1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Experimental methods
2.3 Analysis methods
3. Results and Discussion
3.1 Effect of the degree of amine substitution on the zeta potential and FT-IR of CNFs
3.2 Effect of the degree of amine substitution on the morphology of CNFs
3.3 Effect of the degree of amine substitution on the rheological properties of CNFs
3.4 Effect of the degree of amine substitution on the anion removal properties of CNF aerogels
4. Conclusions
1. Introduction
Cellulose nanofibers (CNFs) are natural nanomaterials obtained by the nanofibrillation of cellulose. Cellulose, synthesized by plants or bacteria, is biodegradable. CNFs, produced through chemical, mechanical, or enzymatic nanofabrication processes, also exhibit biodegradability1) and possess high tensile strength and tear resistance.2) Additionally, they have a high specific surface area and can be substituted with various functional groups at hydroxyl sites.3) They demonstrate excellent morphological and thermal stability.4,5) Based on these properties, research is actively being conducted across various fields, including biodegradable plastics, automotive interior materials, medicine, pharmaceuticals, cosmetics, and packaging.6,7,8,9,10)
CNFs are typically fabricated using mechanical treatments, such as nanogrinding, microfluidization, or high-pressure homogenization.11,12) In the case of nanogrinders, the cellulose suspension is passed between two grinders, applying shear forces that break the hydrogen bonds between cellulose molecules to produce CNFs. Microfluidizers and high-pressure homogenizers pass the suspension at high velocities through chambers with narrow microchannels, causing fragmentation. However, these nanofabrication devices consume significant amounts of energy, prompting the exploration of various pretreatment methods to mitigate this issue.13)
Pretreatment methods include mechanical pretreatment,14) enzymatic treatment,15,16) and chemical pretreatment.17,18) Mechanical pretreatment involves reducing fiber size by refining cellulose fibers before nanofibrillation, thus decreasing the energy requirements of subsequent processes.
Energy savings during nanofibrillation have also been achieved through chemical pretreatment by introducing functional groups. Chemical pretreatment modifies the surface of cellulose fibers using chemical agents. Representative anionic pretreatment methods, such as 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) oxidation and carboxymethylation, introduce carboxyl groups via oxidation or substitution reactions. TEMPO oxidation employs systems like TEMPO/NaBr/NaClO or TEMPO/ NaClO/NaClO2, where TEMPO is oxidized to nitrosonium ions that substitute hydroxyl groups at the C6 position with carboxylate groups.19,20) This modification improves water dispersibility and facilitates nanofibrillation through homogenization or ultrasonication. Carboxymethylated cellulose also yields transparent CNFs after nanofibrillation, with the resulting CNFs exhibiting transparency and mechanical strength depending on the degree of carboxyl group substitution and processing conditions.21,22,23,24)
Cationic pretreatment, in contrast, substitutes quaternary ammonium groups onto the hydroxyl groups of cellulose fibers, rendering the surface cationic. In 2011, Olszewska et al.25) reported the preparation of cationic CNFs. Common cationization agents include glycidyltrimethylammonium chloride (GMA) and 3-chloro-2-hydroxypropyltrimethylammonium chloride (CHPTAC).26,27) The quaternary ammonium groups introduced through these reactions confer antimicrobial resistance28,29,30,31,32) and promote adsorption of anionic contaminants,32,33) thereby expanding the application range of CNFs.
