1. Introduction
The demand of renewable biomass has been increased to produce bio-energy and biomaterials with the issue of climate change and depletion of fossil fuels. Lignocellulosic biomass from agricultural residue and industrial waste is the one of promising substrate as a sustainable resource for bioconversion of biomass with several advantages. For instance, the biomass from residue and waste do not have to compete with the other use as food and also feasible to reduce the greenhouse gas (GHG) emission causing global warming.1,2) The application of paper mill waste stream as a biomass resource provides additional advantages. The cost of the biomass is low or negative since paper mills pay cost for landfill of waste fines. The supply of biomass is more stable as compared to other kinds of biomass because of independence of the waste by a region or season to harvest.3,4) The available amount of paper mill waste has been increased due to the demands of recycling papers and pulp.5) The paper waste and waste fines are more ready to be hydrolyzed without aggressive pretreatments.2)
According to the economic studies, conversion of waste fines in paper sludge to sugars further to produce biofuels and bioplastics provides benefits as compared to incineration and landfill of paper mill waste.6) The case study by Banerjee (2011), however, referred the limitations about the process which preferred feedstock as bleached short fiber with low ash content.6) The ash content could influence the product cost because the non-digestible component affecting on the waste treatment cost after hydrolysis. The results of economic analysis studies are understandable considering the inhibition effects of CaCO3 in the previous study which is deeply connect with the economic disadvantage. CaCO3 is also known as a strong inhibitor in enzymatic hydrolysis. The previous study revealed the three kinds of inhibitory effects of CaCO3 such as non-productive enzyme adsorption, pH level shift, and enzyme deactivation by Ca2+.7) The inhibition effects were significant as much as lignin in lignocellulosic biomass.
The present study would like to suggest a method of utilization of CaCO3 to reduce a cost of enzymatic hydrolysis yield of paper mill waste fines. The suggestion is basis of the role change of CaCO3 from a strong inhibitory chemical to a beneficial buffer material. The buffer substitution method was devised to reduce the cost of buffer and acid supply in hydrolysis process. The idea was based on utilization of CaCO3 as an useful material substituting acetate buffer in hydrolysis process. CaCO3 could be used for pH control and buffer solution in other applications.8,9)
2. Materials and Method
2.1 Materials
The waste fines were procured from a recycled linerboard-manufacturing mill in New York state. The waste fines were solid fraction of screwpressed sludge from rejects out of the whole paper mill process. The paper mill reject fine also can be classified as a dewatered cake from the waste water treatment process in the paper mill. The compositions of waste fines were analyzed to define the contents of carbohydrates, lignin and ash. The amount of carbohydrates and lignin were defined using NMR after hydrolysis with 72% H2SO4 according to the National Renewable Energy Laboratory (NREL) Laboratory Analytical Procedure (LAP, NREL/TP-510-42618, NREL/TP-510-42622). Weight 300 mg of sample and add 3 mL of 72% H2SO4 and incubated the sample for 60 minutes in water bath at 30°C. The acidity was diluted to 4% with 84 mL of deionized water. The samples were autoclaved for 60 minutes at 121°C. The hydrolyzed samples were prepared for nuclear magnetic resonance spectroscopy (NMR) analysis. Glucosamine, trimethylamine hydrochloride (TMA), and 3-(trimethylsilyl) propionic-2,2,3,3-d4 acid (TSP) were used for preparation of internal standard. Glucosamine (4.187%), TMA (0.228%) and TSP (0.053%) were dissolved in D2O (95.532%) after drying glucosamine, TMA and TSP in the vacuum oven at 65°C overnight. The NMR sample was prepared with 100 μL of internal standard solution and 900 μL of sample solution. Monosaccharides standard curves were prepared for glucose, xylose, arabinose, galactose, mannose and Rhamnose at different concentration of 1, 2, 4, 8, 16 g/L for each sugars.10)
2.2 Enzymatic hydrolysis
The hydrolysis was carried out in hydrolysis flasks placed in a shaking incubator (Reciprocal Shaking Bath 51221080, Precision Scientific Co., Denver CO). The waste fines were hydrolyzed at 50°C up to 72 h at 130 rpm. The cellulase enzyme used was commercially available Cellic CTec2 (Novozymes, USA) for the hydrolysis. Substrates are loaded as 5% consistency using 0.05M sodium acetate buffer (pH 5). Nonionic surfactant, 0.15% of Tween 80, was added to measure the effect of surfactant on the hydrolysis condition in different methods. The hydrolysate was separated with a 0.22 μm syringe filter and the liquid fraction was analyzed with NMR.10,11) The hydrolysis yield was described as the mass percentage of the total amount of monomeric sugar production from the raw material.
