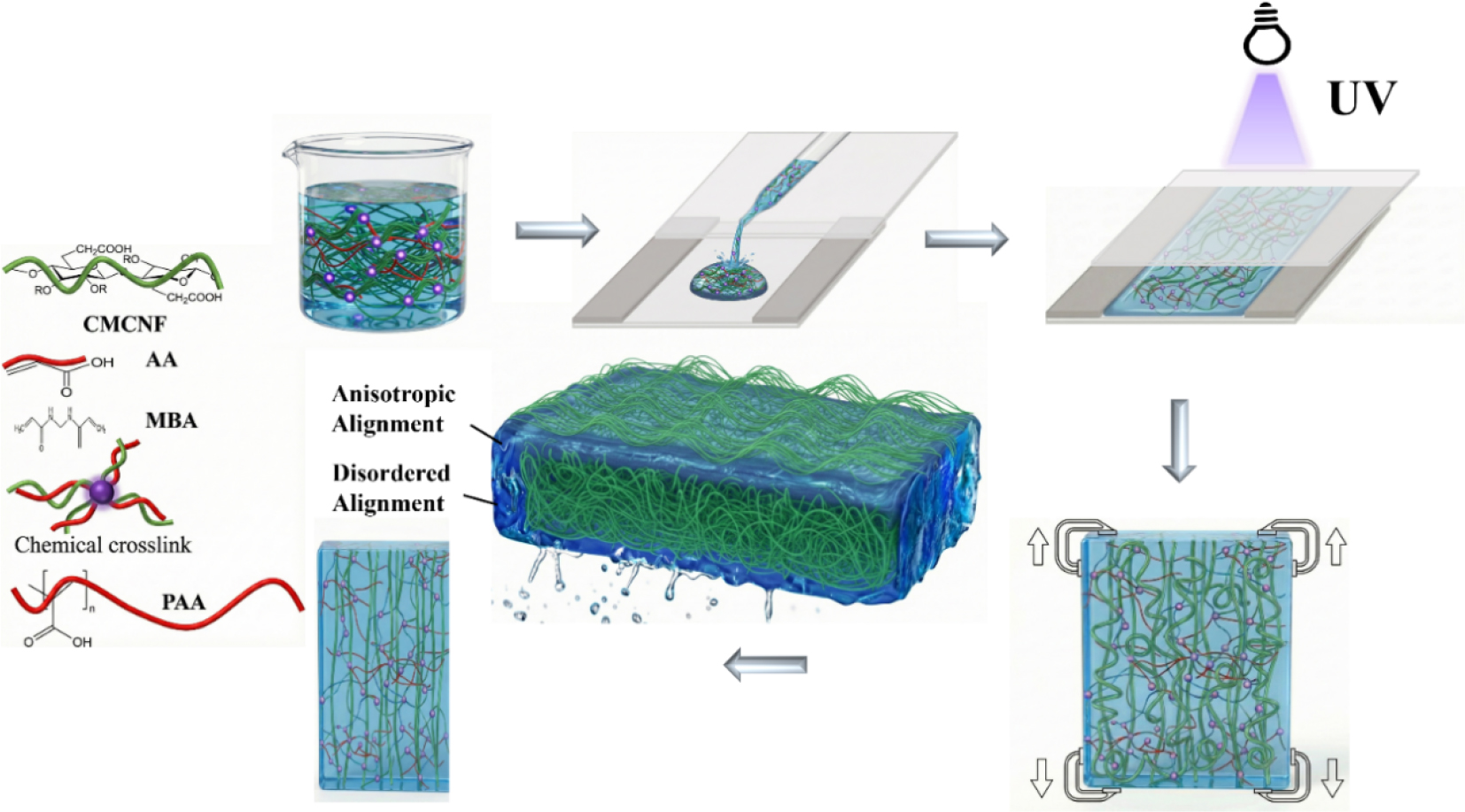
1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Preparation of carboxymethylated cellulose nanofibrils
2.3 Precursor preparation
2.4 Fabrication of PAA/CMCNF hydrogels
2.5 Structural characterization
2.6 Mechanical characterization
2.7 pH-dependent swelling and anisotropy
2.8 Cyclic stability and actuation performance
3. Results and Discussion
3.1 Fabrication strategy and mechanism
3.2 Microstructural evolution and anisotropy
3.3 Structural gradient and shape-morphing behavior
3.4 Chemical composition and crystallinity
3.5 Mechanical testing
3.6 pH-responsive swelling
3.7 Programmable actuation and soft robotics
4. Conclusions
1. Introduction
Stimuli-responsive hydrogels [1,2] are able to alter their physicochemical properties in response to environmental triggers such as pH [3,4], temperature [5], and light [6,7], rendering them promising candidates for biomedical systems [8], soft robotics [9], and wearable devices [10]. Among these systems, pH-responsive hydrogels are particularly appealing for drug delivery [11,12], biosensing [13], and artificial muscles [14] owing to the well-defined pH gradients present in numerous physiological and operational environments. However, most conventional pH-responsive hydrogels possess isotropic network architectures, which generally result in slow solvent diffusion, limited mechanical strength, and poorly controlled deformation. These limitations hinder their use in applications requiring rapid response and programmable actuation.