Freeze-drying cellulose nanofiber suspensions yields aerogels, where water is removed and replaced with air, creating highly porous structures. The surface properties of CNFs can be tailored through chemical modification, enabling control over electrostatic characteristics as well as fiber width and length. Consequently, the pore size of aerogels can be finely regulated, making them promising candidates for water treatment filters.33,34)
CNFs prepared by anionic chemical pretreatments, such as TEMPO oxidation or carboxymethylation, can adsorb cationic heavy metals like Fe, Cu, and Pb.33,34,35) Moreover, nanoscale CNFs significantly enhance heavy metal adsorption from wastewater.36) Their hydrophilic nature and ability to form uniform surfaces contribute to effective pollutant capture.37)
In addition to cationic contaminants, efforts have been made to capture anionic species using cationic CNFs. Halide ions such as Cl- and Br- are commonly found in industrial wastewater. Excessive concentration of these anions in aquatic environments can lead to ecological disturbances, inhibition of plant growth, and the formation of disinfection by-products (DBPs). Br- can react with chlorinated disinfectants to form carcinogenic brominated DBPs, highlighting the urgent need for their effective removal in water and wastewater treatment systems. Water treatment filters prepared from cationized CNFs have demonstrated the ability to capture anionic dyes and ions or to reduce the conductivity of aqueous solutions.38,39)
In this study, to investigate the anion capture performance based on the degree of quaternary ammonium pretreatment, varying amounts of GMA were added to cellulose fibers. The treated fibers were processed into cationic CNFs (CA-CNFs) using a high-pressure homogenizer. The resulting CA-CNFs were freeze-dried to fabricate cylindrical aerogel filters, which were then evaluated for their ability to capture Cl- and Br- ions. Anion capture was assessed by measuring the charge density of filtered solutions and through Energy-dispersive X-ray spectroscopy (EDS) analysis using field emission scanning electron microscopy (FE-SEM).
2. Materials and Methods
2.1 Materials
Hardwood bleached kraft pulp (Hw-BKP) was sourced from S Paper Co. (Korea). The pulp, derived from eucalyptus species, consisted of 79.5 ± 0.5% cellulose and 18.5 ± 0.5% hemicellulose, with minor amounts of lignin and ash. The brightness of the pulp was measured at 88.5 ± 0.3%.
For the quaternization pretreatment, GMA (C6H14NOCl, ≥90%, Sigma-Aldrich, USA), sodium hydroxide (NaOH, 98%, Samchun, Korea), and ethanol (C2H5OH, 95%, Samchun, Korea) were used.
2.2 Experimental methods
2.2.1 Preparation of quaternary ammonium cellulose
A total of 40 g (w/w) of Hw-BKP was treated with 9.88 g of NaOH as a catalyst and varying amounts of GMA (2.0, 3.0, and 4.5 mmol/g) for quaternization. Ethanol (500 mL) was used as the solvent. After the addition of NaOH and GMA, the mixture was stirred at 65°C for 2 h to initiate the quaternization reaction. The modified fibers were subsequently washed with distilled water until the pH of the wash water reached 7.
2.2.2 Production of CNFs
CNFs were produced using a grinder (Supermasscollider, MKZA10-15IV, Masuko Sangyo, Japan) operating at 1,000 ± 50 rpm with a disk gap of 200–250 µm. The pulp suspension was passed through the grinder four times. The resulting material was further processed using a high-pressure homogenizer (Panda PLYS 2000, GEA, Italy) at 1,200 ± 50 bar for five passes to obtain CNFs.
2.2.3 Production of quaternary ammonium cellulose aerogels
A 1 wt% suspension of quaternary ammonium CNFs was poured into cylindrical plastic molds (radius 1 cm, height 10 cm) and frozen at -40°C for 24 h. The frozen samples were then freeze-dried using a freeze dryer (FDU-1200, EYELA, Japan) to obtain aerogels.
2.3 Analysis methods
2.3.1 Characterization of quaternary ammonium CNFs
2.3.1.1 Statistical analysis
All experiments were conducted in triplicate to ensure reproducibility. Results are presented as mean ± standard deviation (SD). Where applicable, error bars representing SD are included in graphical data.
2.3.1.2 Zeta potential and particle size analysis
The zeta potential and particle size distribution of the quaternary ammonium CNFs were measured using a zeta potential analyzer (Zetasizer Nano ZS, Malvern Panalytical, UK). A 0.02% (w/w) dispersion was prepared in deionized water and briefly sonicated to ensure uniformity. Both analyses were conducted in appropriate measurement modes. The average zeta potential and hydrodynamic diameter were recorded.
