous studies have been conducted in search of new antifungal agents among plant secondary metabolites instead of synthetic chemicals.4)
Wood vinegar, which is a liquid by-product of charcoal production by burning wood, is well known for its antibacterial and antifungal efficiency.5-7) In Korea, wood vinegar is one of commonly used folk medicines for preventing and curing skin diseases such as tinea pedis (athlete’s foot). It has also been reported that the primary components of wood vinegar have insecticidal activity against termites, the attackers of wooden cultural heritage objects.8) The traditional and academic applications of wood vinegar suggest that it can be used as a natural fungicide in agriculture and food preservation.9) However, no study has reported the use of wood vinegar in the field of cultural heritage conservation.
In this study, we aimed to evaluate the fungal inhibitory effect of wood vinegar in wood against wood-decaying fungi, white rot fungi (Trametes versicolor) and brown rot fungi (Tyromyces palustris). The concentration of wood vinegar is an important factor that affects the final properties of the treated material, especially defence effect for penetration of fungus in pine wood. The preservative effect of pine wood blocks was confirmed by scanning electron microscope (SEM) analysis. We observed the erosion and thinning of cell walls caused by the growth and penetration of mycelia in pine wood blocks by SEM observation. We focused on the inhibition effect against fungal decaying as wood preservatives of wood vinegar.
2. Materials and methods
2.1 Wood vinegar
Wood vinegar was purchased from Soochonimsan Inc. (Seongju city, Korea). The wood vinegar (20 L) was filtered with filter paper (Whatman No. 2) and concentrated in vacuum at 25°C using a rotary vacuum evaporator (EYELA, Japan). The wood vinegar extract (68.64 g) was diluted with water (2 L), and then it was partitioned with organic solvent of methylenechloride (MC). Organic solvent was dried using a rotary vacuum evaporator. The extract (MC) was dissolved with ethyl alcohol and used as samples for the antifungal activity tests.
The main component analysis of methlyenechloride extract was performed by gas chromatography mass spectrometry of an Agilent model 6890 II series gas chromatograph linked to an Agilent model 5973N selective quadrupole mass detector. Separation was performed on HP-5MS capillary column (30 m × 0.25 mm i.d., 0.25 μm film thickness, J&W Scientific) for all components. The maximum temperature applied in run for this column was 280°C. The temperature of injector and interface were held at 250°C and helium was used as carrier gas under constant flow (1 mL/min). Oven temperature increased linearly at 4°C/min from 50°C to 250°C. The sample was injected in triplicate.
The MC extract was used with various concentrations for decay test. Since then wood vinegar of decay test represents the MC extract. Ethyl alcohol was used as a control.
2.2 Fungal strains
In this study, Trametes versicolor and Tyromyces palustris, which were isolated from the Janggyeong Panjeon in Haeinsa temple, were cultured to obtain multiple fungi samples. The Janggyeong Panjeon contains the Tripitaka Koreana (Palman Daejanggyeong) made of wood. The cultivation of the fungi species was conducted at 28°C of air temperature on Potato Dextrose Agar (PDA, Difco, USA) medium.
2.3 Preparation of test blocks
For wood decay test, test blocks were cut from sapwood of Pinus densiflora and Zelkova serrata Makino, because the wood of this species is most frequently used in the wooden cultural heritage objects in Korea and this wood is susceptible to white rot fungi and brown rot fungi. A sample size was prepared of 10 mm × 10 mm × 10 mm. P. densiflora and Z. serrata specimens were dried for at least 1 week at 105°C to ensure that the wood samples were completely dry. The test blocks were placed into a desiccator, and a vacuum of 100 mbar was applied to remove the majority of air trapped within the wood blocks. The conditioned wood blocks were placed in a glass beaker was filled up with wood vinegar solution and the wood vinegar solution used five different concentrations. The glass beakers were situated inside desiccator connected with a vacuum pump and progressed a process including an initial vacuum phase of 100 mbar for 1 hour and a pressure phase of 12 bar for 3 hours. After submersion, these blocks were dried at approximately 105°C for 48 hours in the oven and the weight was recorded until a constant weight was achieved.
