1. Introduction
Hydrogels have received a lot of attention in various fields due to their unique advantages, such as biocompatibility, higher water content, non-toxic characteristics, flexibility, soft material properties, and ease of handling.1,2) Hydrogels, including cellulose nanofibrils (CNFs) hydrogels, are based on natural materials and have been commonly used in the biomedical sector for a variety of purposes, such as drug delivery, tissue engineering, and biosensors. 1-6) CNFs are manufactured from cellulose fibers through mechanical treatments (blending, grinding, and homogenization), chemical pretreatments (carboxymethylation and TEMPO-mediated oxidation), and enzymatic hydrolysis.7-12)
Recently, studies have focused on CNF hydrogels that are introduced into charge screening by using metal cations to crosslink carboxylated CNFs.13,14) CNF gels are delicate and have low mechanical strength. However, the mechanical properties of CNF gels can be improved by adding metal salts. CNF gels form stronger networks when in an entangled structure due to their ability to form flexible nanofibrils. A bulk investigation is performed on the rheological properties of nanocellulose gel in combination with salt to control the mechanical properties of CNF gels. Tandjawa et al.15) reported an increase in the viscosity and mechanical strength of cellulose gels by adding 50 mM sodium chloride (NaCl) and calcium chloride (CaCl2). However, the mechanical properties of carboxymethylated cellulose gradually decreased from 0.1 to 10 mM.16) CNF gelation rapidly occurs with the addition of divalent (Ca2+, Zn2+, or Cu2+) and trivalent cations (Al3+, Fe3+).14) A higher cation valency enables a greater tendency for crosslinking between fibrils, creating a stronger nanofibril network structure. Furthermore, the same concentration of CNF (ranging from 0.01 to 3 M) containing CaCl2 shows a higher viscosity and storage modulus than when containing NaCl.17) The mechanical strength of the hydrogel can be controlled by adding salt. However, there is a research gap regarding the rheological properties of CNF gels prepared using divalent cations.
This study investigates the rheological properties of CNFs by using various concentrations of divalent cations to understand how divalent cations influence the CNF gel strength and the maximum concentration of cations that can be added to form the strongest gel.
2. Materials and Methods
2.1 Materials
In this study, dried kraft pulp (M. Company) was used to prepare CNFs. 95% Ethanol (EtOH; Samchun Pure Chemical Co., Ltd., Korea), 98% sodium hydroxide (NaOH; OCI Company Ltd., Korea), 98% monochloroacetic acid (MCA; Denak Co., Ltd., Japan), 99% barium chloride anhydrous (BaCl2; Junsei Chemical Co., Ltd., Japan), 98% magnesium chloride hexahydrate (MgCl2; Showa Co., Ltd., Japan), and calcium chloride dihydrate: extra pure (CaCl2; Junsei Chemical Co., Ltd., Japan) were used.
2.2 Preparation of CNF
CNFs were prepared according to the method reported by Wågberg et al.18) The fiber obtained from the dried kraft pulp (100 g) was suspended in a NaOH solution (14.11 g NaOH in EtOH) at room temperature for at least 1 h. The 0.6 mmol/g cellulose MCA solution was prepared by mixing monochloroacetic acid (28.9 g) and EtOH. The MCA solution was added to the suspension and reacted at 75℃ for 2 h. After a reaction time of 2 h, the fiber suspension was washed thoroughly using distilled water and filtered several times until neutral. The CNF was prepared by passing the cellulose fiber (2% wt) through a supermasscolloider (MKZA10-15IV; Masuko Sangyo, Japan) twice, and then homogenizing the fiber twice by using a GEA Homogenizer processor (Panda Plus, GEA, Italy).
2.3 Preparation of CNF gels
The CNF suspension (2%) was diluted using a divalent salt solution to obtain 1.5% CNF gel. The CNF gel was mixed with ionic crosslinking BaCl2, CaCl2, and MgCl2 to obtain various concentrations (100, 500, 1,000, 5,000, 8,000, and 10,000 ppm) for Ba2+, Ca2+, and Mg2+. After the salt was added, the mixtures were homogenized by hand, shaking approximately 5 times/s.