2.3.1.3 FT-IR analysis
To confirm the chemical modification of the CNFs, Fourier transform infrared (FT-IR) spectroscopy was performed using an instrument equipped with an attenuated total reflectance (ATR) unit (Cary 670, Agilent Technologies, USA). Freeze-dried samples were analyzed over the range of 4,000–500 cm-1, and transmittance (%) spectra were recorded.
2.3.1.4 Viscosity analysis
The viscosity of the cellulose nanofiber suspensions was measured using a rheometer (MCR 102, Anton Paar, Austria) equipped with parallel plates (25 mm diameter). The gap between the plates was maintained at 1 mm. Measurements were taken over shear rates ranging from 1 to 100 s-1 at a constant temperature of 25°C, controlled by a Peltier system. The sample concentration was fixed at 1.0% (w/w), and measurements were performed in triplicate.
2.3.2 Evaluation of anion capture by quaternary ammonium cellulose aerogels
A 1,000 ppm anion solution containing Cl- and Br- was prepared by dissolving NaCl and KBr in deionized water. The solution was subsequently diluted to 500 ppm by mixing equal volumes of the 1,000 ppm solution and deionized water.
Filtration experiments were conducted by passing 500 mL of the 500 ppm solution through the prepared quaternary ammonium cellulose aerogel. After filtration, the aerogel was freeze-dried, and cross-sectional samples were collected. The distribution of Cl and Br ions was analyzed using EDS coupled with FE-SEM (Ultra Plus, Carl Zeiss, Germany).
Additionally, the charge density of the filtered solution was compared with that of the original 500 ppm solution. Samples (10 mL each) were titrated using a 0.001 N poly-diallyldimethylammonium chloride (Poly-DADMAC) solution, and the volume required to neutralize the charges was recorded.
3. Results and Discussion
3.1 Effect of the degree of amine substitution on the zeta potential and FT-IR of CNFs
Cylindrical aerogels for anion capture were fabricated using CA-CNF 1, CA-CNF 2, and CA-CNF 3 with varying degrees of quaternary amine substitution, as well as untreated CNF.
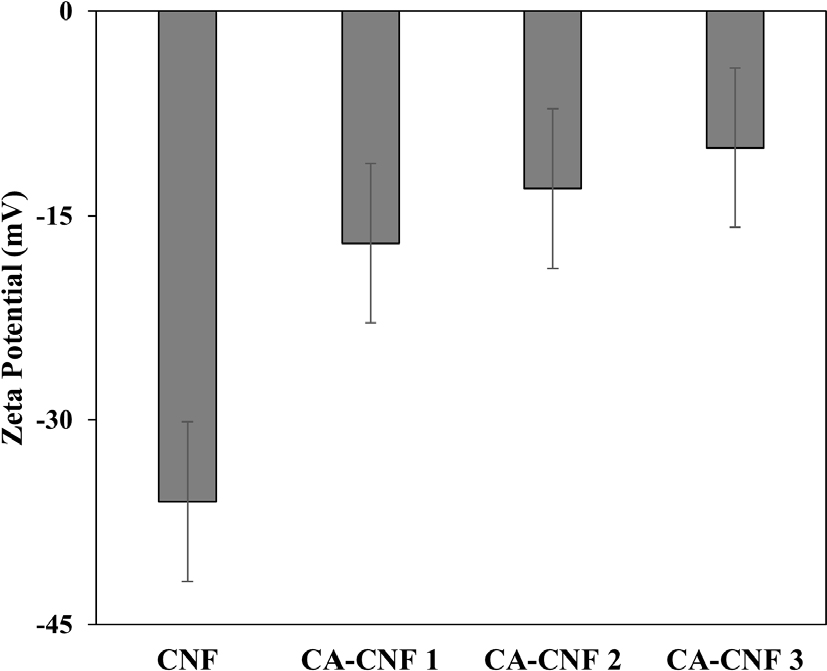
The zeta potential measurements of the samples are presented in Fig. 1. The untreated CNF exhibited a zeta potential of -36.4 mV, while CA-CNF 1, CA-CNF 2, and CA-CNF 3 showed progressively less negative values of -17.2 mV, -13.4 mV, and -10.6 mV, respectively. This trend confirms that the anionic nature of the CNFs decreased following quaternary amine treatment.