2.4 Weight loss and SEM observation for decay test
The decay test was conducted by the American Society for Testing and Materials (ASTM D1413-99) method except that the specimens were 10 mm × 10 mm × 10 mm in size.10) Screw-capped culture bottles were filled with 120 g of soil mixture and 70 mL distilled water. Two feeder strips were placed directly on surface of the soil. The prepared bottles with slightly loosened lids were sterilized and autoclaved for 30 min at 121°C. The sterilized soil culture bottles were thoroughly cooled. The fungal inoculums (diameter, 8 mm pieces) from freshly grown culture were taken from the outer edge of mycelium and placed on the two edges of the feeder strip in sterilized culture bottles. They were incubated at 28°C and 70% relative humidity for approximately 3 weeks until the mycelia mat had covered the feeder strip. The wood blocks with cross section face down were placed on feeder strips in contact with mycelia in each culture bottle. The bottles containing the test blocks were incubated for a period of 15 weeks in an incubator maintained at 28°C and a relative humidity of about 70%, and in dark conditions. At the end of the incubation period the wood blocks were removed from the culture bottles, and the mycelium was carefully brushed off. The wood blocks were dried at room temperature for 4 days and then in the hot-air oven and weighed until the constant weight (W2) was obtained. The extent of fungal attack was determined by weight loss. The weight of wood blocks before exposure (W1) was recorded. Then, the wood blocks were removed from the culture bottles after 15 weeks and the weight of wood blocks after exposure (W2) was recorded. Percentage weight loss was calculated using Eq. 1 below:
After the fungal deterioration of wood blocks treated with the wood vinegar, wood blocks were fixed by immersion in 2% paraformaldehyde + 2% glutaraldehyde in 0.05 M cacodylate buffer (pH 7.2) at 4°C for 4 hours or overnight, and subsequently rinsed three times at 30 min intervals with 0.05 M cacodylate buffer (pH 7.2) solution at 4°C. They were washed at 30 min intervals with 30%, 50%, 75%, 90% and 95% alcohol series at room temperature. Wood blocks were cut to observe the morphological changes in growth and penetration of mycelia and avoid the effect of surface. Sections of samples were obtained by a rotary microtome and checked for their SEM compatibility using stereomicroscopy (Stemi-2000C, Zeiss). Thereafter, they were dehydrated in 100% alcohol and dried completely in a critical point dryer. The wood blocks were dried in a critical point dryer mounted on a metal stub and coated with gold. The microphotographs of wood blocks were obtained using JEOL JSM-5910LV (Japan).
3. Results and Discussion
3.1 Component analysis of wood vinegar
Wood vinegar represents the MC extract. The major components of wood vinegar were furfural (20.8%), guaiacol (19.2%), 4-methylguaiacol (15.3%), 5-methyl furfural (9.3%), and 4-ethylguaiacol (7.5%), by gas chromatography-mass spectrometry measurement (Table 1). The compounds are derivatives of furfural and guaiacol, and are the naturally occurring organic compounds. Furfural is a colourless oily liquid with the odor of almonds, which is quickly darken when exposed to air. Guaiacol is present in wood smoke, resulting from the pyrolysis of lignin. Also, it is darken upon exposure to air and light. Thus, wood vinegar could be used no more than 0.3 g/mL concentration due to their dark colour.
Table 1
The main components of wood vinegar (MC extract) though gas chromatography mass spectrometry analysis
3.2 The extent of deterioration caused by T. versicolor and T. palustris
The mean weight loss of wood blocks was shown in Table 2. The weight loss values indicate the extent of degradation that wood blocks are decayed by T. versicolor and T. palustris.
Table 2
Mean weight loss (±SD) for wood decay test of P. densiflora and Z. serrata blocks after 15 weeks treated with wood vinegar against T. versicolor and T. palustris
P. densiflora blocks treated with the highest concentration of wood vinegar recorded a mean weight loss about 14.5%, while those treated with 0.25, 0.10, 0.05 and 0.01 g/mL concentration recorded a mean weight loss of about 19.5, 33.8, 37.8 and 45.0%, respectively. Wood vinegar at 0.30 g/mL concentration provides 69.0% protection (14.5% mean weight loss), as compared to the control, where 47.0% mean weight loss was observed when exposed to T. versicolor.