2.4 Rheological properties of CNF gels
The rheological properties of all the samples were determined using a rheometer (MCR 102; Anton Paar, Austria). A parallel plate (25-mm diameter) was used and the gap between both plates was set to 1 mm. The viscosity of the samples was measured at a shear rate ranging from 1 s-1 to 100 s-1. The strain sweep was used to determine the linear viscoelastic region at a frequency of 10 rad/s and over a strain range of 0.01-100%. The flow point (strain γf) was evaluated where the storage modulus equaled the loss modulus (G′=G″). The measurements were performed at 25°C.
3. Results and Discussion
The rheological measurements were studied to observe how the divalent salt content affected the properties of the CNF gels. The CNF gels were produced by introducing metal cations as crosslinkers to bind with the carboxylic groups, starting the gelation process. The bulk investigation was performed by adding metal salts to increase the strength of the hydrogel.11,15,17,19,20)
The effects of divalent salts were investigated using various divalent salts and concentrations. Detailed CNF gel compositions and divalent ions are listed in Table 1. The change in viscosity because of the influence of the divalent ions added to the CNF is summarized in Figs. 1-3. The divalent ions significantly affect the gel strength by increasing the viscosity. The viscosity of the hydrogel was increased because of the network structure formed by crosslinking the positively charged cation (added) and the negatively charged CNF.21) All the samples show typical shear thinning behavior with a decrease in viscosity when the shear rate increases. An increase in the shear rate leads to a gradual breakdown in the network, which results in a higher viscosity. However, the viscosity is relatively low when the shear rate is reduced because the interfibril network does not change as quickly.23)
Table 1.
Characteristics and compositions of cations in the hydrogel
Ionic radii were taken from a previous study.22)
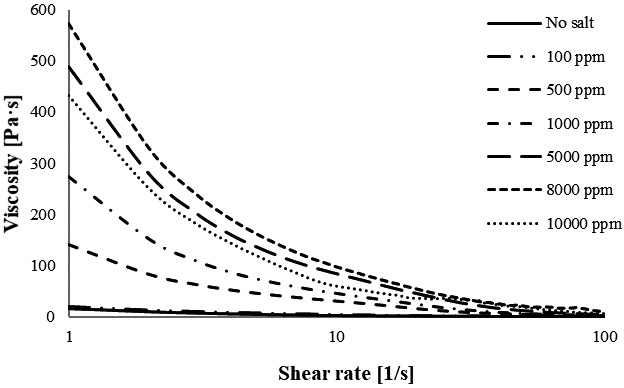
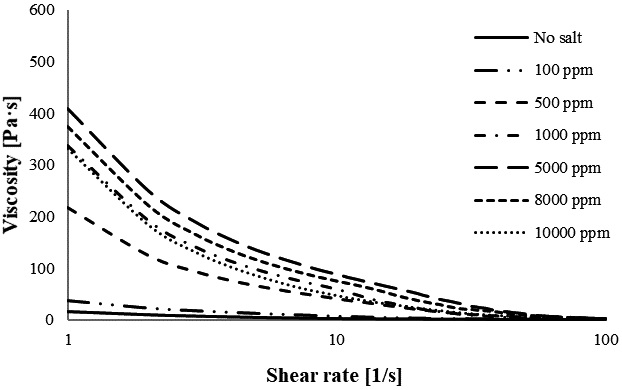
Fig. 1 shows the viscosity of CNF at various concentrations of Ba2+; the viscosity gradually increases by increasing the concentration of Ba2+ from 100 to 8,000 ppm. However, the continuous addition of Ba2+ from 8,000 to 10,000 ppm, decreases the viscosity. The viscosity increases from 19.89 Pa·s (100 ppm Ba2+) to 572.16 Pa·s (8,000 ppm Ba2+). Fig. 2 displays the viscosity of CNF and Mg2+ at concentrations ranging from 100 to 10,000 ppm. Fig. 2 indicates that the viscosity increases as the concentration of Mg2+ increases from 100 to 5,000 ppm and then decreases as the concentration of Mg2+ increases from 8,000 to 10,000 ppm. This trend shows that the highest viscosity of CNF containing Mg2+ is 5,000 ppm. Mg2+ allows for the generation of higher viscosities than Ba2+ between 100 and 1,000 ppm; however, at 5,000 ppm, Ba2+ has a higher viscosity than Mg2+.