Previous research by Im et al.40) reported that increasing the amount of GMA enhances the charge density of pulp fibers, reflecting a higher incorporation of cationic functional groups. Similarly, in the case of TEMPO-oxidized CNFs, an increase in the degree of oxidation correlates with greater anionic group content.24)
These findings suggest that hydroxyl groups (-OH) on the CNF surface were partially substituted with quaternary ammonium groups through GMA pretreatment. As the GMA content increased, the degree of substitution rose accordingly, resulting in more cationic CNFs.
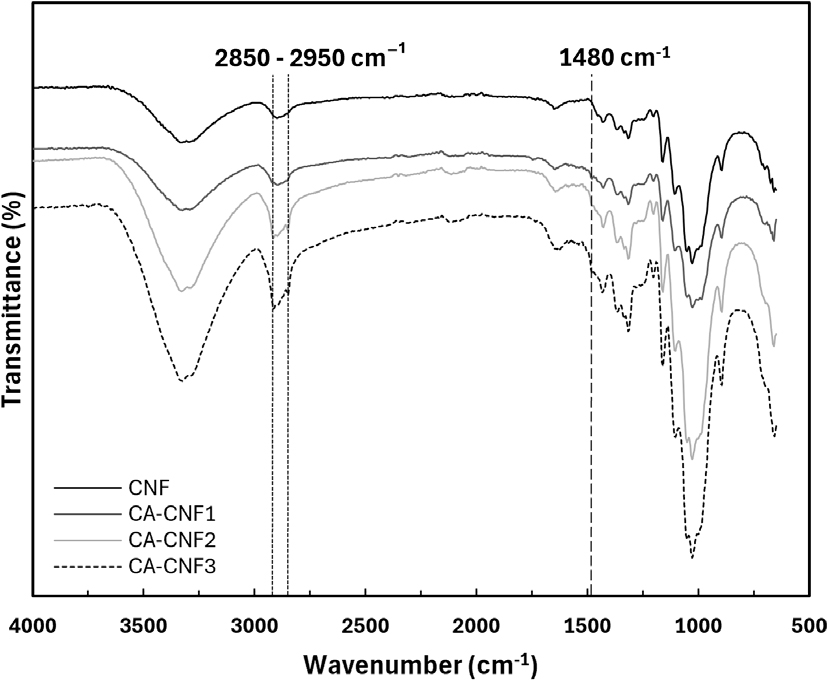
To further confirm the chemical modification of CNFs, FT-IR analysis was performed (Fig. 2). All cationized CNF samples showed distinct absorbance bands at 2,850–2,950 cm-1, which are attributed to the C-H stretching vibrations of methyl and methylene groups in the quaternary ammonium structure. Additionally, the appearance of a new band at 1,480 cm-1, corresponding to the C-H bending vibration of the -N+(CH3)3 group, supports the successful incorporation of quaternary amine functionalities. The intensity of these characteristic peaks increased with higher GMA addition, confirming the progressive substitution of functional groups.
