1. Introduction
Aerogel is a highly porous material with up to 99% porosity, low density (as light as air), high surface area (200-1,000 m2/g), and reaching up to nanoscale pore size. Due to these unique properties, a wide range of aerogel applications has been reported, such as contamination of nuclear waste, water repellent coatings, chemical sensors, heterogeneous catalysts, acoustic transducers, and energy storage devices. Aerogel is originally produced from inorganic components of silica, alumina and tin oxide, followed by organic polymers, in which resorcinol/melamine and formaldehyde are as representatives.1) More recently, cellulose aerogel has gained great interest for being derived from cellulose raw material. Therefore, the application fields of cellulose aerogel have been further extended where biocompatibility and biodegradability are particularly requested, i.e., biodegradable carriers for drug delivery systems and advanced food materials.2,3) Cellulose aerogel can be prepared from regenerated cellulose, cellulose derivatives and native cellulose. Among them, nanofibrillated cellulose (NFC) as one of the native celluloses, preserves the structure of cellulose I, characterized as good crystallinity, large specific surface area and inherently entangled network structure, thus it is a great skeleton for cellulose aerogel. Typically, NFC is produced by mechanical high-shear disintegration and homogenization with enormous energy required. When applied with pretreatment, e.g. TEMPO-mediated oxidation (2,2,6,6-tetramethylpiperidine-1-oxyl) before homogenization, the energy consumption is heavily reduced over 100 times (from 700-1,400 MJ/kg to 7 MJ/kg), leading to more economical and efficient production of NFC of good quality.4) The principle of TEMPO process is based on the selective oxidation of the C6 primary hydroxyl groups of cellulose, with the NaClO and NaOH consumed. As a result, the repulsive forces among the ionized carboxylate separate the nanofibrils within the fibers from each other.5) The oxidation efficiency of TEMPO process (usually characterized by carboxylate content) and the concentration of cellulose suspension have strong impact on the properties of NFC, consequently affecting the structure of dried cellulose aerogel.6,7)
Assorted applications of cellulose aerogel mainly depend on its porous structure. The existing hydrogen bonding between the cellulose molecules is susceptible to the drying process, causing the collapse of the original pore structure and accordingly weakening the porous nature of the aerogel.8) Therefore, the drying process is crucial for retaining the native structure. In the present study, the aerogel is prepared from NFC by adjusting concentration of NFC and applying two different freezing methods. Via investigation, the porosity, surface area and related characterizations of prepared cellulose aerogel will be discussed to elucidate the influence mechanism of variables on the porous structure of cellulose aerogel.
2. Materials and Methods
2.1 Materials
A commercial softwood bleached kraft pulp (BSKP) was used in this study. 2,2,6,6-tetramethylpiperidine- 1-oxyl (TEMPO), sodium bromide, sodium hypochlorite solution were of laboratory grade, purchased from Aladdin Chemicals Co. Ltd. (Shanghai, China).
2.2 Preparation of NFC
Cellulose surface oxidation was carried out according to Saito et al.7) with TEMPO as catalyst and sodium chlorite as a primary oxidant. In brief, BSKP (20 g of oven dried weight) was suspended in water (2 L) containing TEMPO (0.25 g) and sodium bromide (2.5 g). The TEMPO-mediated oxidation of the cellulose slurry was started by adding NaClO solution (5 mmol/g pulp) and conducted at room temperature under gentle agitation. The pH was maintained at 10.5 by adding 0.5 M NaOH. The reaction was finished until no more decrease in pH was observed, and adjusted the pH to 7 by adding 0.5 M HCl. The TEMPO-oxidized product was thoroughly washed with water by filtration and stored at 4°C before further treatment or analysis. The yield was ~90%, and the carboxylate content in the oxidized solid product was ~0.4 mmol/g pulp. A domestic blender was first applied to mix the suspension for 5 min. Different concentrations of TEMPO-oxidized pulp slurry (0.5 wt%, 1.0 wt% and 2.0 wt%) were passed through the homogenizer at 60 MPa pressure for minutes until a clear gel was obtained. The concentrations of obtained gels were corrected to 0.6 wt%, 1.0 wt% and 2.0 wt% by solid content determination.