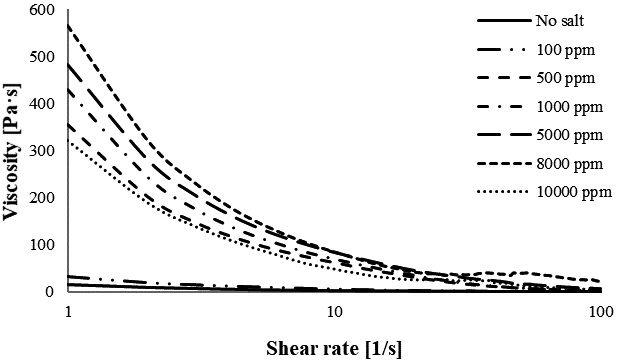
The apparent viscosities of CNF with various concentrations of Ca2+ are shown in Fig. 3. Fig. 3 shows a trend similar to that observed for Ba2+. The increase in Ca2+ from 100 to 8,000 ppm increases the viscosity of the gel; after the increase to 10,000 ppm, the viscosity decreases. This also indicates that 8,000 ppm of Ca2+ leads to a stronger hydrogel. These results show that the viscosity of CNF containing a divalent ion improves progressively when the concentration is increased from 100 to 8,000 ppm and, thereafter, starts to decrease when adding cations up to 10,000 ppm. Overall, an increase in the cation concentration leads to an increase in viscosity until the CNF gel ceiling is reached, causing the CNF to start to coagulate and lose viscosity. A similar result is observed while gradually increasing the CaCl2 concentration from 0 to 3 M; the viscosity of CNF gel gradually increases too.17) Liu et al.21) reported that a significantly higher viscosity is obtained when using a more concentrated crosslinked hydrogel. However, an excessive cation content in hydrogel reduces the strength of the hydrogel.
In this study, Mg2+ had the highest viscosity, Ca2+ the second-highest, and Ba2+ the lowest, at 100 ppm. This might be attributed to the difference in cation radii, where Mg2+ has a smaller radius compared to Ca2+ and Ba2+. CNF containing Ca2+ has a higher viscosity than Mg2+ and Ba2+ (Ca2+>Mg2+>Ba2+) at concentrations of 500 and 1,000 ppm. At higher concentrations (5,000 to 8,000 ppm), Ba2+ had the highest viscosity, Ca2+ the second-highest, and Mg2+ the lowest. These trends indicate that the impact of divalent ions on the CNF viscosity is directly correlated with the kind of cation used, shear rate, and concentration of cation. In addition, the critical ionic strength of the cellulose suspension depends on the charge content, mass fraction and dimension of the fibril.11)