3.2 Effect of the degree of amine substitution on the morphology of CNFs
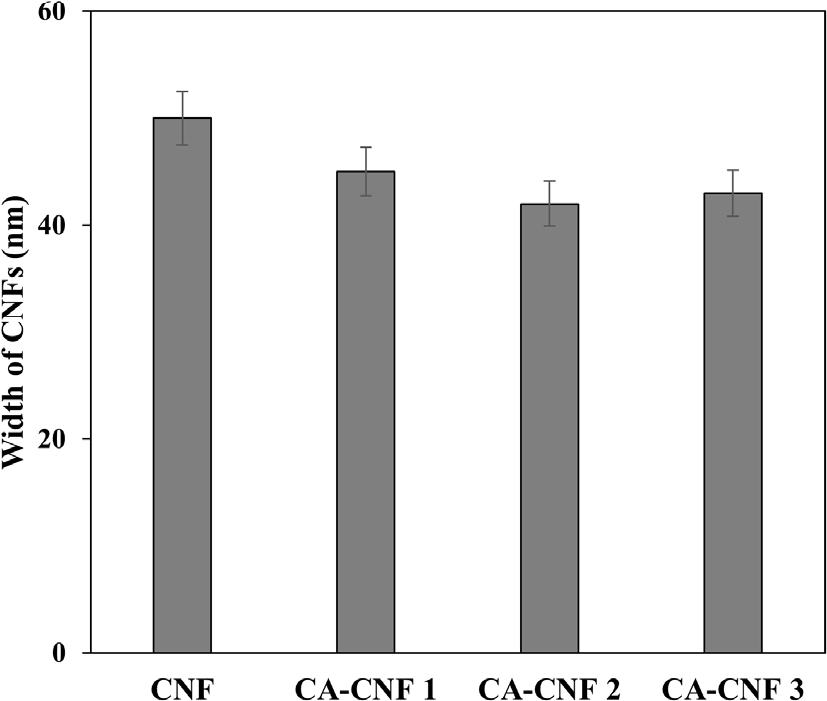
The fiber diameters of the samples are shown in Fig. 3. The untreated CNF exhibited an average fiber diameter of approximately 50 nm, while the diameters for CA-CNF 1, CA-CNF 2, and CA-CNF 3 were approximately 42 nm, 41 nm, and 39 nm, respectively, indicating successful nanofibrillation in all samples.
A comparison between untreated and treated CNFs revealed that fiber diameters decreased with higher degrees of amine substitution. This phenomenon is attributed to fiber swelling caused by NaOH during pretreatment and to electrostatic repulsion induced by GMA, facilitating the production of finer fibers.20) In addition, the amphiphilic nature of GMA likely enhanced fibrillation efficiency by reducing interfibrillar adhesion.
Similar trends have been observed in TEMPO-oxidized and carboxymethylated CNFs, where greater degrees of pretreatment and processing yield finer nanofibers.41,42)
While further investigation is required to directly correlate pretreatment degree with aerogel pore size, these morphological changes suggest the potential for tailoring pore structures through chemical modification.42)
3.3 Effect of the degree of amine substitution on the rheological properties of CNFs
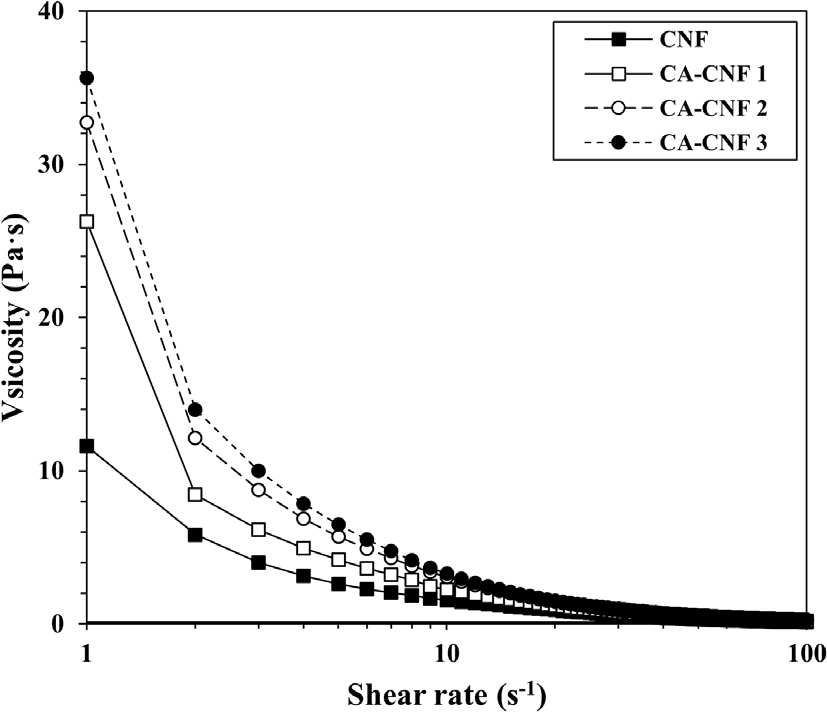
The viscosities of the CNF suspensions under low shear conditions are shown in Fig. 4. All samples exhibited shear-thinning behavior, and the values presented here represent the viscosity measured at low shear rates, where the effect of molecular interactions is more prominent. The viscosity increased with increasing amine substitution, indicating enhanced intermolecular interactions between CNFs and water molecules.