2.3 Characterization of prepared NFC
Microscopic observation of NFC was performed by transmission electron microscopy (TEM). Diluted cellulose dispersion was mounted on a glow-discharged carbon-coated electron microscopy grid. The excess liquid was absorbed by a filter paper, then the sample was drying at room temperature for 2-3 h. The sample grid was observed at 79 kV using a JEOL electron microscope (JEM- 1010).
A Thermo HAAKE rotational rheometer (Rheo- Stress 600) was used to examine the flow properties of NFC. The sample was transferred to the titan cone-plate sensor system with a 4° cone angle and a 40 mm diameter. Measurements were carried out at 25°C for 3 min at sheer rate range of 0.01-1000 s-1. Flow curves were recorded by HAAKE rheometer software (RheoWin version 3.14).
2.4 Preparation of NFC aerogel
The NFC dispersion was frozen in an aluminum mould by submersion either in liquid nitrogen (temperature -196°C) for several seconds or in a freezer (temperature -18°C) for overnight, then transferred into a freeze-dryer (Boyikang FD-1D- 50 model, Beijing, China). The sample was kept frozen during drying at a pressure of approximately -20 Pa. The drying was typically finished within 24 h.
2.5 Characterization of prepared aerogel
The specific surface area was determined from the adsorption results using the Brunauer- Emmet-Teller (BET) method, and determined by nitrogen gas adsorption using a NOVA 2000e system (Quantachrome, US). Before analysis, 0.02- 0.03 g sample was degassed at a temperature of 115°C for 4 hours. BET analysis was carried out for a relative vapor pressure of 0.01-0.3 at -196°C. From the experimental BET specific surface area values (BET), the corresponding diameter of the fibril d in the aerogel was estimated according to Eq. 1 assuming a cylindrical shape of the fibrils and assuming that the density of cellulose ρc is equal to 1,460 kg/m3. The average pore size of the NFC aerogels was estimated from the nitrogen desorption isotherm according to the analysis of Barrett-Joyner-Halendar (BJH).9)
The bulk density of the cellulose aerogels (ρ*) was determined from the dimensions and weight of the specimen. Their porosity was calculated from Eq. 2 where the ratio ρ*/ρc is the relative density.
Hg-porosimetry was also performed to measure the pores size and their distribution of cellulose aerogel, the characterization was done by AutoPore IV 9500 under low pressure (max. pressure setting: 30,000 psi).
To study the micro-structure of the NFC aerogels, the specimens were analyzed with a HITACHI S-3400N SEM (scanning electron microscopy) to obtain electron images, operated at an acceleration voltage range of 15 kV. The specimens were fixed on a metal stub with black double-sided tape and coated with gold/palladium layer using a KYKY SBC-12 Sputter Coater before analysis.