To address these limitations, introducing anisotropy through the alignment of internal microstructure alignment has emerged as an effective strategy [15]. Similar to natural tissues (e.g., tendons and plant cell walls), aligned networks can generate direction-dependent mechanics and deformation. For example, anisotropic hydrogel sheets reinforced with cellulose nanofibrils (CNFs) can exhibit strong in-plane reinforcement together with large out-of-plane deformation [16]. Fibrillar hydrogel actuators have been reported to expand uniaxially by 60–100 times with an initial rate of 100–130% s-1, clearly outperforming isotropic gels. Nevertheless, many established routes for constructing anisotropy, including directional freezing [17], electric/magnetic field alignment [18,19], and 3D printing [20], either require specialized equipment or are difficult to scale up [21]. Although these strategies are effective, they are not always suitable for routine, large-area, or high-throughput fabrication. Recent reviews have also highlighted that cellulose-based hydrogels and nano-scale cellulose materials offer strong sustainability and functionality, but further advances in process simplicity, mechanical robustness, and scalable fabrication are still needed [22,23,24].
Mechanically guided alignment has attracted attention as a simpler alternative. Shear-induced orientation [25], a concept borrowed from polymer processing, can align nanocellulose and thereby introduce anisotropy into hydrogel systems. Yang et al. [26] showed that shear flow aligned cellulose nanocrystals and increased the alignment degree by approximately 24% compared with vacuum filtration, leading to improved tensile properties. In parallel, pre-stretching has been widely used as a practical method to enhance fatigue resistance and stabilize network structures. Sun et al. [27] reported that pre-stretched poly(vinyl alcohol) hydrogels retained ~80% of the initial modulus after 1,000 cycles. However, shear alignment alone does not necessarily provide fast and reconfigurable stimulus response, whereas pre-stretching alone cannot create intrinsic structural anisotropy. This suggests that combining the two mechanical treatments may be necessary to obtain both high alignment and reliable pH-driven actuation.
Despite this potential, shear alignment together with pre-stretching has rarely been explored for pH-responsive systems [28,29]. Most existing studies focus on static mechanical reinforcement rather than on dynamic, repeatable pH-driven switching and programmable shape transformation. Accordingly, there remains a need for an integrated material design that couples anisotropic structure with rapid and reversible actuation [30].
In this work, a mechano-guided strategy is developed to fabricate anisotropic, pH-sensitive hydrogels through shear-assisted pre-stretching. Carboxymethylated cellulose nanofibrils (CMCNF) serve as an oriented template to direct the alignment of poly(acrylic acid) (PAA) chains. Following shear-induced orientation, a controlled pre-stretching step reorganizes and densifies the network, while hydrogen bonding, covalent crosslinking, and temporary Ca2+-assisted fixation help maintain structural integrity. By varying shear rate and pre-stretch ratio, the degree of microstructural anisotropy [31,32], pH-triggered swelling kinetics, and deformation behavior can be adjusted. Additionally, the introduced prestress improves cyclic stability under repeated pH switching [33]. Overall, this strategy provides a scalable route to constructing anisotropic hydrogels with programmable pH responsiveness for soft actuation and related applications.
2. Materials and Methods
2.1 Materials
Acacia hardwood sulfite chemical pulp (beating degree = 25°SR) were purchased from Rizhao Huatai Paper Co., Ltd. Acrylic acid (AA, 99%), N,N′-methylenebisacrylamide (MBA, crosslinker), Irgacure651 (initiator) were provided by Shanghai Maclin Biochemical Technology Co., Ltd., and hydrochloric acid (HCl, 0.1 M)/sodium hydroxide (NaOH, 0.1 M) solutions was supplied from the Tianjin Damao Chemical Reagent Factory. Deionized water was produced in the laboratory. All chemicals were used as received without further purification.
2.2 Preparation of carboxymethylated cellulose nanofibrils
CMCNF were prepared by first carboxymethylating the sulfite pulp fibers and then mechanically fibrillating the modified fibers [34]. Briefly, the carboxymethylated pulp was diluted to 1 wt% with deionized water. The resulting suspension was processed 15 times using Supermasscolloider (MKCA6-5J, Masuyuki, Japan) at 1,500 rpm and with a gap cleanliness of -100 µm. The pretreated cellulose dispersion was then further processed three times through 200 and 87 µm chambers of a high-pressure Microfluidizer (M-110EH-30, MFIC, USA) [35]. The degree of substitution (DS) of the CMCNF used in this study was 0.4651.