This trend is attributed to the higher affinity of quaternary ammonium groups (-CH2CHOHCH2N+(CH3)3) for water compared to native hydroxyl groups (-OH), resulting in stronger interactions and higher viscosity.
Similar behavior has been reported for TEMPO-oxidized and carboxymethylated CNFs, where viscosity increases with the degree of functionalization.17,43,44,45,46)
These findings suggest that tuning the GMA content can be used to control the viscosity of CNF suspensions, thereby influencing aerogel shape formation during fabrication.47)
3.4 Effect of the degree of amine substitution on the anion removal properties of CNF aerogels
3.4.1 Qualitative analysis using EDS
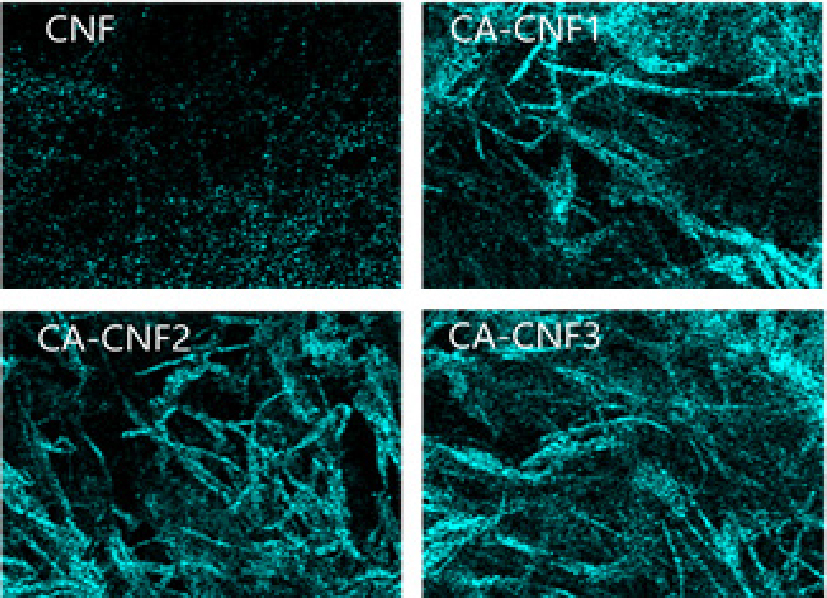
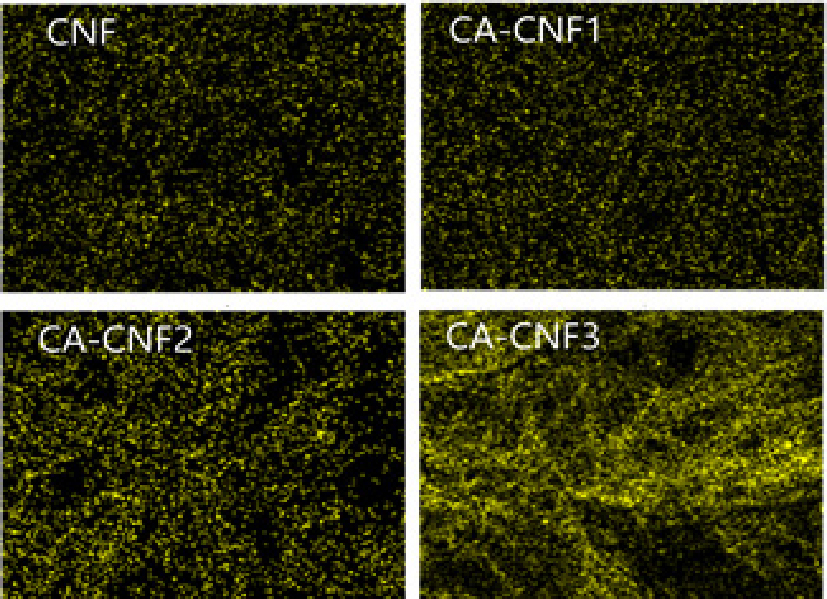
To evaluate anion capture performance, cylindrical aerogels were fabricated from CNFs with varying degrees of quaternary ammonium substitution. The distribution of nitrogen (N) in the aerogels, as analyzed by EDS, is shown in Fig. 5. Aerogels produced from untreated CNF exhibited negligible N content, whereas aerogels from cationized CNFs demonstrated increasing N distribution with higher degrees of modification.