2.3 Buffer substitution and pH adjustment
The buffer material, sodium acetate, was replaced by CaCO3 existing in the raw material for the buffer substitution (BS) method. Paper mill waste fine was mixed with water as the ration of 1:20. Sulfuric acid or acetic acid (glacial) were added to the mixture to control the pH to 5.2. Another method, pH adjustment (PA), for pH control of conventional buffer solution was tested to compare the amount of acid demand compared to the BS method.
3. Results and Discussion
The composition of the linerboard paper mill waste fine was analyzed and described in the Table 1. The waste fine was composed by 32.0% of glucan, 12.1% of xylan, 3.8% of lignin, 41.3% of ash and 10.8% of other contaminants. The total amount of woody fiber was less than 50% of raw material due to the high portion of ash and contaminants by recycling and separation process. Byung-Gwan Jeon and Youn-Sop Kim (1992) reported the ash content in waste fines from a Korean paper mill as 23.67% and Gyeong-Youn Kim and others (2007) showed that the amount of ash in the three different kinds of paper mill waste fines were 32.99 – 61.79%.12,13) Ash contained significant amount of CaCO3 as much as 26.0% of the raw material. Contaminants included plastics, metals, fabrics, and other substances.
Table 1.
Compositions of waste fines rejected from a linerboard paper mill as a weight percentage of the raw material
| Glucan | Xylan | Lignin | Ash (CaCO3) | Others |
|---|---|---|---|---|
| 32.0 | 12.1 | 3.8 | 41.3 (26.0) | 10.8 |
Buffer solutions have been regarded as an essential additive for enzymatic hydrolysis process due to the specific activity of the enzyme affected by the environmental conditions such as temperature and pH. Fig. 1 shows the importance of buffer to get the higher hydrolysis yield. The sugar production yields drastically increased from 11.9% to 33.7% with addition of buffer. The effect of the surfactant can also be seen more clearly in the hydrolysis process with the buffer solution.
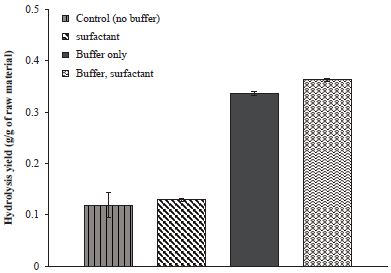
Fig. 1.
Enzymatic hydrolysis yields of waste fine depending on the addition of buffer and surfactant. The waste fine was hydrolyzed by 20
The presence of CaCO3 in the raw material raises the pH of the hydrolysis solution from pH 5 to pH 6.5-7.0. As a result, the activity of the enzyme reduced and hydrolysis yield decreased. The results for the PA method using acetic acid supports that optimization of pH is required for higher hydrolysis yield as showing the results in Fig. 2. When PA method applied using additional acetic acid to reach pH 5.2, the hydrolysis yield increased to 41.4%. The increased yield was 7.7% higher than compared to the hydrolysis using buffer only. The effects of surfactant were not significant in the PA method results.
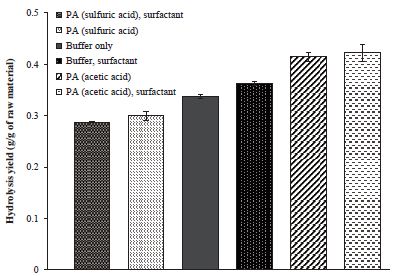
Fig. 2.
Enzymatic hydrolysis yields of waste fine using pH adjustment (PA) method. PA method applied sulfuric acid and acetic acid to maintain optimum pH shifted by CaCO3.
On the other hand, the PA method using sulfuric acid resulted a decrease in the hydrolysis yield rather than only using the buffer solution. The hydrolysis yield decreased from 33.7% to 28.8%. Even though the surfactant was added, the yield loss could not be prevented. From these results, it can be seen that sulfuric acid has a negative effect on enzyme activity. These negative effects are greater than the positive effects of pH control.