Mean weight loss of 51.6, 43.7 and 41.1% was observed in P. densiflora samples treated with 0.05, 0.10 and 0.25 g/mL concentrations of wood vinegar, when these were tested against T. palustris. When blocks subjected to this fungus were treated with 0.30 g/mL wood vinegar, a mean weight loss of 19.3% was recorded, whereas in control blocks a 56.4% mean weight loss was observed.
In Z. serrata blocks, wood vinegar tested at 0.10 and 0.25 g/mL concentration against T. palustris recorded 64.9 and 56.0% mean weight loss, whereas at higher concentration, a 13.2% mean weight loss was caused by the test fungus. In control block, 73.7% mean weight loss was observed.
The highest concentration (0.30 g/mL) of wood vinegar provides 33.1% protection (54.4% mean weight loss), as compared to the control, where 81.3% mean weight loss was observed when exposed to T. versicolor.
Weight loss analysis of blocks decayed by T. versicolor and T. palustris revealed that treatment with wood vinegar significantly improved decay resistance. The data on statistical analysis revealed that wood vinegar provided protection in P. densiflora blocks against T. versicolor. A significant difference was observed in the percentage of protection provided by the highest concentrations of wood vinegar in both woods against both the test fungi. The extent of deterioration caused by T. versicolor and T. palustris in P. densiflora blocks was significantly different and that brown rot fungus caused more damage than did white rot fungus. Whereas, Z. serrata blocks didn’t showed remarkable difference that deterioration caused by both the test fungi.
3.3 SEM observation for the degree of degradation
The results are presented in Figs. 1-4. The experimental SEM data showed that only P. densiflora blocks treated with wood vinegar was protected against wood rot fungi for wood blocks of 3 weeks and 9 weeks. Because it was a significantly different in P. densiflora blocks, morphological analysis was conducted only on P. densiflora.

Fig. 1
Scanning electron microscopy micrographs of P. densiflora block decayed by T. versicolor for 3 weeks (A-D: 0.05 g/mL wood vinegar; E-H: 0.25 g/mL wood vinegar).
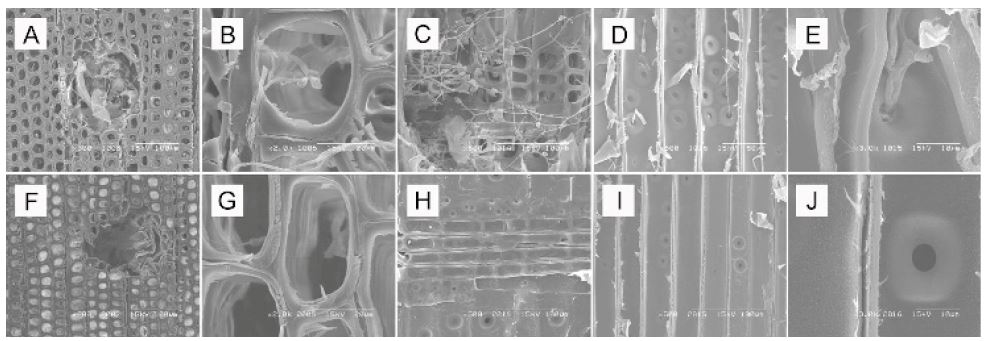
Fig. 2
Scanning electron microscopy micrographs of P. densiflora block decayed by T. palustris for 3 weeks (A-E: 0.05 g/mL wood vinegar; F-J: 0.25 g/mL wood vinegar).
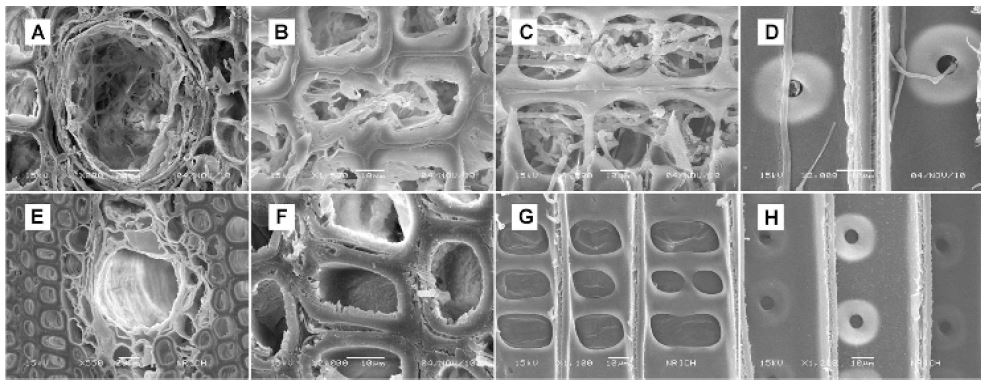
Fig. 3
Scanning electron microscopy micrographs of P. densiflora block decayed by T. versicolor for 9 weeks (A-D: 0.05 g/mL wood vinegar; E-H: 0.25 g/mL wood vinegar).