Similar to what was observed for viscosity, adding divalent ions increases the storage modulus and loss modulus of the CNF gels (Figs. 4-6). The effect of ionic strength on the storage modulus and loss modulus of 1.5% CNF caused by adding Ba2+ is shown in Fig. 4. The G′ value increased from 417.9 to 17,334 Pa as the concentration of Ba2+ increased from 100 to 8,000 ppm, confirming that a stronger network of CNF gel is created at 8,000 ppm. However, when Ba2+ surpassed 8,000 ppm, the G′ value decreased. This suggests that by increasing the concentration of cations, the gel stiffens and becomes stronger until the CNF gel ceiling is reached; further increase in the concentration decreases the strength of the hydrogel. The stiffening of the gel is caused by the intimate contact between nanofibrils due to the increased cation content in the CNF gel. This removes water from the network, making it stronger.16) A similar trend is observed when Ca2+ is added. The gel strength increases as the concentration of cations are increased from 100 to 8,000 ppm, after which the strength decreases as the concentration is increased to 10,000 ppm (Fig. 5). The presence of Ca2+ ions in the CNF induces screening on the charged surface of the fibrils because the Ca2+ ions are closely associated with the carboxyl groups.24) Gelation of CNFs occurs when 50 mM of Ca(NO3)2 is added. This has a significant effect on the viscoelastic properties, caused by network formation.14) Sim et al.17) reported that storage modulus and yield stress increase with increasing CaCl2 concentration, due to flocculation that results in the formation of a strong network in the CNF gel. Maestri et al.19) reported that the storage modulus increases as the CaCl2 concentration of cations increases from 10 to 100 mM in cellulose nanocrystals gel.19)
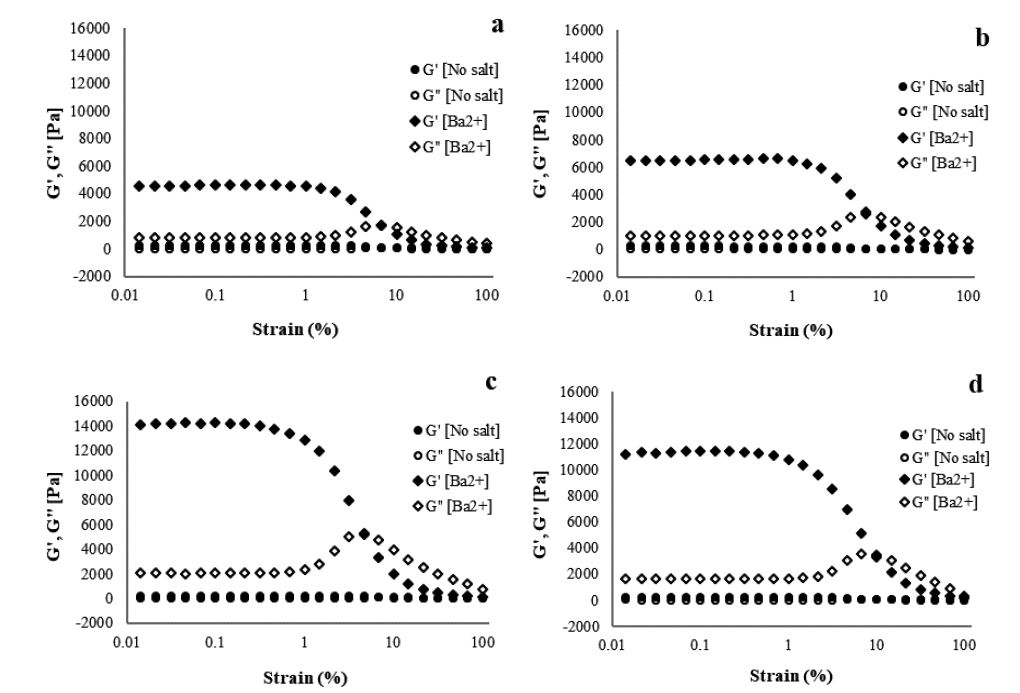
Fig. 4.
Storage and loss modulus as a function of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with Ba2+ concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
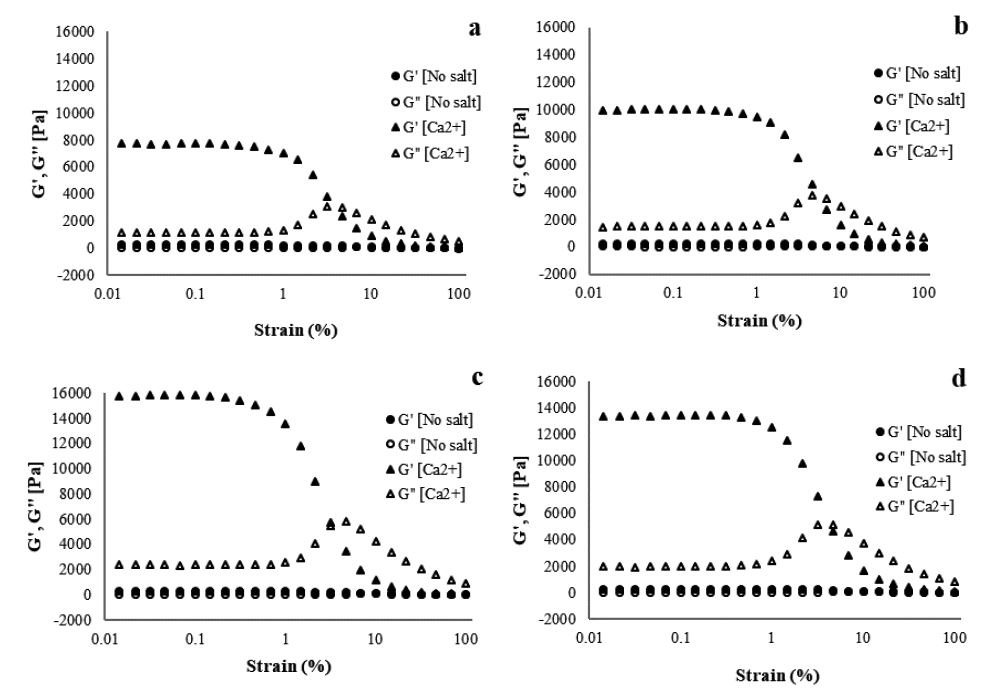
Fig. 5.