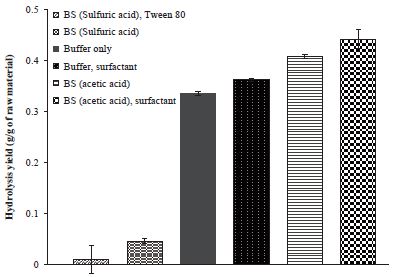
Buffer substitution (BS) using acetic acid also improved the hydrolysis yield like PA method (Fig. 3). The hydrolysis yield of waste fine increased from 33.7% to 40.8% when only acetic acid was added to the water and waste fine mixture to reach to pH 5.2. The degree of yield increase was very close to the result of PA. The effect of surfactant was observed as more effective in the BS method than PA method. The hydrolysis yield increased up to 44.2% by combination of BS method and addition of Tween 80.
BS method using sulfuric acid resulted negative effect as same as PA method. The yield reduction effects were much more significant than PA method. BS method with sulfuric acid decreased the yield to 1.0%. The result indicated that the enzyme activity decrease was more vulnerable to the sulfuric acid without buffer solution. From the results, sulfuric acid was not a proper chemical for pH adjustment of hydrolysis process.
Table 2 shows the amount of chemicals requiring for buffer and pH adjustment. Preparation of 0.05M of sodium acetate buffer solution (pH 5.0) required 8.2 kg of sodium acetate and 2.0 kg of acetic acid before adding substrate for 100 kg of waste fine hydrolysis. Additional acid was demanded as much as 6.3 kg of acetic acid for the solution optimized pH to 5.2 since CaCO3 in waste fines shifted pH level. The sum amount of acetic acid to get pH 5.2 of waste fines and buffer solution mixture was 8.3% (w/w) of waste fines. In contrast, BS method required only 7.7 kg of acetic acid for 100 kg of waste fine hydrolysis without sodium acetate supplement even including the pH optimization step. The BS method was possible to reduce the cost for 8.2 kg of sodium acetate and 0.6 kg of acetic acid for 100 kg (OD) of waste fine hydrolysis.
Table 2.
Requirement of buffer and acid amount for hydrolysis of paper mill waste fines
| Method | Buffer solution | Waste fine (kg, OD) | Buffer solution | pH adjustment to pH 5.2 | |
|---|---|---|---|---|---|
| Sodium acetate (kg) | Acetic acid (kg) | Acetic acid (kg) | |||
| PA | Sodium acetate | 100 | 8.2 | 2.0 | 6.3 |
| BS | Water | 100 | 0 | 0 | 7.7 |
The amount of acetic acid demand maybe still high for waste fine hydrolysis even with BS method requiring 7.7% (w/w) of acetic acid per raw material. The solutions were considered as setting the pH level a little bit higher than optimal pH for the hydrolysis process. According to the recent study, the elevated range of pH level between 5.2-6.2 was suggested to improve the hydrolysis yield of pretreated lignocellulosic biomass.14)
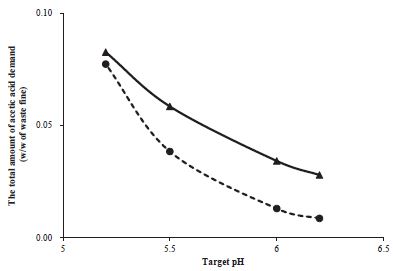
The total amount of acetic acid for PA method including buffer solution was 2.8%, 3.4%, 5.8% and 8.3% (w/w of waste fines) to reach of pH to 6.2, 6.0, 5.5, and 5.2 when 0.05 M sodium acetate buffer was used for hydrolysis (Fig. 4). In the case of BS method for water solution, the required amount of acetic acid was 0.9%, 1.3%, 3.8%, and 7.7% for the same pH levels. If operation pH is set as 5.5 from 5.2, the acetic acid demand reduces from 8.3% to 5.8% for PA method and to 3.8% for BS method. In the case of set point with pH 6, the requirement of acetic acid is only 3.4% for PA method and 1.3% for BS method which encouraged significant reduction of the operation cost with lower demand of acetic acid. The feasibility of the higher pH for BS method needs to be demonstrated for the waste fine hydrolysis further.
4. Conclusion
The new approach about the buffer substitution method was from an idea based on utilization of the major inhibitor, CaCO3, as a useful buffer material. The buffer substitution method requires none of sodium acetate and even less amount of acetic acid as compared to conventional enzyme hydrolysis using buffer solution and pH adjustment. Hydrolysis experiments resulted in almost same yields between traditional and the buffer substitution methods. Therefore, the buffer substitution could be applied to reduce the significant amount of cost of chemicals for the hydrolysis of paper mill waste fines. Acetic acid is more suitable for buffer substitution method than sulfuric acid.