2.3 Precursor preparation
A homogeneous precursor solution was prepared on a total-mass basis by dispersing CMCNF (1.0 wt%), AA monomer (30 wt%), N,N′-methylenebisacrylamide (MBA, 0.1 wt% as crosslinker), and Irgacure 651 (0.05 wt% as photoinitiator) in deionized water. The mixture was stirred at 500 rpm for 2 h under a nitrogen atmosphere at room temperature (25 ± 2°C) to ensure homogeneity and remove dissolved oxygen.
2.4 Fabrication of PAA/CMCNF hydrogels
Shear-induced Alignment: The precursor solution was injected into a custom-designed rectangular mold composed of two parallel glass slides separated by 4 mm spacers. The representative hydrogel strips used for actuation tests had initial dimensions of 50 mm × 10 mm × 4 mm (length × width × thickness). To induce initial alignment of the CMCNF, the top slide was reciprocated horizontally for 50 cycles at a controlled shear rate (0–6 cm/s). Immediately after shearing, the precursor was subjected to UV polymerization for 10 min. Notably, the reaction temperature was strictly maintained at 0°C using a cooling stage to prevent the thermal relaxation of the oriented CMCNF networks. The resulting shear-aligned hydrogel was designated as CPS.
Pre-stretching and Stabilization: The as-prepared CPS hydrogel was clamped onto a uniaxial mechanical tensile stage and pre-stretched to target stretch ratios of 100–300% at a constant rate of 10 mm min-1. For example, a representative strip with initial dimensions of 50 mm × 10 mm × 4 mm was stretched to approximately 100 mm × 7 mm × 1.8 mm for the CPS-1 state. To help preserve the aligned structure and introduce prestress, the stretched hydrogel was immersed in a 0.5 M CaCl2 solution for 1 h while maintaining the strain. This step facilitated temporary ionic interactions between Ca2+ and carboxyl groups in the network. After CaCl2 treatment, the samples were gently rinsed with deionized water 1–3 times to remove loosely bound surface Ca2+ before subsequent characterization. Upon releasing the external load, the hydrogel retained its structural anisotropy. The final samples were labeled as CPS-X, where X denotes the pre-stretch ratio (e.g., CPS-1).
2.5 Structural characterization
The microstructural morphology of the anisotropic hydrogels was visualized using a field-emission scanning electron microscopy (FE-SEM). Prior to observation, the samples were freeze-dried and sputter-coated with a thin conductive gold layer. Chemical interactions within the hydrogel network were analyzed by Fourier transform infrared spectroscopy (FT-IR) in ATR mode using freeze-dried samples. To evaluate the macroscopic alignment of the CMCNF, the precursor solution and the resultant hydrogel were observed in situ using a polarized optical microscope (POM, Nikon Eclipse LV100-POL) equipped with a 10× objective lens under crossed polarizers. Birefringence () was determined at the center of the hydrogel using a Berek compensator and calculated according to Eq. (1):
where represents the optical retardation (nm) and is the sample thickness (nm).
2.6 Mechanical characterization
Uniaxial tensile tests were performed using a universal testing machine on dumbbell-shaped specimens (gauge length: 25 mm, width: 2 mm). The tests were conducted at a crosshead speed of 10 mm/min. To investigate mechanical anisotropy, tensile loads were applied both parallel and perpendicular to the CMCNF orientation direction. The elastic modulus, tensile strength, and elongation at break were determined from the resulting stress–strain curves. Additionally, the macroscopic load-bearing capacity was demonstrated by hoisting a standard weight at the end of the hydrogel strip.
2.7 pH-dependent swelling and anisotropy
The equilibrium swelling ratio (ESR) [36] was determined gravimetrically. Freeze-dried xerogels were weighed () and subsequently immersed in solutions with varying pH values (pH = 2, 7, and 12) for 24 h to reach swelling equilibrium. Surface water was gently removed with filter paper, and the wet weight () was recorded. The ESR was calculated using Eq. (2):
To quantify the structural anisotropy, the dimensional changes of the hydrogel upon swelling were measured using a vernier caliper (accuracy: 0.02 mm). The anisotropic ratio (AR) [37] was defined as the ratio of the dimensional swelling rate parallel to the alignment direction (SR∥) to that in the perpendicular direction ( SR⊥), as shown in Eq. (3):
2.8 Cyclic stability and actuation performance
The reversible pH-responsiveness was evaluated by alternately immersing the hydrogel in pH 2 and pH 12 solutions for 10 consecutive cycles, with 30 min per step to ensure sufficient solvent exchange and a stable swelling/deswelling state before switching to the next medium. For the actuation test, a hydrogel strip was immersed in pH 12 solution, and its bending behavior was recorded in real time using a high-speed camera combined with image analysis software. The response speed was defined as the time required for the strip to reach a stable curled state; in the representative sample, actuation was initiated within 0.5 s and the bending process reached a stable state within about 15–20 s.