Subsequent filtration of 500 ppm Cl- and Br- solutions through the CNF aerogels followed by EDS analysis (Figs. 6 and 7) revealed that the distributions of Cl- and Br- were markedly higher in cationized CNF aerogels compared to untreated CNF aerogels. Moreover, greater degrees of amination resulted in greater anion capture.
These results are consistent with previous reports that cationic CNFs exhibit enhanced adsorption capacity for anionic dyes,48) and that cationized cellulose and sawdust effectively adsorb anionic contaminants.38,39)
Similarly, for cationic heavy metals, anionic TEMPO-oxidized CNFs and carboxymethylated CNFs demonstrate increased adsorption capacity when bearing higher negative zeta potentials.49,50,51,52)
3.4.2 Quantitative analysis using charge density measurements
The charge densities of the Cl- and Br- solutions after filtration through the CNF aerogels were measured using titration with 0.001 N poly-diallyldimethylammonium chloride (Poly-DADMAC).
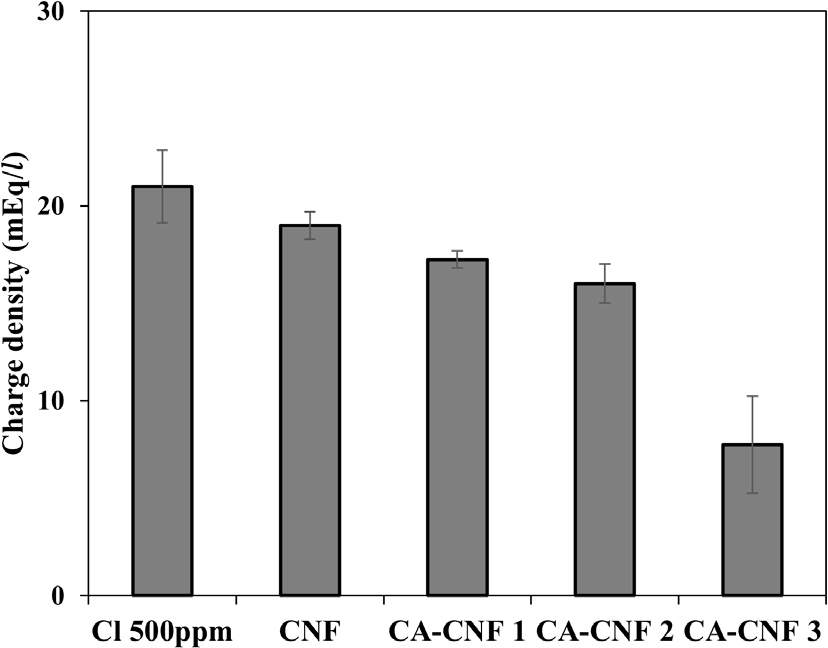
Fig. 8 shows the charge density of Cl- solutions before and after filtration. The unfiltered 500 ppm Cl- solution exhibited a charge density of approximately 21.0 mEq/L. After filtration through CNF aerogels with increasing degrees of quaternary ammonium substitution (untreated CNF, CA-CNF 1, CA-CNF 2, and CA-CNF 3), the charge density decreased to 18.2, 16.3, 14.9, and 9.1 mEq/L, respectively.
These values correspond to Cl- removal amounts of 2.8, 4.7, 6.1, and 11.9 mEq/L, resulting in removal efficiencies of approximately 13.3%, 22.4%, 29.0%, and 56.7%, respectively.