3. Results and Discussion
3.1 General description of TEMPO-oxidized cellulose
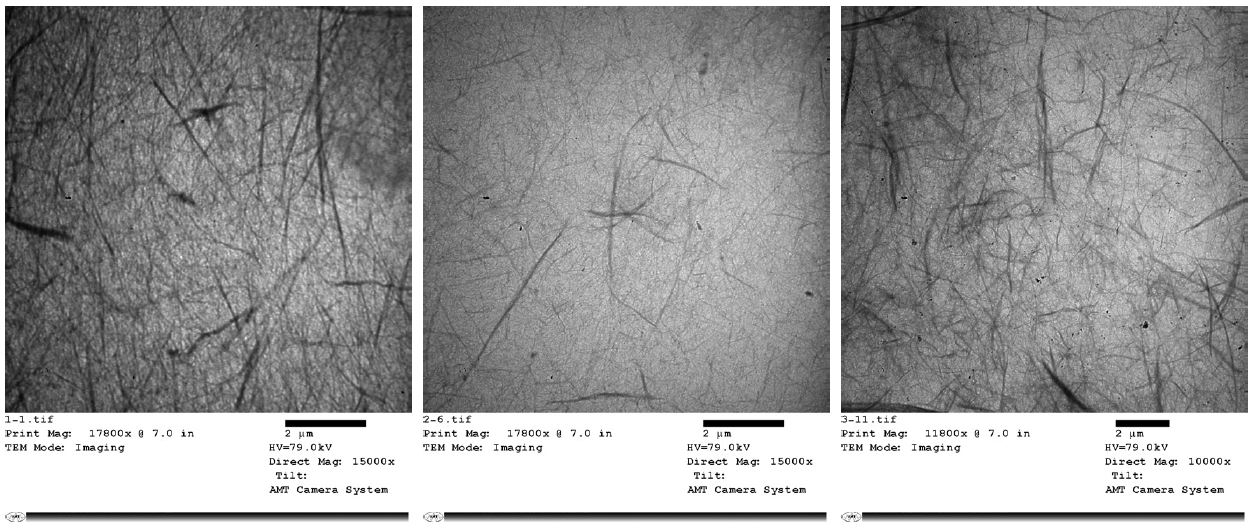
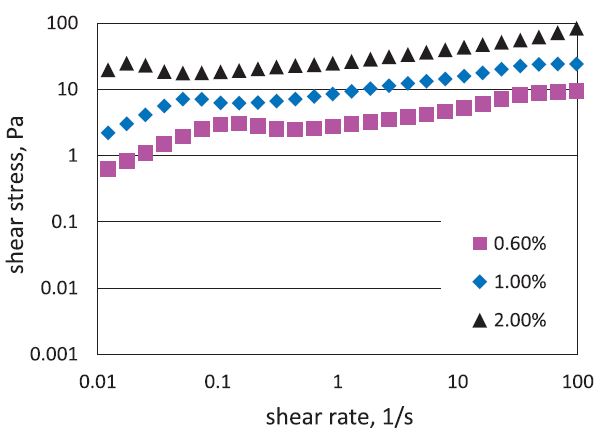
Fig. 1 shows TEM images of TEMPO-oxidized cellulose fibrils. The isolated cellulose fibrils prepared under different concentrations exhibited two classes of cellulose fibrils, one with nanofibers 10- 20 nm in width, the other with 100-200 nm in width, and several micrometers in length, implying the relatively high aspect ratio of prepared NFC and inherently entangled network structure. As for the flow properties of prepared NFC (shown in Fig. 2), dispersions exhibited pseudo-plastic flows for all three samples. With the increasing concentration, the yield stress at lower shear rates was more in the flat region, which is often observed in gel-like structure.10) The results are well corresponded to the appearance of prepared NFC, more transparent and viscous NFC was observed at higher concentration. It should be noticed that under proper oxidation (i.e., ~0.4 mmol/g pulp of carboxylate content in this study), the gel-like behavior could be retained even at low concentration of TEMPO-oxidized cellulose fibrils. The obtained aqueous nanofibrils gels suggested the structural integrity of NFC, which ensures the skeleton for the porous structure of cellulose aerogel.
3.2 The structure properties of cellulose aerogel
Fig. 3 illustrates the variation in average density of the aerogels was from 4.92 mg/cm3 to 17.48 mg/cm3, as a function of NFC mass under both freezing methods (Fig. 3 Right Y-axis). This trend is in consistent with the results reported in Health and Thielemans.11) Meanwhile, the porosity decreased in line with the increasing density from 99.66% to 98.91%. The possible reason is that the aggregation of NFC is prone to occurring at higher concentration and forming lamellar structure due to the effect of hydrogen bonding, consequently, a lower porosity was attained.