3. Results and Discussion
3.1 Fabrication strategy and mechanism
Fig. 1 illustrates the fabrication process for the anisotropic CMCNF/PAA hydrogel. In brief, an aqueous precursor containing CMCNF, AA, crosslinker, and photoinitiator was injected into a rectangular mold formed by two parallel glass plates spaced 0.40 mm apart. Alignment was introduced through two consecutive mechanical steps. First, the upper glass plate was moved back and forth along the long axis of the mold to generate a shear field (approximately Couette flow). In this flow, the high-aspect-ratio CMCNFs experience hydrodynamic torque and align with the flow direction to minimize drag. The aligned structure was then frozen in situ by UV-induced polymerization of AA, embedding the oriented CMCNF framework within the resulting PAA matrix. Second, the cured hydrogel was uniaxially pre-stretched. This additional mechanical treatment further straightened the nanofibrils and densified the network, thereby enhancing both the degree of orientation and the overall toughness of the material. The formulation was selected to balance precursor processability and network reinforcement. In this study, the CMCNF content was fixed at 1.0 wt% (water content 62.5%). Based on our preliminary experimental optimization, at higher CMCNF loadings, the precursor became too viscous and the fibrils entangled strongly, which reduced the effectiveness of shear alignment. In contrast, at lower loadings, the fibril concentration was insufficient to establish a percolated anisotropic network, resulting in diminished mechanical reinforcement.
3.2 Microstructural evolution and anisotropy
The shear-assisted pre-stretching strategy effectively induced anisotropic alignment within the CMCNF/PAA network. To evaluate the effects of shear and pre-stretching, the hydrogels were observed by SEM and POM. As shown in Fig. 2a. The control sample (CP) showed a randomly oriented, rough surface morphology, indicative of an isotropic network. In contrast, the shear-aligned hydrogel (CPS) exhibited an incipient directional texture along the shear direction. After the subsequent pre-stretching step (CPS-2), the hydrogel displayed a highly ordered, wave-like fibrillar architecture, confirming that the combination of shear and mechanical stretching significantly enhances the orientation of the CMCNF/PAA network.
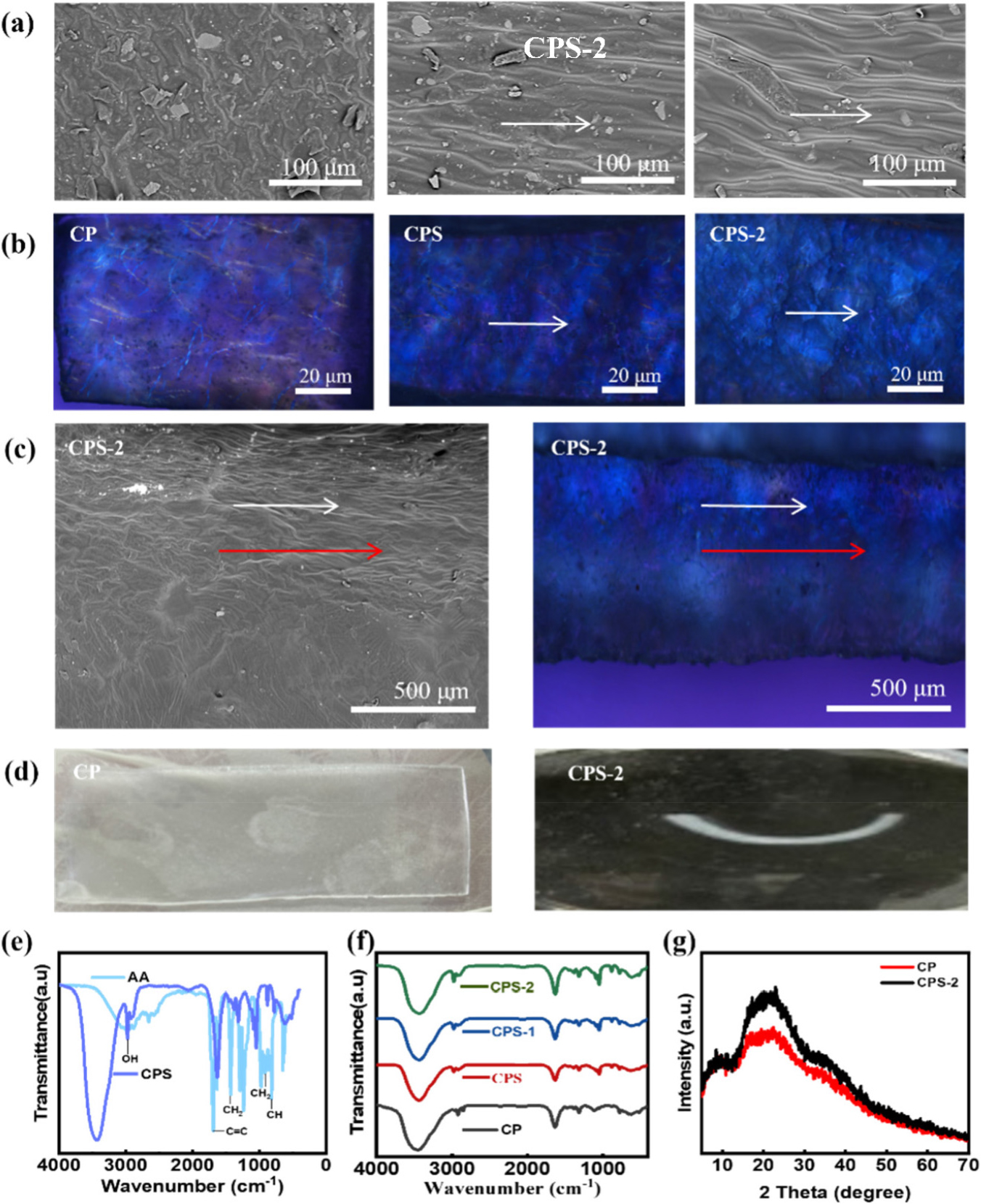
Fig. 2.
As shown in the SEM and POM images of CMCNF/PAA hydrogel (a, b), the orderly oriented structure of the surface could be obviously observed. (c) POM and SEM images of the cross-section of CPS-2. CNF alignment direction is shown with a white arrow. The red dashed line separates the regions with alignment (monodomain) and without alignment (polydomain) (d) Anisotropic shape-morphing of CMCNF/PAA hydrogel actuators with Shear and Pre-Stretching before and after. (e) FTIR spectra of CPS hydrogel and AA, (f) CP, CPS, CPS-1, and CPS-2 of hydrogel. (g) CP and CPS-2 of XRD.
