1. Introduction
The Fischer-Tropsch synthesis (FTS) is a catalytic process for converting syngas (hydrogen (H2) and carbon monoxide (CO)) derived from coal, natural gas and biomass resources into valuable hydrocarbons with a wide range of molecular weights (C1-C50) for use as liquid fuels and fine chemical building-blocks.1-3) Iron, cobalt (Co), ruthenium and nickel catalysts have been used as active species in FTS processes, which involve the sequential addition of methylene intermediates based on polymerization-like elongation mechanisms. 4) FT products such as FT diesel fuels have become more attractive than petroleum-based diesels, because of their high cetane index and extremely low contents of sulfur and aromatic impurities. 5) However, the FTS reaction is highly exothermic; therefore requires careful process control to promote smooth gas flow, avoid the formation of local hot spots inside the catalyst layers and prevent sintering of the catalysts.6)
Recently, structured catalysts with micrometer-scale pores, including monoliths7), foams8), microchannels9) and honeycombs10), have attracted much interest for their potential applications in various catalytic processes. Microstructured catalysts possess several advantages, such as effective heat and gas diffusion, low pressure drop and high surface-to-volume ratios, all of which enable high catalytic performances.11) However, these support materials can have inherent drawbacks, e.g., heaviness, difficulty in processing and inadequate lateral gas diffusion, resulting in poor performance in practical reactor systems.12)
We have previously reported lightweight and flexible paper-like catalyst materials, namely paper- structured catalysts, prepared from ceramic fibers as a main framework by a high-speed and low-cost papermaking technique.13-15) Various types of metal nanoparticles (NPs), e.g., gold16), copper17), platinum18) and ruthenium19) NPs, were successfully synthesized in situ on a layered ceramic fiber network of papers via a facile impregnation method. The paper-structured catalysts demonstrated the high catalytic performances in a variety of chemical reactions. This was attributed to the micrometer-scale porous architecture of the paper-structured catalysts with layered fiber networks, which promoted smooth gas and heat diffusion, as in a microreactor.20) Furthermore, the tunability of the catalyst loading, layer thickness and layer-stacking pattern allowed us to tailor the catalyst layers for the optimal reaction conditions. 21)
In this study, Co-based paper-structured catalysts with micrometer-size pores were prepared via a papermaking technique and an impregnation method. The FTS reaction was performed with Co-containing paper composites, having different Co loadings, residence times and stacking patterns, to investigate the effects of the catalyst layer design on the FTS catalytic efficiency. In particular, we arranged the catalyst layers by stacking paper composites in a flow reactor to optimize catalytic FTS reaction. Our findings highlight the advantages of such “paper-based catalytic systems” for enhanced process engineering.
2. Materials and Methods
2.1 Materials
Alumina/silica ceramic fibers (B80, Al2O3 80 wt% and SiO2 20 wt%, Denka Co. Ltd., Tokyo, Japan) were cut to an average length of 0.5 mm. An alumina sol (AS-520, Nissan Chemical Industries Ltd., Tokyo, Japan) was used as an inorganic binder to improve the mechanical strength of the paper composites after thermal treatment. Two types of polyelectrolyte flocculants, i.e., cationic poly (diallyldimethylammonium chloride) (poly DADMAC; molecular weight ca. 3×105 g mol-1, charge density 5.5 meq g-1, Sigma-Aldrich Co., Ltd., St. Louis, MO, USA) and anionic polyacrylamide (anionic PAM; HH-351, molecular weight ca. 4×106 g mol-1, charge density 0.64 meq g-1, Kurita Water Industries, Ltd., Tokyo, Japan), were used in a dual polyelectrolyte system to form uniform fiber networks of ceramic fibers. Commercial bleached hardwood kraft pulp (Eucalyptus grandis natural hybrids >90%, Brazil) was beaten to meet the Canadian Standard Freeness of 300 mL with a Technical Association of Pulp and Paper Industry (TAPPI) standard beater (No. 2505 BE- 23, Kumagai Riki Kogyo Co., Ltd., Tokyo, Japan). 22) The metal precursor, i.e., Co(NO3)2·6H2O (99.5%), was purchased from Wako Pure Chemical Industries Ltd., Osaka, Japan. All the chemicals were reagent grade and used without further purification.
2.2 Preparation of paper-structured catalyst supports
Ceramic papers as catalyst supports were prepared by an established papermaking technique using a dual polyelectrolyte retention system according to our previous reports (Fig. 1).23,24) Ceramic fibers (5.0 g) were suspended in water (700 mL), and then polyDADMAC (0.5 wt% of total solid, 25 g), alumina sol (20 wt% consistency, 2.5 g), anionic PAM (0.1 wt% of total solid, 27.5 g) and pulp fibers (0.25 g by dry weight) were added in turn to the fiber suspension at 3 min intervals. The wet-state sheet was obtained by dewatering the suspension using a 200-mesh wire, and then the sheet was pressed at 350 kPa for 3 min and dried at 105°C for 1.5 h. The dried sheet was cut into disk-shaped pieces (diameter: 32 mm; thickness: 0.8 mm), and finally calcined at 500°C for 1 h to improve the mechanical strength by binder welding and to form porous structures by removing the pulp fibers used as a temporary matrix.