Storage and loss modulus as a function of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with Ca2+ concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
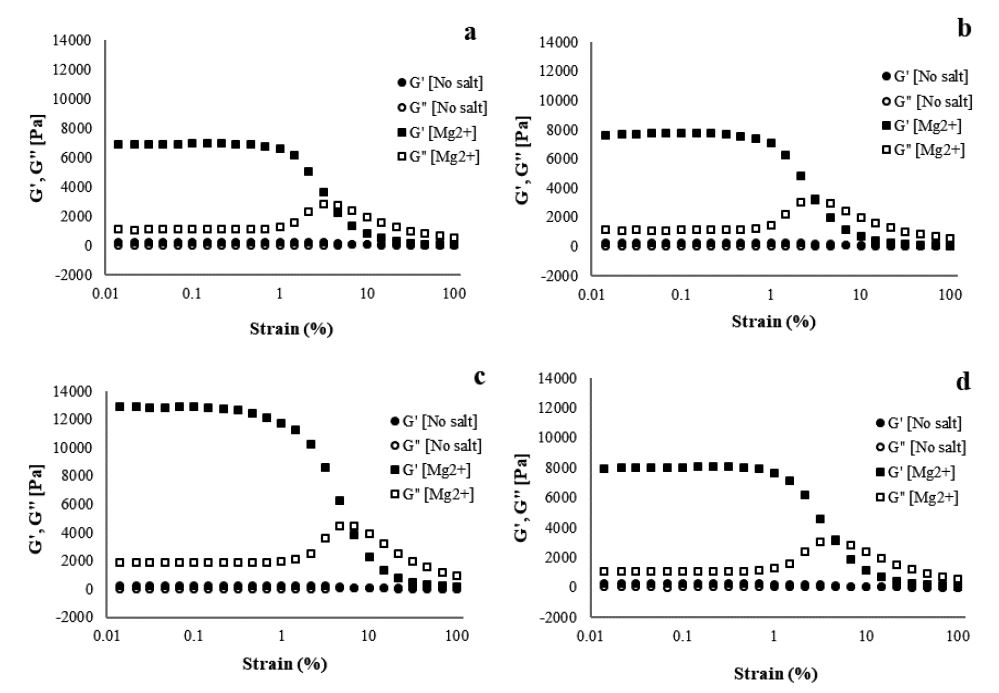
Fig. 6.
Storage and loss modulus as a function of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with Mg2+ concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
Fig. 6 shows the storage and loss modulus of the CNF gel as a function of strain amplitude for various concentrations of Mg2+. As shown in Fig. 6, the gel strength increases as the Mg2+ concentration is increased from 100 to 5,000 ppm, and reaches a maximum of 12,888 Pa, followed by decrease to a low of 10,134 Pa at 8,000 ppm. The Mg2+ concentration of 5,000 ppm formed the strongest gel (12,888 Pa). Storage moduli G′ as a function of different cation concentrations of Ba2+, Ca2+, and Mg2+ at a frequency of 10 rad/s is summarized in Fig. 7. As shown, Mg2+ has a more significant effect on the hydrogel strength than Ca2+ and Ba2+ (Mg2+>Ca2+>Ba2+) at low concentrations (100 ppm). This result can be explained by the difference in the ionic radii (Mg2+<Ca2+<Ba2+), as shown in Table 1, in which smaller ionic radii leads to a higher strength. Small ionic radii of Mg2+ implies a hard cation, which works better when increasing stiffness than soft cations such as Ca2+ and Ba2+. However, this observation is in contrast to the work presented by Chau et al.,20) who reported that storage and loss modulus of the CNF gels increase as the ionic radii of cation increase (Ca2+>Mg2+).