This structural anisotropy was further corroborated by POM observations (Fig. 2b). Under crossed polarizers, the unsheared CP hydrogel remained completely dark, characteristic of an optically isotropic material with disordered CNF arrangement. Conversely, the CPS and CPS-2 samples exhibited distinct birefringence (interference colors), which shifted from low-order to high-order brightness with the application of stretching. These observations provide direct evidence for the macroscopic alignment of semi-crystalline CMCNFs within the hydrogel matrix.
3.3 Structural gradient and shape-morphing behavior
To clarify the bending mechanism during actuation, we examined the cross-section of CPS-2 (Fig. 2c). Both SEM and POM reveal a pronounced structural gradient through the thickness. The top region, which is in direct contact with the moving plate, exhibits strong alignment (appearing bright in POM and oriented in SEM), whereas the bottom region remains comparatively disordered (appearing dark). This gradient is reasonable because the shear stress decays across the gap, and the stress near the bottom plate can fall below the threshold required to reorient CNFs.
This asymmetric architecture explains the shape-morphing behavior in Fig. 2d. Aligned cellulose nanofibril framework restricts swelling along the alignment direction but allows greater expansion perpendicular to it. Because the aligned top layer constrains swelling more than the less ordered bottom layer, the mismatch in swelling strain generates an internal bending moment, causing the strip to curl toward the aligned side.
3.4 Chemical composition and crystallinity
The chemical formation of the hydrogel network was verified by FTIR spectroscopy. As shown in Fig. 2e, the spectrum of the polymerized CPS hydrogel was compared with that of the AA monomer. The polymerization of AA was confirmed by the marked weakening of the characteristic C=C stretching vibration peak at 1,634 cm-1 in the PAA spectrum. Additionally, the broad peak at 3,028 cm-1 (O-H stretching) became more pronounced in the hydrogel, indicating enhanced hydrogen-bonding interactions among the carboxyl groups. Fig. 2f further supports the presence of CMCNF in the hydrogel. While the bands at 3,295 cm-1, 2,913 cm-1, and 1,035 cm-1 correspond mainly to the cellulose backbone, the absorption bands appearing around ~1,600 cm-1 and ~1,420 cm-1 are assigned to the asymmetric and symmetric stretching vibrations of COO− groups, respectively, which are consistent with carboxymethyl modification. The influence of mechanical stretching on the XRD pattern was also examined (Fig. 2g). Compared to the CP sample, the CPS-2 hydrogel exhibited a slight increase in diffraction intensity. This change may reflect improved orientation and packing of the CMCNF/PAA domains after mechanical treatment.