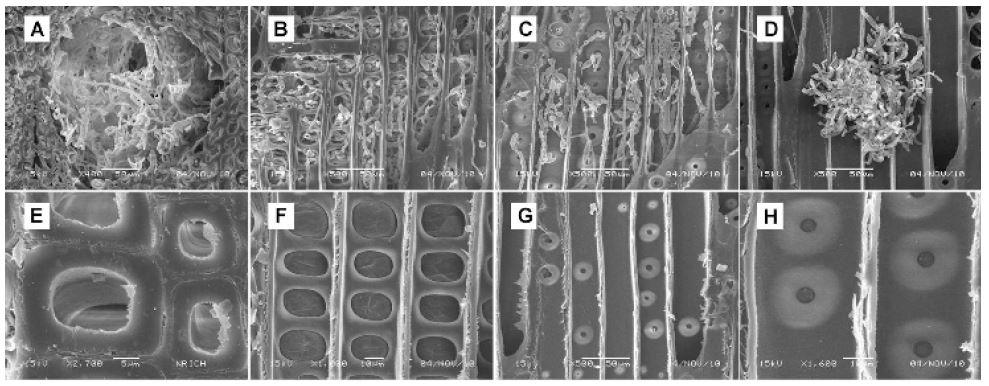
Fig. 4
Scanning electron microscopy micrographs of P. densiflora block decayed by T. palustris for 9 weeks (A-D: 0.05 g/mL wood vinegar; E-H: 0.25 g/mL wood vinegar).
Inhibition of the growth and penetration of mycelia was clearly affected by the concentration value of wood vinegar. Higher concentrations provided good fits to the experimental SEM data. In the control samples (treated with ethanol) subjected to the decaying effect of T. versicolor and T. palustris for 3 weeks, P. densiflora blocks were damaged by mycelia mainly in their resin canals and window-like shafts of the cross-field pitting. Degradation of the cell wall in cross section was also discovered. Application of 0.05 g/mL wood vinegar gave similar results to the ethanol control; no differences in morphology were observed. The concentration of 0.05 g/mL exerted only small antifungal effect on T. versicolor and T. palustris after 3 weeks of application (Figs. 1 and 2). The resin canals surrounded by epithelial cells in wood proved more susceptible to the fungal attack than the tracheid’s and were especially affected in their radial section. Mycelia penetration was also observed at the cell lumen in cross section. Ray parenchyma cells of the window-like pit were harmed, but there was no erosion or thinning of cell walls. Moreover, the growth and penetration of mycelia in cross-field pitting was observed. The full extent of the antifungal activity of the wood vinegar against T. versicolor and T. palustris after 3 weeks of incubation could be observed at a concentration of 0.25 g/mL (Figs. 1 and 2). Test wood blocks had normal structure, and no changes in micro -morphological characteristics were visible. Fungal contamination of the resin canals and ray parenchyma cells observed at the concentration of 0.05 g/mL was not detected at 0.25 g/mL. Thus, as the concentration of the wood vinegar was lowered, the growth and penetration of mycelia, not only on the surface but also in the inner parts of wood blocks, increased dramatically.