2.3 Preparation of cobalt-impregnated papers
Paper-structured catalyst composites, i.e., Co-impregnated papers, were prepared by an impregnation method.25) As-prepared ceramic paper composites were immersed in an aqueous solution of Co(NO3)2·6H2O at different concentrations (0.01-1.0 M, 80 mL) for 4 h at room temperature. The treated disks were then oven-dried at 105°C for 16 h, calcined at 400°C for 2 h and pretreated in an H2 flow (50 mL min-1) at 400°C for 6 h to reduce the Co oxides to catalytically-active Co(0).
2.4 Analytical characterization
The cobalt content of the Co-loaded papers was determined by atomic absorption spectrophotometry (AA-6600F, Shimadzu Corp., Kyoto, Japan). The paper samples prepared at different precursor concentrations were soaked in 35% nitric acid, and the concentration of metal ions eluted from the paper samples was quantitatively determined. The crystalline structures of the catalyst samples were determined by X-ray diffraction (XRD; XD-D1, Shimadzu Corp., Kyoto, Japan) with Ni-filtered Cu Kα radiation (λ = 1.5418 Å) and a scanning angle (2θ) range of 30°-75° at 30 kV and 40 mA. The crystallite sizes of Co-oxide and metallic Co(0) were estimated from the Scherrer equation [(D(hkl) = 0.89λ/βcosθ], where β is the full width at half maximum of the most intense diffraction peak. The microscopic images of the sample surfaces were obtained by scanning electron microscopy (SEM; Zeiss ULTRA55, JFE Techno-Research Co., Ltd., Tokyo, Japan), and elemental composition analysis was performed with energy-dispersive X-ray spectroscopy (EDS; NSS300, Thermo Electron Co., Ltd., Aichi, Japan). A gold coating was applied on the surfaces of all the paper samples to provide an ultra-thin conductive layer for the SEM measurements. Sample porosity and pore size distributions were determined with a mercury intrusion apparatus (Autopore IV 9500, Micromeritics Instrument Co., Chiba, Japan).
2.5 FTS reaction tests with Co papers and catalyst-free ceramic papers
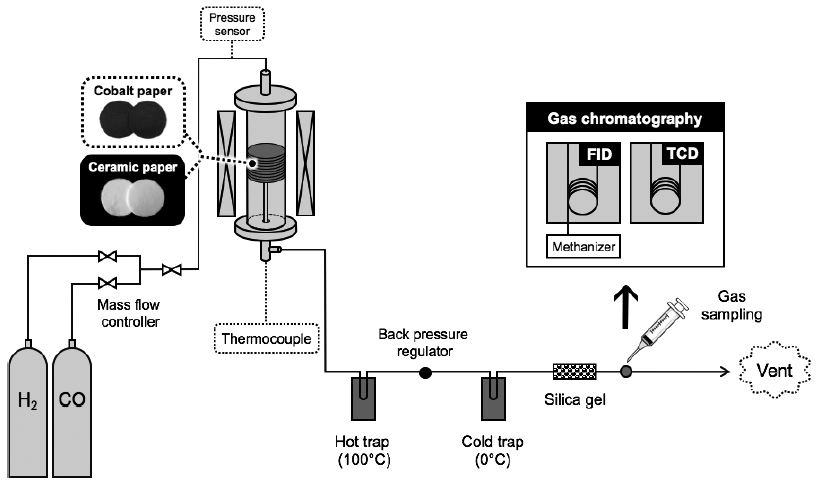
FTS reaction tests were performed in a tubular fixed-bed flow reactor system at 220-260°C and 0.12 MPa, as illustrated in Fig. 2. Various pieces of Co papers and catalyst-free ceramic papers were stacked vertically inside a stainless-steel cylindrical reactor (catalyst occupied volume: 6.4× 102-2.0×104 mm3). Prior to the FTS reaction test, the Co papers were reduced with H2 gas at 400°C at a constant gas flow rate of 50 mL min-1. Then a mixture of H2 and CO gases (molar ratio: 2/1) was fed into the reactor at 220-260°C at a constant gas flow rate of 150 mL min-1. During the FTS process, the obtained liquid products and water were condensed by two consecutive traps at 100°C and 0°C, respectively. The outlet concentrations of residual gas components, i.e., CO, CO2 and other hydrocarbons, were periodically analyzed by two types of gas chromatograph systems: a thermal conductivity detector (TCD; GC-14B, Shimadzu Corp., Kyoto, Japan) equipped with a 5A molecular sieve column and a Porapak Q column at a furnace temperature of 60°C, and a flame ionization detector (FID; GC-17A, Shimadzu Corp., Kyoto, Japan) equipped with an Rt-Al2O3 BOND/ Na2SO4 column and an online methanizer (MTN-1, Shimadzu Corp., Kyoto, Japan). The catalytic performance was evaluated in terms of CO conversion. The CO conversion (%) was calculated with Eq. 1.26,27)
3. Results and Discussion
3.1 Fundamental characteristics of paper composites