3.5 Mechanical testing
3.5.1 Mechanical anisotropy and reinforcement
The hierarchically aligned structure endows the hydrogels with exceptional mechanical performance and distinct anisotropy. Fig. 3a and b present the tensile stress-strain curves measured parallel and perpendicular to the CMCNF alignment direction, respectively. The isotropic CP hydrogel exhibited weak mechanical properties with low tensile stress. In contrast, the shear-aligned and pre-stretched samples (CPS-0 to CPS-2) demonstrated a significant enhancement in mechanical strength. Notably, the tensile modulus and ultimate strength measured parallel to the alignment direction (Fig. 3a) were substantially higher than those measured in the perpendicular direction (Fig. 3b). This discrepancy highlights the critical role of CMCNF alignment: the oriented nanofibers effectively bear the load along the axial direction, serving as rigid reinforcement skeletons, whereas the load-bearing capacity is limited by the polymer matrix in the transverse direction.
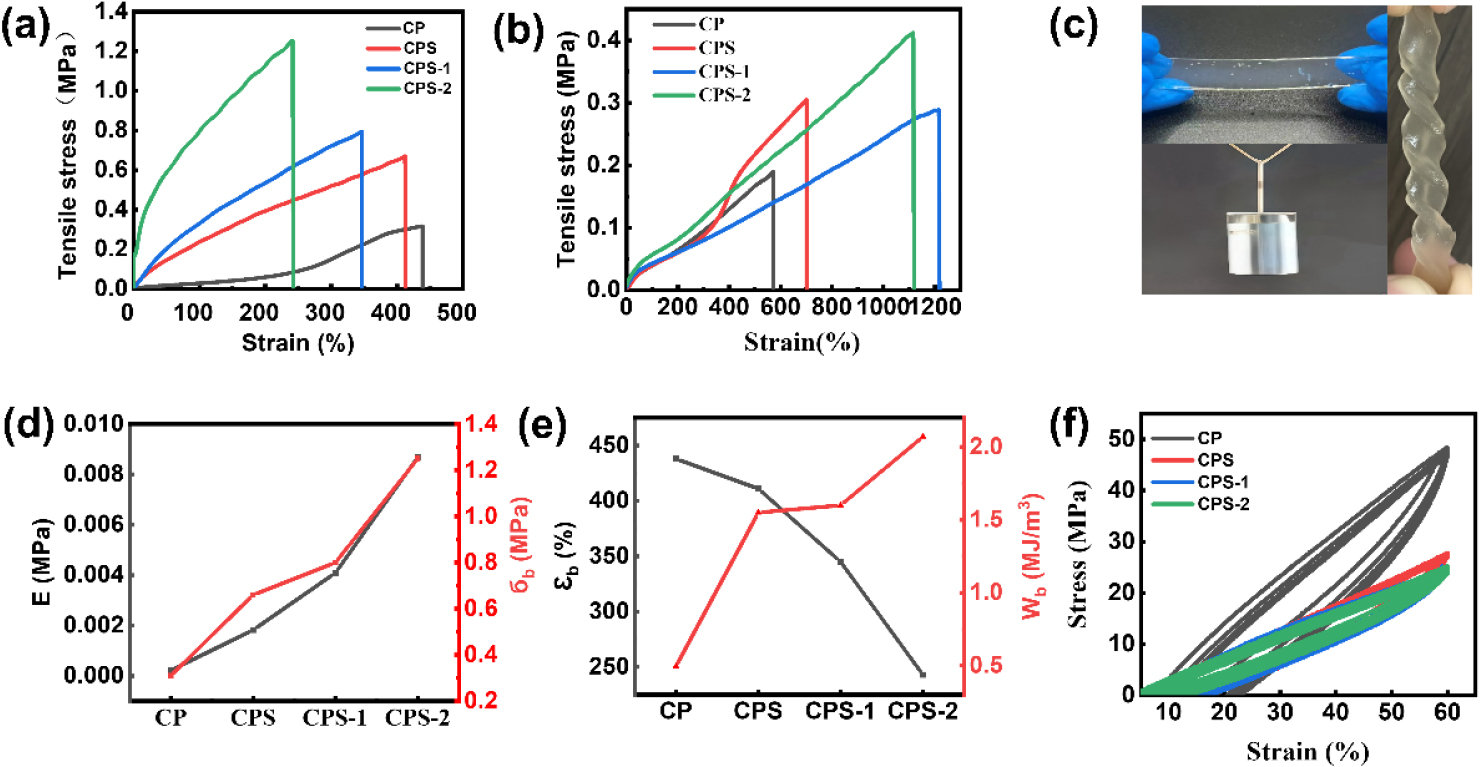
Fig. 3.