In ethanol control and the 0.05 g/mL samples incubated with T. versicolor and T. palustris for 9 weeks, the wood blocks were increasingly destroyed (Figs. 3 and 4). Although cell wall degradation was not seen in 3 weeks, it was clearly observed in 9 weeks. With progression of time, the cells around resin canals showed erosion and thinning. Ray cells were outstandingly penetrated by mycelia and the damage of cross-field pitting increased. The surface of blocks treated with the 0.25 g/mL wood vinegar for 9 weeks showed damage by T. versicolor and T. palustris (Figs. 3 and 4). However, the inner part of the wood blocks was protected against fungi. The surface was an easy attack target, as it was directly exposed to the fungi. Nonetheless, the wood vinegar inhibited the penetration and spreading of the fungi in the inner part of the wood blocks. Thus, no inner micro-morphological changes were observed in the wood blocks. After 15 weeks, further structural changes caused by T. versicolor and T. palustris in pine blocks were detected (data not shown). Mycelial damage in the vessels was surveyed in cross and ray section. The cell wall around the vessels was demolished and mycelial penetration was inspected. Once the mycelia penetrated the cell wall, they reformed as a sharp line. Under these conditions, cell penetration by the fungus may have been easy. After application of the 0.25 g/mL wood vinegar for 15 weeks, mycelia were formed and generated changes in wood structure of pine blocks (data not shown). The blocks are damaged. However, cell wall degradation by mycelia in cross section was not observed, and the mycelia did not penetrate into the inner part, as the outside resin canals and ray parenchyma cells were free of fungal growth. Thus, after 15 weeks of incubation, severe degradation and destruction of wood blocks was detected in case of the 0.05 g/mL concentration, whereas antifungal activity could still be observed in the 0.25 g/mL concentration of wood vinegar (Fig. 5).
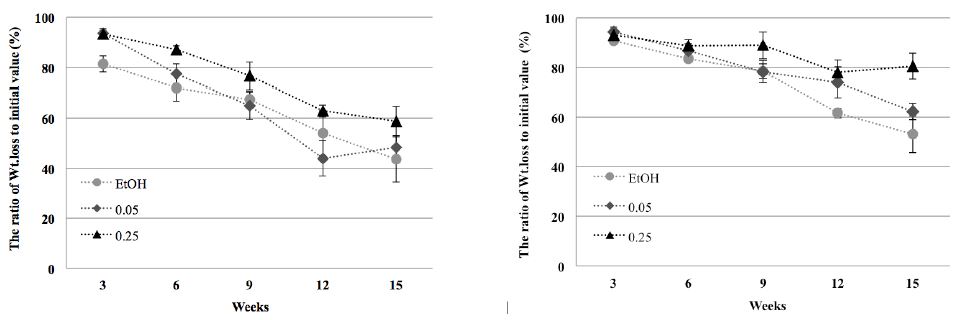
Fig. 5
The ratio of weight loss to initial value after 15 weeks against Trametes versicolor (A) and Tyromyces palustris (B).
Brown rot fungi are well known for their damaging effect on softwood. However, recent experiments have demonstrated that white rot fungi could also attack softwood and cause structural changes in wood. Therefore, we tried to verify the antifungal effect against white rot attack in pine blocks. The wood blocks analysed in our study were damaged by both T. versicolor and T. palustris. It was difficult to determine which fungus caused more serious destruction under SEM observation. However, the growth and mycelial penetration of T. versicolor was surveyed in pine blocks, and the fungal damage was inhibited by the 0.25 g/mL wood vinegar. Thus, our investigation clarified that softwood could be damaged by white rot fungi. Also, we confirmed that the wood vinegar had preservative effect of pine blocks against both brown rot and white rot fungi.
4. Conclusions
The preservative effect of wood blocks treated with wood vinegar has been investigated by numerous studies in diverse fields. Based on these reports, wood vinegar has been widely used as a bio-fertilizer and a preservative.11) An especially interesting recent study showed that the activity of neutralised wood vinegar with water extracts was responsible for fungal growth inhibition in wood elements and exerted insecticidal effect against termites.12,13) However, no research has been conducted on fungal damage to wooden cultural heritage objects. In this study, we evaluated the potentiality of wood vinegar as a useful preservative for wood used as the materials of traditional architecture in Korea. The wood vinegar was found to inhibit the erosion and thinning of cell walls caused by the growth and penetration of mycelia in pine blocks. The strong antifungal activity and the inhibition of wood cell destruction are important factors in the preservation of wood blocks. Pine block with the 0.25 g/mL concentration of wood vinegar did not show any substantial deviation from the normal structure after incubation with fungi for 9 weeks. Thus, we suggest that the wood vinegar should be developed as a resistance agent to prevent fungal deterioration of wooden cultural heritage objects.