Fig. 3a-e shows optical images of the catalyst-free ceramic papers and Co papers prepared at different Co precursor concentrations of 0.01, 0.05, 0.1 and 1.0 M. The paper-structured composite, consisting of a framework of ceramic fibers, was prepared by an established papermaking protocol. 23,24) Active Co species were successfully loaded on the paper matrix by a simple impregnation method (Fig. 1), and the Co paper composites showed a gradation in color from light to dark brown as the precursor concentration was increased (Fig. 3b-e). At Co(NO3)2·6H2O precursor concentrations of 0.01, 0.05, 0.1 and 1.0 M, the Co-loadings of each paper composite were measured to be ca. 0.5, 0.9, 1.2 and 2.4 wt%, respectively, as determined by atomic absorption spectrometry. Prior to the FTS process, the obtained Co papers were subjected to an H2 reduction treatment. The H2-treated Co papers appeared to be dark gray-brown, which suggested the formation of metallic Co NPs on the paper-structured matrix (Fig. 3f); we refer to this fabrication process as on-paper synthesis in this study. These paper composites were lightweight, with an apparent density of 0.42 g cm-3 for the Co paper prepared at 1.0 M, flexible, easy-to-handle, easy-to-cut and stackable (Fig. 3g-k).

Fig. 3.
Optical images: (a) ceramic paper, (b-f) Co paper prepared with the Co(NO3)2·6H2O precursor at concentrations of (b) 0.01 M, (c) 0.05 M, (d) 0.1 M and (e) 1.0 M, (f) Co paper prepared at 1.0 M after H2 reduction and (g-k) flexible and easy-to-cut (h, j) ceramic and (g, i, k) Co papers.
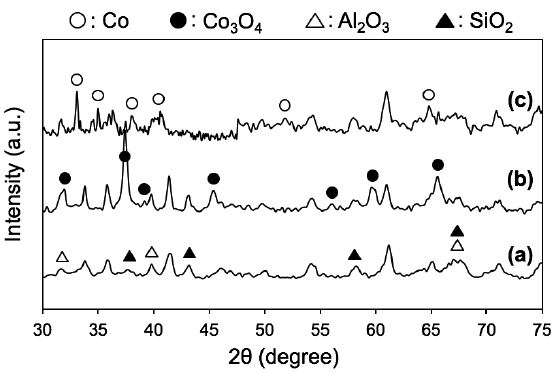
The crystalline structures of the paper composites were determined by XRD (Fig. 4). The characteristic peaks of Co3O4 were detected at 2θ = 31.8°, 37.4°, 39.4°, 45.4°, 56.2°, 60.0° and 65.6° [PDF-ICDD #43-1003]. The average size of Co3O4 crystallites calculated by the Scherrer equation was ca. 13 nm, indicating that Co3O4 particles formed on the ceramic paper matrix (Fig. 4b). In the XRD pattern of the H2-reduced Co paper, new peaks were detected at 33.1°, 35.0°, 38.0°, 40.8°, 51.7° and 64.9°, which confirmed the formation of catalytically-active metallic Co(0) with crystallite sizes of ca. 13 nm (Fig. 4c). Thus, our on-paper synthesis provided paper-structured catalysts containing active Co NPs, which may be effective catalysts in FTS processes.28)
3.2 Porous structures of ceramic and Co papers
SEM images of the catalyst-free ceramic paper and Co-treated paper are shown in Fig. 5. The ceramic paper consisting of inorganic ceramic fibers possessed a porous fibrous network structure with multiple layers of entangled fibers on top of one another (Fig. 5a). In the paper fabrication process, the alumina sol binder assisted in bonding the fibers to reinforce the catalyst papers by binder thermowelding29), and the organic pulp fibers acted as a temporary template to give the wet sheet strength during the papermaking process and were completely removed by calcination at 500°C to form the unique porous structure of these papers. In this study, we used hardwood pulp fibers with dry weight of 0.25 g according to our established papermaking method.29,30) Since the unique porous microstructure of the paper-structured catalyst plays an important role in promoting smooth diffusion of reactant gas and heat, the appropriate pulp dosage results in the excellent catalytic performance over various heterogeneous reactions, possibly including FTS process. The SEM image in Fig. 5c shows that small agglomerates were deposited on the fibers, possibly corresponding to Co3O4 species. The deposits were analyzed by EDS, as shown in Fig. 6b, and the elemental composition spectrum exhibited characteristic X-ray peaks from Co as well as from constituent elemental peaks of the inorganic ceramic fiber (Si, Al and O) and the surface conductive layer (Au). These results indicate the successful formation of Co catalysts on the ceramic fibers.