(a, b) Tensile stress-strain curves of CMCMNF/PAA hydrogels in the tensile direction versus perpendicular to the tensile direction when pre-stretched at 0 to 2. (c) Photograph showing that a hydrogel with a thickness of ~0.4 mm lifted a 500 g weight without crack propagation. (d, e) Comparison of mechanical parameters: (d) Elastic modulus (E) and tensile strength (); (e) Elongation at break () and work of extension (). (f) Consecutive cyclic loading–unloading curves of CP and CPS-2 hydrogels at a fixed strain of 60% (stretch rate: 10 mm min-1).
The mechanical robustness of the anisotropic hydrogel is visually demonstrated in Fig. 3c. The CPS-2 hydrogel (thickness ~0.4 mm) could withstand complex deformations, such as tight knotting and twisting, without fracture. Furthermore, it easily hoisted a 500 g weight, showcasing its high specific strength. These changes in mechanical parameters are summarized in Fig. 3d–e. With increasing pre-stretch ratios (from CP to CPS-2), the hydrogel network undergoes a transition from soft and ductile to stiff and strong. The Elastic Modulus (E) and Tensile Strength () exhibited a sharp increase. Specifically, the Elastic Modulus of CPS-2 significantly exceeded that of the neat CP hydrogel, attributed to the strain-induced alignment and densification of the polymer network. The Work of Extension (), representing toughness, also improved remarkably (Fig. 3e), indicating that the aligned structure can effectively dissipate energy and resist crack propagation. However, as expected for anisotropic materials, the Elongation at Break () decreased for CPS-2, reflecting the trade-off between stiffness and ductility.
3.5.2 Cyclic stability and elastic recovery
Cyclic loading–unloading tests (Fig. 3f) were carried out to evaluate the elasticity and fatigue resistance of the hydrogels. The CP sample exhibited a large hysteresis loop during the first cycle and showed a clear residual strain after unloading, indicating irreversible chain slippage and/or localized structural damage within the disordered network.
In contrast, CPS-2 displayed a much more elastic response. The hysteresis loops are narrow and nearly overlap over repeated cycles, and the residual strain is negligible. This result is consistent with the role of pre-stretching as a “mechanical annealing” process that removes network slack and stabilizes the aligned structure, which is highly beneficial for repeated actuation.
3.6 pH-responsive swelling
3.6.1 pH-responsive swelling behavior
The pH-responsive behavior of the hydrogel originates from the abundant carboxylic acid groups (–COOH) within the PAA network. As illustrated in Fig. 4a, the ionization state of these groups is governed by the external pH. At low pH (pH < pKa ≈ 4.7), the carboxylic groups remain protonated, promoting the formation of intramolecular hydrogen bonds, which leads to network shrinkage. At high pH (e.g., pH 12), the –COOH groups deprotonate into negatively charged carboxylate ions (–COO−). The resulting strong electrostatic repulsion between polymer chains, coupled with increased osmotic pressure, drives significant water absorption and network expansion. As shown in Fig. 4b, the swelling ratio increased dramatically as the pH increased from 2 to 12. This trend confirms the strong pH dependence of the hydrogel network. A slight anisotropic swelling behavior was also observed in the CP sample, likely due to weak structural bias introduced during casting, with swelling in the transverse direction being slightly greater than that in the longitudinal direction.
3.6.2 Anisotropic swelling kinetics and actuation mechanism
For the anisotropic CPS-2 hydrogel, swelling becomes clearly direction-dependent (Fig. 4c). Expansion perpendicular to the fibril alignment is much larger than expansion parallel to it. This is the expected “corset effect” of aligned CMCNFs: the stiff nanofibrils constrain chain extension along the alignment direction while leaving more freedom for transverse swelling.