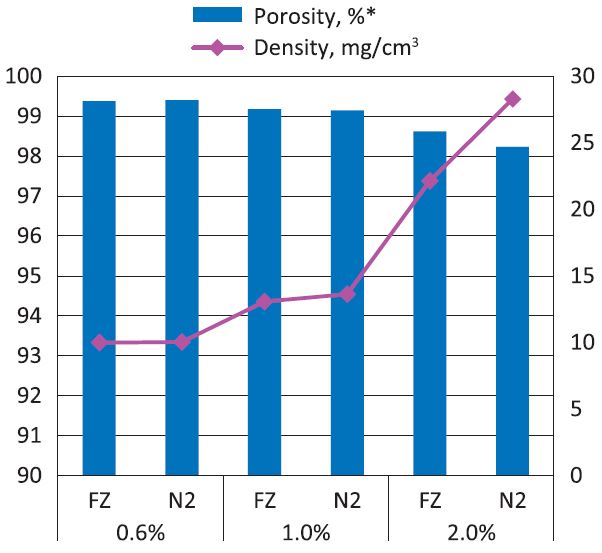
Fig. 3.
The porosity and density results of prepared cellulose aerogels, where FZ stands for regular drying in the freezer, while N2 stands for rapid drying in the liquid nitrogen; *Porosity was calculated based on Eq. 1.
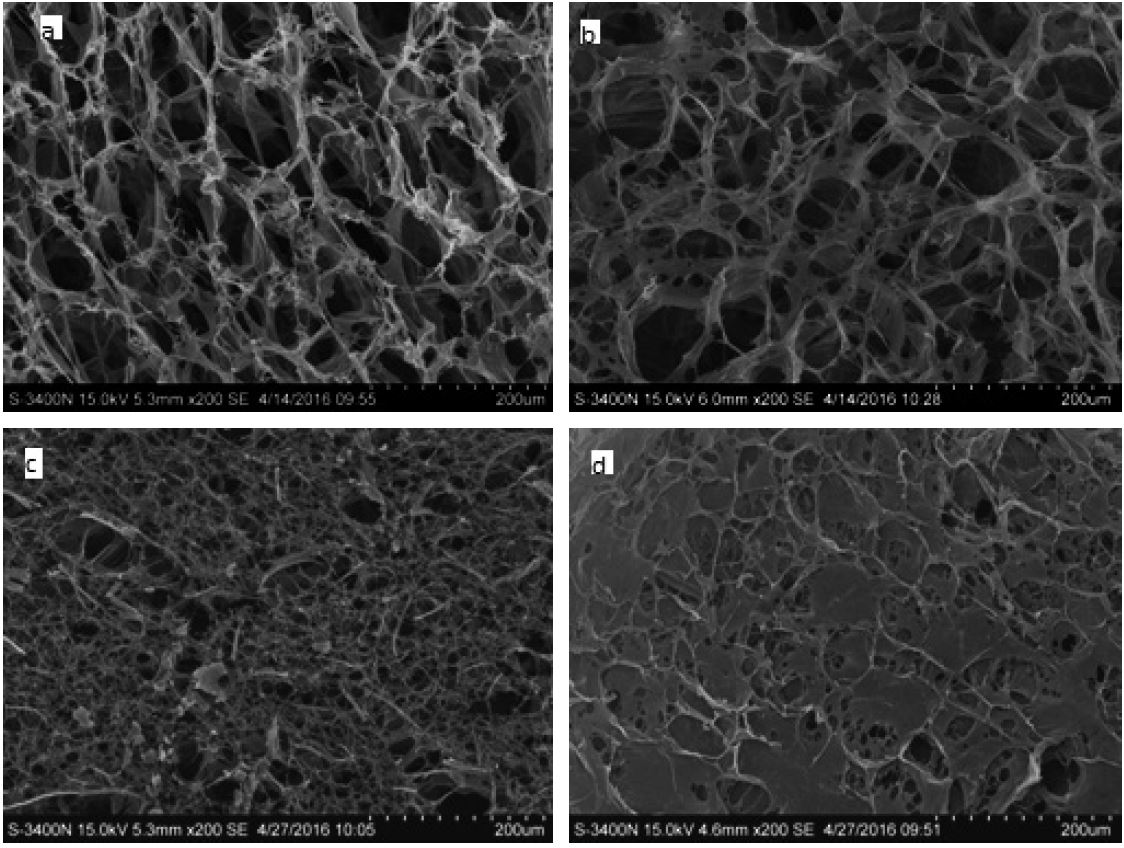
The comparison of the effects of two freezing methods on the porosity of cellulose aerogel was also shown in Fig. 3. In general, regular freezing (Method 1) resulted in lower density and higher porosity of cellulose aerogel than rapid freezing in the liquid nitrogen (Method 2). After complete water removal, the resulting sponge-like aerogel had some volumetric shrinkage for the sample prepared by Method 1, i.e. approximately 20%. For sample prepared by Method 2, much smaller shrinkage was observed during the drying, i.e., approximately 4%. The shrinkage which took place during Method 1 led to the major changes in the porous structure of cellulose aerogel, as shown by SEM (Fig. 4d): 2-dimensional extended sheet-like structures were presented due to the aggregation of nanofibers. The SEM and porosity results indicate that an internal porosity may exist in the sheet-like structure, which possibly explain that the moderately high porosity was observed for sample prepared by Method 1 even when 20% shrinkage took place. It is also in accordance with SEM micrographs observation in Pääkkö12), where the various smaller pores within the sheet-like structure can be found after regular freeze-drying.