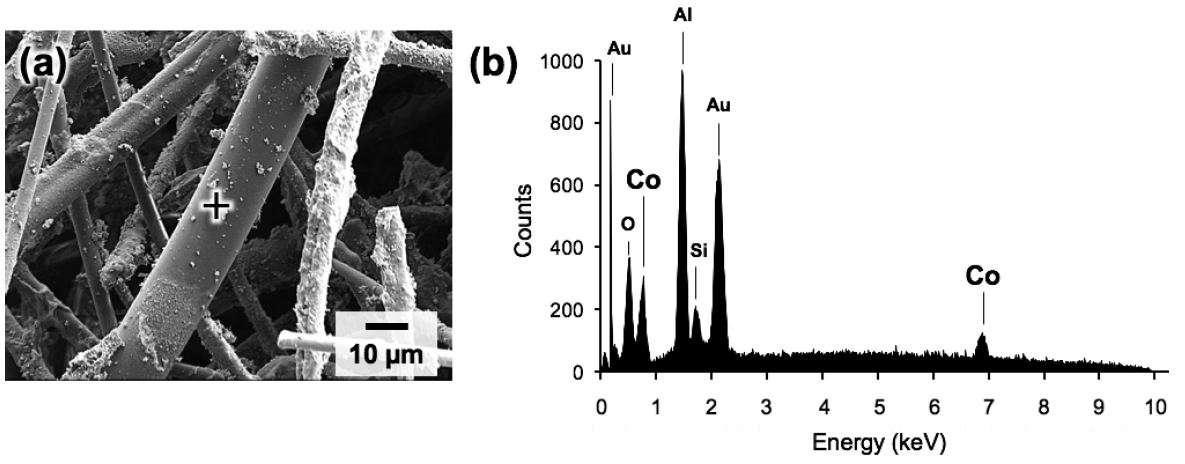
Fig. 6.
(a) SEM image of Co paper and (b) EDS profile acquired from the position of the black cross in the image.
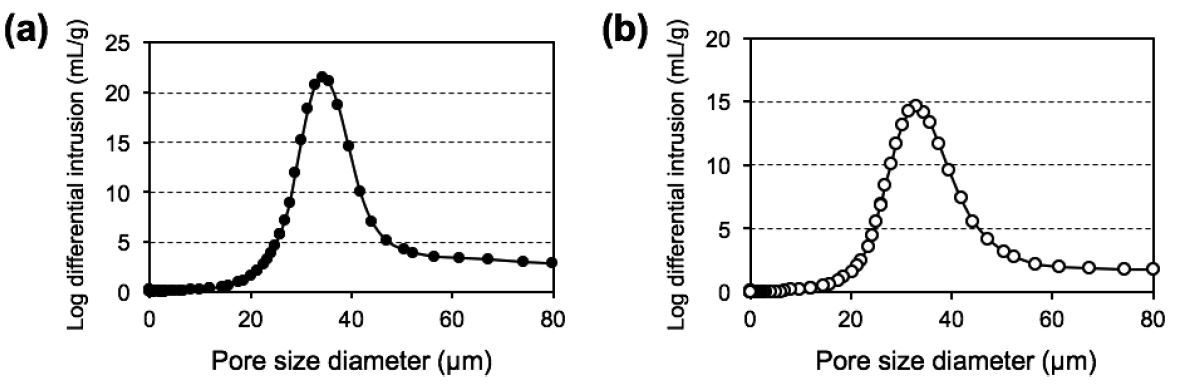
Fig. 7 shows the pore size distribution profiles of the paper composites as measured by a mercury intrusion method. In the case of the catalyst-free ceramic paper matrix, a sharp peak was detected at ca. 35.2 μm, and the porosity was determined to be 94.5%. After the on-paper synthesis, the pore size diameter of the Co paper was measured to be ca. 34.1 μm with a porosity of 92.4%. These results indicated that the micrometer-ordered pores of the paper composites remained unfilled even after the on-paper synthesis of Co NPs. In our previous researches, the pore sizes of paper-structured catalysts were successfully tuned by varying the amount of the pulp template13,30) or by using finer fibers such as SiC fibers with an average diameter of 0.5 μm29). The results indicated that the average pore size increased with the increase of the pulp dosage, while decreased with the use of the finer fibers. In this research, we have used 0.25 g of pulp fibers for preparing the paper-structured catalysts by referring to our previous reports for heterogeneous catalytic reactions. 14,15,19-21) Moreover, our previous studies have shown that the unique structural properties of porous paper-structured catalysts improved gas and heat diffusion, creating an environment similar to that of a microreactor31); hence, we expected these improvements could be useful in exothermic FTS processes.