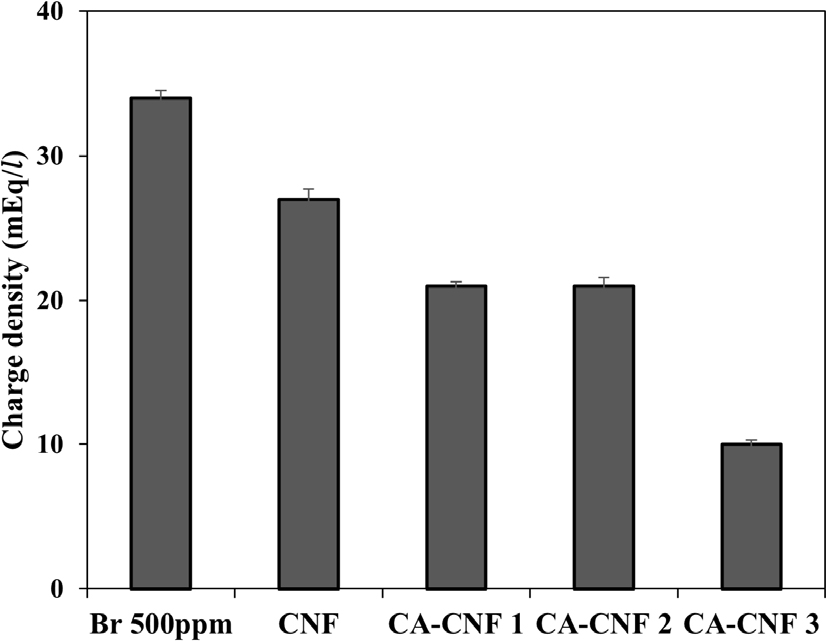
Similarly, Fig. 9 presents the charge density of Br- solutions. The initial 500 ppm Br- solution exhibited a charge density of about 34.0 mEq/L, which decreased to 28.6, 22.5, 20.1, and 10.4 mEq/L after passing through untreated CNF, CA-CNF 1, CA-CNF 2, and CA-CNF 3 aerogels, respectively.
This corresponds to Br- removal amounts of 5.4, 11.5, 13.9, and 23.6 mEq/L, with removal efficiencies of 15.9%, 33.8%, 40.9%, and 69.4%, respectively.
These results demonstrate that increasing the degree of cationic functionalization via quaternary ammonium groups significantly enhances the ability of CNF aerogels to capture anions. The observed reduction in charge density and the calculated removal efficiencies provide quantitative evidence of the improved anion-binding performance.
4. Conclusions
In this study, cylindrical aerogels for anion capture were fabricated using both untreated CNFs and quaternary amine-treated CNFs. The degree of quaternary amine substitution was found to significantly influence key material properties, including zeta potential, fiber morphology, viscosity, and anion adsorption performance.
The zeta potential measurements revealed that untreated CNFs exhibited a potential of -36.4 mV, whereas CA-CNF 1, CA-CNF 2, and CA-CNF 3 demonstrated increasingly less negative values of -17.2 mV, -13.4 mV, and -10.6 mV, respectively, corresponding to the degree of amine treatment.
The anion adsorption performance of the aerogels was evaluated through qualitative EDS analysis and quantitative charge density measurements following filtration of Cl- and Br- solutions. EDS analysis confirmed that the distribution of N in the aerogels increased with the degree of quaternary amine substitution. Correspondingly, greater Cl- and Br- capture was observed in aerogels treated with higher levels of GMA.
Charge density analysis provided more precise quantification, indicating that the charge density of the filtered solutions decreased as the degree of amination increased, corroborating the EDS findings.
Overall, this study demonstrates the potential of quaternary amine-functionalized CNFs as effective materials for anion removal in water treatment applications. The degree of quaternization directly enhanced adsorption efficiency, highlighting the critical role of surface functionalization in optimizing filter performance. Future research should focus on scaling up fabrication processes, evaluating regeneration and reuse potential, and assessing filtration performance under diverse water chemistries and real-world conditions.