As for the sample prepared by Method 2, Figs. 4a-c show an interconnected fibrillar skeleton structure, i.e. the 3-dimensional open fibrillar network structure of the aqueous gel was qualitatively preserved after the water extraction, suggesting the aggregation of nanofibers may be prevented by rapid freezing in liquid nitrogen. This finding is consistent with Jin’s publication in 200413), reporting that rapid freezing leads to more uniform structure than regular freezing which is hindered by the growth of ice crystals. As the concentration of NFC increased, the entangled network structure was preserved well as shown in more stable gel.
The internal specific surface area determined through nitrogen gas adsorption experiments of the obtained aerogels is shown in Fig. 5. The BET analysis describes the microporous area (2-200 nm test range for the equipment in this study) of sample that is accessible to gas adsorption. It may indicate that pore volume measurements by nitrogen adsorption are not precise enough for samples containing macropores (pores of width larger than 200 nm in this study). The value was increased with the concentration of NFC, implying that more microporous structure was formed due to the condensed structure of interconnected and layered sheets at a higher concentration of cellulose aerogel. Therefore, the formed condensed structure was characterized as smaller pore size at higher concentration of cellulose aerogel, which is shown in Fig. 5.
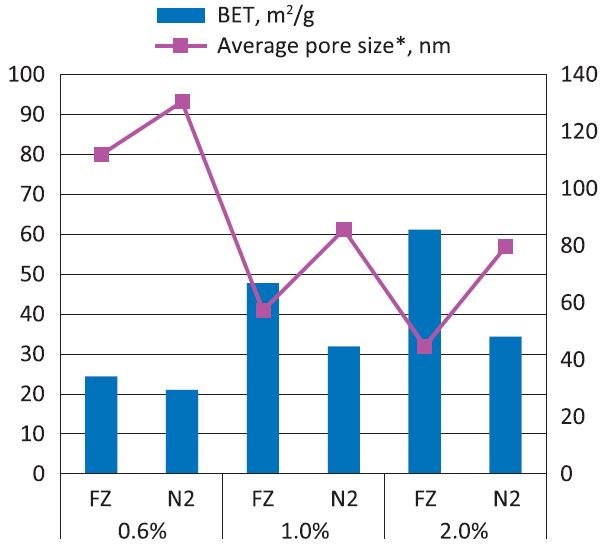
Fig. 5.
BET and average pore size results of prepared cellulose aerogels, where FZ stands for regular drying in the freezer, while N2 stands for rapid drying in the liquid nitrogen; *Average pore size was calculated based on Eq. 2.