3.3 FTS reaction with paper-structured Co composites
To investigate the FTS behavior, the FTS reactions were carried out using Co-impregnated and Co-free ceramic paper composites under four different testing conditions; (1) different reaction temperatures, (2) different Co loadings, (3) different gas residence times and (4) stacking patterns. A series of the FTS reaction tests were conducted with a fixed-bed tubular reactor system and the outlet gas composition was measured with two gas chromatography systems (Fig. 2). Prior to the reaction tests, each of the Co-impregnated paper samples was treated in an H2 flow at 400°C to activate the Co-oxide and form Co(0) NPs in the paper composites.
3.3.1 Effects of reaction temperatures on catalytic FTS efficiency
The FTS reaction tests were conducted over a wide range of operating temperatures of 220-260°C to investigate the effects of the reaction temperatures on the FTS. Five pieces of H2-reduced Co papers, prepared at the same precursor concentration of 1.0 M, were placed in the flow reactor with the occupied volume of 3.2×103 mm3 and a Co loading of 32.9 mg, as illustrated in the inset of Fig. 8. A mixture of H2 and CO gases (molar ratio: 2/1) was then fed into the reactor at 0.12 MPa at a constant gas flow rate of 150 mL min-1.
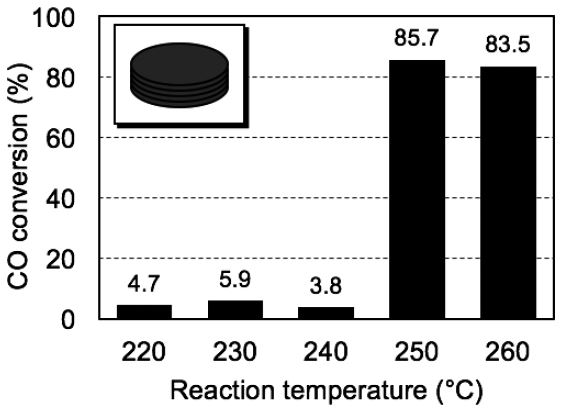
Fig. 8.
CO conversion after 25-h FTS reaction at different temperatures in the range 220-260°C over 5 pieces of the same Co paper composite; the inset figure is a schematic illustration of the stacked paper composites. Co content: 32.9 mg; volume occupied by catalyst: 3.2 ×103 mm3; gas hourly space velocity: 2,800 h-1; feed composition: H2/CO gas (molar ratio: 2/1); flow pressure: 0.12 MPa.
Fig. 8 compares the CO conversion ratios after 25 h on stream. The CO conversion was below 6% in the operating temperature range of 220-240°C, whereas increased to over 83% in the range of 250-260°C. This increase in conversion may be attributed to the activation energy of the Co-mediated catalytic FTS reaction. The reaction tests performed at lower operating temperatures had insufficient thermal energy to overcome the energy barrier between reactants and products in the catalytic FTS reaction. On the contrary, the FTS reaction proceeded efficiently on the surfaces of the Co(0) NPs at higher temperatures of 250 and 260°C. However, a slight decrease in CO conversion was observed at 260°C, possibly due to the onset of catalyst deactivation. On the basis of these results, we estimated the optimum working temperature of the Co catalysts in this study to be 250°C; thus, all the following tests were conducted at 250°C.
3.3.2 Effects of cobalt loadings on catalytic FTS efficiency
In this section, the FTS reaction tests were performed over 1, 5 and 20 pieces of 1.0 M-impregnated Co paper samples, corresponding to total Co loadings of 6.6, 32.9 and 132 mg, respectively (Fig. 9a). Fig. 9b shows the CO conversion determined from the outlet gas composition. A single piece of the Co paper composite exhibited the lowest CO conversion of 3.4%. By contrast, the CO conversion increased when 5 and 20 pieces of Co paper were used, reaching 85.7% and 99.6%, respectively. Since the catalytic efficiency of the FTS process strongly depends on the catalyst loading32), higher Co content of the paper composites improved the overall catalytic efficiency. Moreover, the increase in CO conversion was most notable between the reactions containing 1 and 5 pieces of Co paper, though much smaller increase in CO conversion was observed when the number of sheets was increased from 5 to 20 pieces of Co paper. These results suggested an optimum catalyst loading existed for promoting the FTS efficiency.