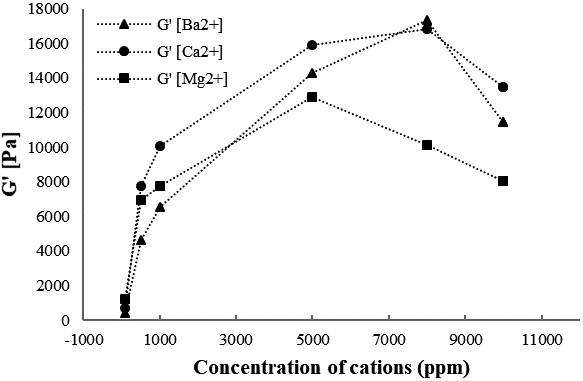
Fig. 7.
Storage moduli G′ as a function of different cation concentrations (Ba2+, Ca2+, and Mg2+) at a frequency of 10 rad/s and a strain of 0.1%.
Compared to Ba2+ and Ca2+, the increase in Mg2+ concentration from 5,000 to 10,000 ppm resulted in a lower-strength hydrogel. The strongest CNF gel containing Mg2+ was formed at 5,000 ppm Mg2+ concentration. The gel containing Ca2+ was stronger than the gel containing Mg2+ and Ba2+ between 500 and 5,000 ppm, and exhibited a maximum strength of 16,843 Pa at 8,000 ppm. Ba2+ produced the strongest gel (17,334 Pa) at 8,000 ppm (Ba2+> Ca2+>Mg2+). This was attributed to a stronger bridging of Ba2+ in CNF at high concentrations when compared to that of Mg2+ and Ca2+. This also indicated that MgCl2 is the most effective at increasing the hydrogel strength at low concentrations. A higher concentration of BaCl2 was required to trigger gelation in comparison with MgCl2 and CaCl2. The presence of cations as ionic crosslinking enhanced the mechanical properties of carboxymethylated CNF by creating intra/interfibril repulsion.14,25)
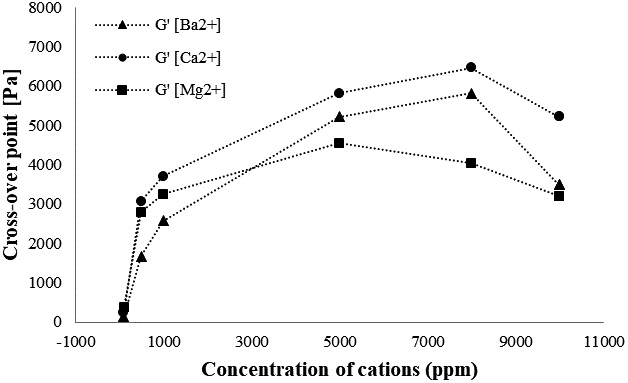
The flow point (γf) for all the samples at a frequency of 10 rad/s is shown in Fig. 8. The higher the crossover point value, the stiffer the sample. In Fig. 8, the value of the flow point increased as the concentration of the cation is increased, indicating that the sample becomes more rigid and strong as the concentration increases. However, a decrease is observed at 10,000 ppm after maximum strength is achieved. A cross over point at the strain sweep can be used to describe the flexibility of the samples. 26)
4. Conclusions
CNFs were modified by adding divalent cations (BaCl2, MgCl2, CaCl2) and using ionic crosslinking to form stronger gels at various concentrations (100, 500, 1,000, 5,000, and 10,000 ppm) of Ba2+, Mg2+, and Ca2+, respectively. As expected, the addition of cations and the increase in cation concentration significantly increased the viscosity, storage modulus, and loss modulus of the gels. However, the gel strength decreased after the maximum concentration was reached. Mg2+ had a greater effect on hydrogel strength when compared to Ca2+ and Ba2+ at 100 ppm (Mg2+<Ca2+<Ba2+), and achieved a maximum strength at 5,000 ppm. Ca2+ produced the greatest strength (Ca2+<Mg2+<Ba2+) between 500 and 1,000 ppm, and achieved a maximum strength at 8,000 ppm. Lastly, Ba2+ exhibited the highest strength at 8,000 ppm. Rheological properties of CNFs with divalent ions showed a positive correlation among viscosity, storage modulus, and loss modulus of the gels, which increased and decreased following the concentration trends of cations.