Comparing the two freezing methods in Fig. 5, Method 1 resulted in larger BET values than Method 2 at various concentrations, which may indicate the more microporous structure was created and observed when applying Method 1. The results can be attributed to a) the limitation of BET analysis and b) regular freezing leads to the aggregation into sheet-like structure, probably containing internal porosity. Results of mercury intrusion analysis (Fig. 6) show that the sample displayed heterogeneous porous structure in the inner areas with wide pore size distribution from several micrometers to hundreds of micrometers. According to the analysis, the sample prepared by Method 2 was characterized with the average pore radius of 72.96 μm and the total cumulative volume of 42.74 cm3/g, which was slightly larger than that of the sample prepared by Method 1 (39.24 cm3/g) characterized by relatively large median pore radius (80.11 μm). The cumulative pore volume is the indication of numbers of large pores, which may imply more macroporous structures exhibited in the sample prepared by Method 2.14) On the other hand, the wider pore size distribution was observed in sample prepared from Method 1, indicating that rapid freezing leads to more uniform structure than regular freezing.
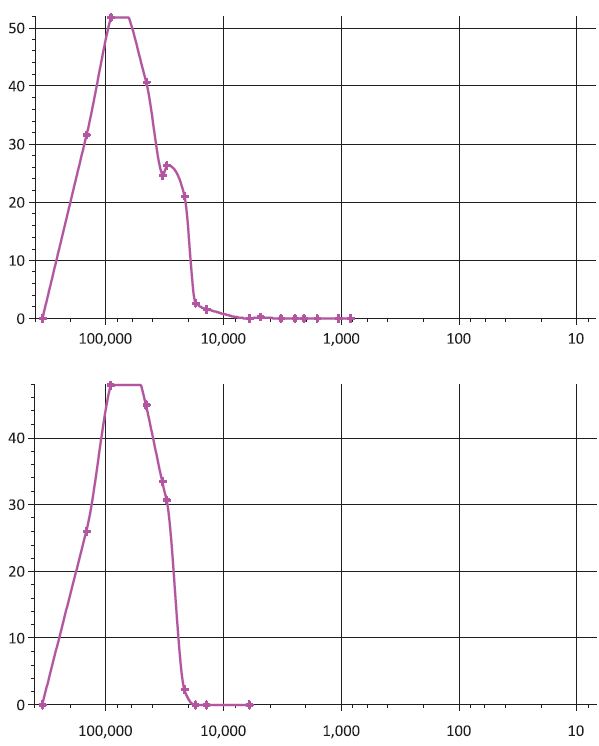
Fig. 6.
Pore size distribution of cellulose aerogel obtained from mercury intrusion measurements (top: 1.0% FZ-Method 1, bottom 1.0% N2-Method 2); x-axis: pore size diameter (nm); yaxis: log differential intrusion (mL/g).
During freeze drying process, the water is removed from the cellulose suspension so that a porous low-density NFC aerogel is created. Because of the hydrogen-bond effect, the aggregation occurs in the drying process indicated by the shrinkage of prepared samples. Under regular freezing, the growth of ice crystal took place with the generation of large pores; as a result the heterogeneous structure of aerogel was attained. Meanwhile, more aggregation may occur in accordance with growth of ice crystal in the regular freezing and drying. By contrast, rapid freezing may eliminate the generation of ice crystal by much quicker preserving the structure, leading to more uniform porous structure of dried cellulose aerogel.
4. Conclusion
This study reports preparation of cellulose aerogel by varying concentrations of TEMPO-oxidized cellulose, making gel-like behavior of NFC, followed by different freezing processes and drying. The higher concentration of NFC can result in more condensed porous structure with larger specific surface area but may also lead to some aggregation of cellulose fibrils with lower porosity. The cellulose aerogels prepared by both freeze-drying methods (regular and rapid freezing) exhibited heterogeneous porous structure. Compared to the regular freezing, rapid freezing in the liquid nitrogen may eliminate the aggregation resulted from the growth of ice crystal, leading to more uniform structure of dried cellulose aerogel. Characterization of porous structure of cellulose aerogel cannot be achieved by single approach, but with the combination of BET, mercury intrusion and SEM together for a comprehensive evaluation.