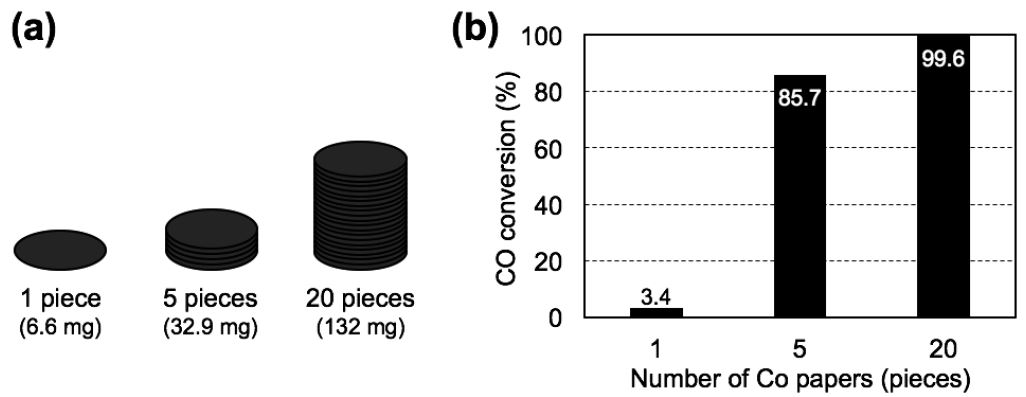
Fig. 9.
(a) Schematic illustration of the stacked paper composites from 1 to 20 pieces, (b) CO conversion after 25-h FTS reaction with different Co loading of 6.6, 32.9 and 132 mg, over 1, 5 and 20 pieces of Co paper composites, respectively. Volume occupied by catalyst: 6.4×102, 3.2×103 and 1.3×104 mm3, respectively; gas hourly space velocities: 14,000, 2,800 and 700 h-1, respectively; feed composition: H2/CO gas (molar ratio: 2/1); flow pressure: 0.12 MPa; reactor temperature: 250°C.
3.3.3 Effect of gas residence times on catalytic FTS efficiency
The effect of gas residence times on the FTS efficiency was investigated with 5, 12, 20 and 31 pieces of Co paper prepared at different Co(NO3)2· 6H2O precursor concentrations of 1.0, 0.1, 0.05 and 0.01 M, respectively. Each catalyst layer possessed a different residence time of 1.3, 3.1, 5.2 and 8.1 s, respectively, while the total Co content was maintained at ca. 33-34 mg (Fig. 10a). We assumed that the longer residence times of the reactant gas within the catalyst layer may lead to the higher CO conversion by promoting the formation of methylene intermediates and sequential addition of hydrocarbons, resulting in higher catalytic efficiency.
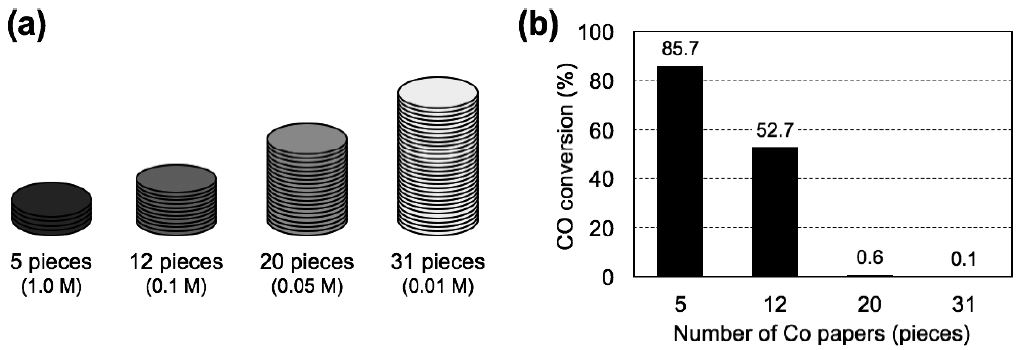
Fig. 10.
(a) Schematic illustration of the stacked paper composites; 5, 12, 20 and 31 pieces of Co paper composites prepared at 1.0, 0.1, 0.05 and 0.01 M of Co(NO3)2·6H2O precursor concentration, respectively. (b) CO conversion of 5, 12, 20 and 31 pieces of Co papers after 25-h FTS reaction with different residence times of 1.3, 3.1, 5.2 and 8.1 s, respectively, with the same total Co loading of ca. 33 mg. Volume occupied by catalyst: 3.2× 103, 7.7×103, 1.3×104 and 2.0×104 mm3, respectively; gas hourly space velocities: 2,800, 1,400, 700 and 450 h-1, respectively; feed composition: H2/CO gas (molar ratio: 2/1); flow pressure: 0.12 MPa; reactor temperature: 250°C.
Fig. 10b shows the CO conversions over stacked papers after 25 h on stream using a mixture of H2 and CO gases (molar ratio: 2/1) at 250°C and 0.12 MPa. The CO conversion with 5 pieces of Co paper (Co loading of ca. 33 mg) reached 85.7% at a gas residence time of 1.3 s; however, the CO conversion significantly decreased with further increases in gas residence time. In the case of 12 pieces, CO conversion decreased to 52.7%, and more pieces of Co papers had no effect on the catalytic FTS reaction, despite the catalyst layers having the same Co content of ca. 33 mg and longer gas residence times.
We observed a substantial decrease in CO conversion as gas residence times increased; such unexpected result may be related to the distribution and density of active sites within the catalyst layers. When the H2/CO gas flows through the stacked paper composites (Fig. 10a), initial contact between the H2/CO gas and catalytically-active sites occurs at the top layer of the catalyst papers. The high density of active Co catalysts at the top layer of the stacked catalyst papers likely contributed to the formation of the methylene intermediates and subsequent hydrocarbon chain elongation by sequential addition of intermediates. In the case of 31 pieces, the catalytically-active sites were three-dimensionally distributed in the reactor, but the lower Co content in the early stage of the reactor was insufficient to generate methylene intermediates. By contrast, 5 pieces of Co paper provided a short pathway for the reactant gas through the catalyst layers with a high density of accessible catalytic active sites. This higher density of active sites resulted in preferable formation of intermediates, leading to higher CO conversion than that occurring in the catalyst layers containing 12, 20 and 31 pieces of Co paper with the same total Co content of ca. 33 mg. Therefore, both the density and distribution of active catalyst sites are important considerations for promoting a smooth FTS reaction. Hence, we next examined tuning these factors by adjusting the stacking of the paper- structured Co catalysts.
3.3.4 Effects of stacking patterns on catalytic FTS efficiency
The above-mentioned catalytic FTS reaction tests revealed that the CO conversion strongly depended on the catalyst distribution within the catalyst layer, i.e., the catalyst density in the porous fiber networks. Thus, we investigated an optimized catalyst layer design to improve the CO conversion efficiency by varying the gas residence times, while maintaining the catalyst density in each constitutive paper. We examined alternate stacking patterns of Co-loaded paper-structured catalysts and catalyst-free ceramic papers, with total layer numbers of 5, 12, 20 and 31 pieces, as shown in Fig. 11a. All the stacking catalyst layers contained 5 pieces of Co paper with a total Co content of 32.9 mg. The remaining sheets were made up of catalytically-inert ceramic paper, which acted as spacing layers. Thus, the catalyst layer with 5 pieces possessed only the 5 Co papers in the reactor and no inert ceramic papers. The catalyst layers with 12, 20 and 31 pieces included with 5 pieces of the Co-loaded papers each with corresponding 7, 15 and 26 pieces of catalyst-free ceramic paper. A single piece of Co paper and a fixed number of spacing layers were stacked together in a repeating manner, i.e., a quadruple-layered architecture. The catalyst layers containing a total of 5, 12, 20 and 31 papers possessed gas residence times of 1.3, 3.1, 5.2 and 8.1 s, respectively, each with the same total Co loading of 32.9 mg. The FTS reaction test was performed under the same reaction conditions used in previous tests, at 250°C and 0.12 MPa with H2/CO (molar ratio: 2/1) for 25 h on stream.
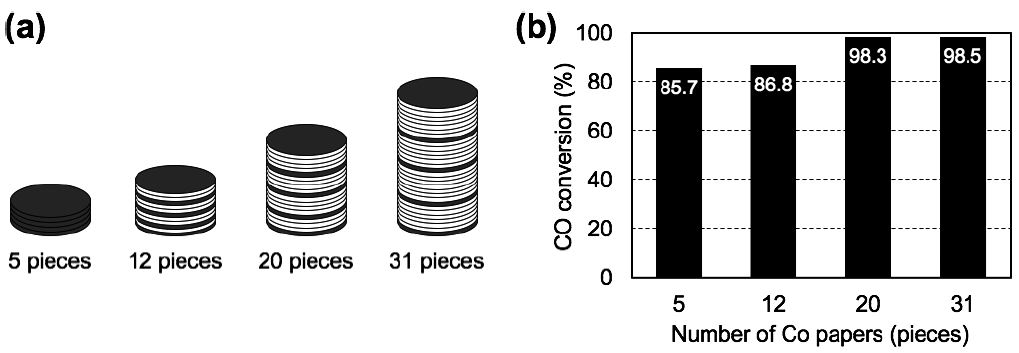
Fig. 11.
(a) Schematic illustration of the stacked paper composites with 5, 12, 20 and 31 pieces of paper composites with alternating Co papers (gray disk) and catalyst-free ceramic papers (white disk). The stack of 12 pieces consisting of 1 Co paper (P), 2 ceramic spacer layers (S), 1P, 2S, 1P, 2S, 1P, 1S and 1P, stacked in the order. The stack of 20 pieces was arranged: 1P, 4S, 1P, 4S, 1P, 4S, 1P, 3S and 1P. The stack of 31 pieces was arranged 1P, 7S, 1P, 6S, 1P, 7S, 1P, 6S and 1P. (b) CO conversion of the 5, 12, 20 and 31 pieces paper stacks after 25-h FTS reaction, with residence times of 1.3, 3.1, 5.2 and 8.1 s, respectively. The total Co loading of 32.9 mg was the same in each case. The stacks occupied volumes of 3.2×103, 7.7×103, 1.3×104 and 2.0×104 mm3, respectively; gas hourly space velocities were 2,800, 1,400, 700 and 450 h-1, respectively. Feed composition: H2/CO gas with (molar ratio: 2/1); flow pressure: 0.12 MPa; reactor temperature: 250°C.
Fig. 11b shows the relationship between the gas residence time and CO conversion over the alternately stacked paper-structured Co catalysts and catalyst-free ceramic papers. Five pieces of Co papers alone, without any spacing layers, exhibited 85.7% CO conversion. However, the Co papers with ceramic paper spacers showed even higher FTS efficiency, with CO conversion values of 86.8%, 98.3% and 98.5%, corresponding to 12, 20 and 31 layer stacks of paper, respectively, even though all the stacked paper pieces had the same total Co content of 32.9 mg. These results indicate that the gas residence time and stacking pattern were important factors affecting the FTS process efficiency. As described in the previous section (Fig. 10b), experiments based on 12, 20 or 31 pieces of Co-treated paper with lower Co contents per piece as the number of pieces increased, showed a decrease in CO conversion of 52.7%, 0.8% and 0.1%, respectively, as the increase in the number of pieces. Thus, the considerable increase in the FTS reactivity achieved by combining Co-loaded papers and catalyst-free ceramic papers was presumably attributed to the formation of methylene intermediates inside the high-density Co papers and effective gas/heat diffusion through the spacing layers, resulted in a more favorable isothermal environment for the FTS process in the reactor. In summary, the paper-structured catalysts containing active Co NPs were effective for the FTS reaction, and stacking the Co treated paper composites with untreated ceramic spacers allowed process control of the gas residence times for improving the catalytic efficiency by promoting heat diffusion inside the catalyst layers in the reactor.
3.3.5 Paper-based FTS reaction; catalytic efficiency and selectivity
In this section, the catalytic efficiency and selectivity of the FTS products are discussed briefly with a view to potential uses of our paper-structured catalysts in future applications. In the FTS reaction, products are categorized by their hydrocarbon chain length, i.e., light hydrocarbon products (C1- C4), middle-distillate products (C5-C20) and heavy oil products (C21+). Owing to the wide product distribution of C1-C50 hydrocarbons, the development of the FTS catalysts with high catalytic efficiency and high selectivity for a particular target product is a challenging task. The production of high-quality liquid fuels, such as gasoline, jet fuels and diesel fuels, strongly requires increased selectivity for C5+ hydrocarbons and decreased formation of CH4 and C2-C4 paraffins. In this respect, our paper- structured catalysts require further development. Although we achieved a high CO conversion >98%, which may be suitable for practical applications, our paper-structured catalysts showed poor selectivity (<1%) for the target C5+ products, and undesirable light hydrocarbons such as C1 products were obtained in large quantities. This was likely due to the low pressure conditions (0.12 MPa) used in this study, as compared with typical flow and pressure requirements of FTS processes (~2.0 MPa).32) Of several arrangements tested, such a single piece of Co paper at 250°C or 5 pieces of Co paper at 220-240°C, we found a higher selectivity for C2-C5+ products (>10%), but lower CO conversion (<1%). Many FTS processes are aimed at developing efficient FTS catalysts with high C5+ selectivity as well as high CO conversion; hence, our present work provides some fundamental insights into the development of highly-active catalysts and process engineering for the practical FTS processes by proposing a new strategy based on stacking arrangements of porous paper-structured catalysts. In future work, thermoconductive paper-structured catalysts and high-pressure flow testing will be investigated to achieve both high CO conversion and high selectivity for longer hydrocarbons.
4. Conclusions
We successfully prepared paper-structured catalysts for catalytic FTS reaction, using a papermaking technique to form micrometer-size pores and a facile impregnation method to synthesize Co NPs in situ. The FTS behavior was investigated with regard to Co loadings, gas residence times and stacking patterns of the paper composites. These factors influenced the formation of methylene intermediates during the exothermic FTS reaction and the gas/heat diffusion in the porous catalyst layers. Alternate stacking of 5 Co paper pieces (Co content: 33 mg) and 7-26 catalyst-free ceramic paper sheets demonstrated high CO conversion over 98% at 250°C. Our paper-structured catalysts with unique porous architectures, which allow on-demand tailoring of the catalyst layers by stacking of paper composites. Our new approach may be useful for improving the energy efficiency and process economics of high-performance catalytic FTS processes.