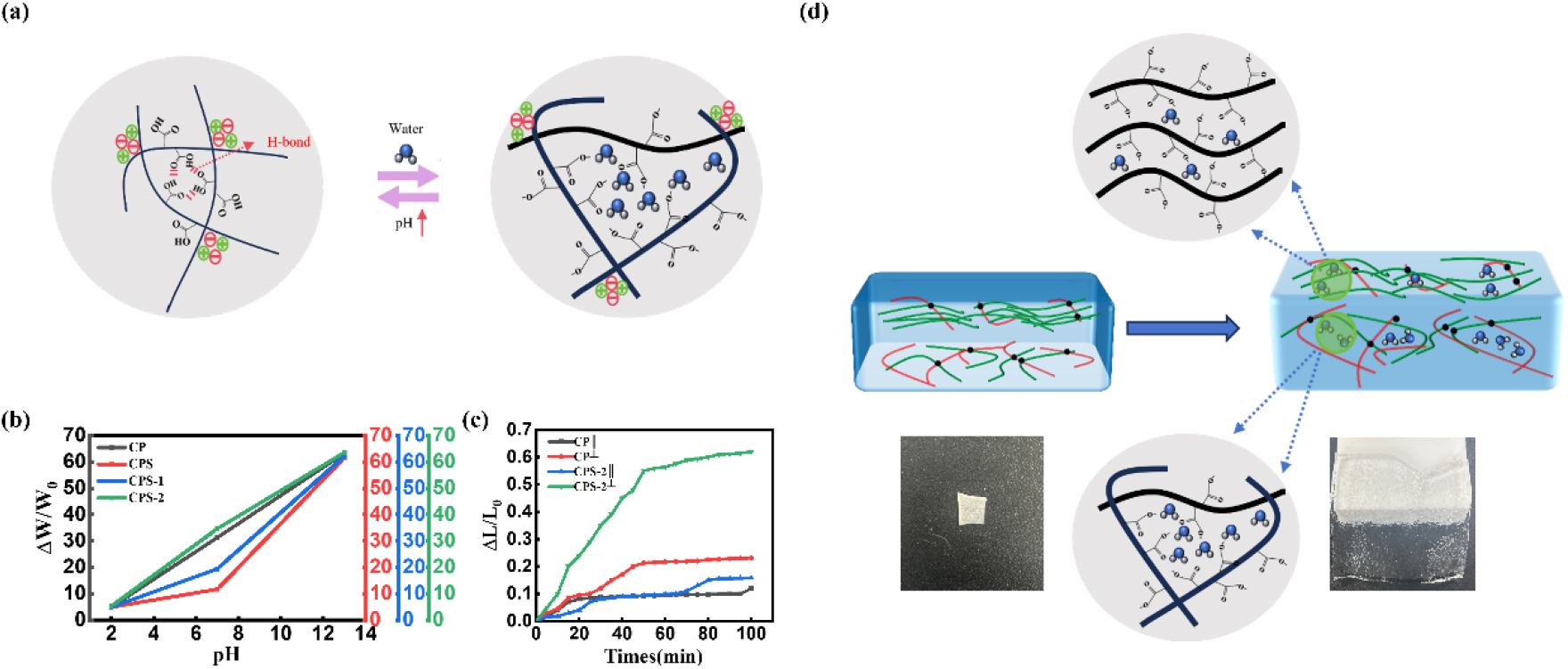
Fig. 4.
pH-responsive swelling behavior and actuation mechanism. (a) Schematic illustration of the swelling/deswelling mechanism driven by the protonation/deprotonation of carboxylic groups. (b) Equilibrium swelling ratios of CP and CPS hydrogels at pH 2, 7, and 12. (c) Time-dependent swelling kinetics of CP and CPS-2 hydrogels measured parallel (∥) and perpendicular (⊥) to the alignment direction. (d) Schematic diagram explaining the structural gradient-induced anisotropic actuation and accelerated water diffusion in aligned nanochannels.
Fig. 4d summarizes the origin of the rapid and programmable actuation behavior. First, the aligned nanofibrils form ordered nanochannels that facilitate solvent transport relative to the isotropic network, thereby accelerating the pH response. Second, the structural gradient across the thickness creates an intrinsic asymmetry in swelling. Under alkaline conditions, the aligned top layer resists expansion more strongly than the less ordered bottom layer, resulting in a mismatch in swelling strain and the generation of an internal bending moment. This stress gradient drives the strip to curl reproducibly toward the aligned side.
3.7 Programmable actuation and soft robotics
The reversible actuation behavior of the hydrogel was evaluated under alternating alkaline and acidic conditions (Fig. 5a). Upon immersion in NaOH solution (pH 12), the hydrogel strip rapidly curled into a “C” shape configuration as a result of anisotropic swelling across its thickness. When transferred to HCl (pH 2), protonation of the network induced deswelling, allowing the strip to recover nearly to its original straight shape. The actuation response was initiated within about 0.5 s, and a stable curled configuration was typically reached within 15–20 s under alkaline conditions. After 10 pH-switching cycles, the actuator retained approximately 92% of its initial bending amplitude. This fast and repeatable switching behavior demonstrates the material’s suitability for cyclic actuation.
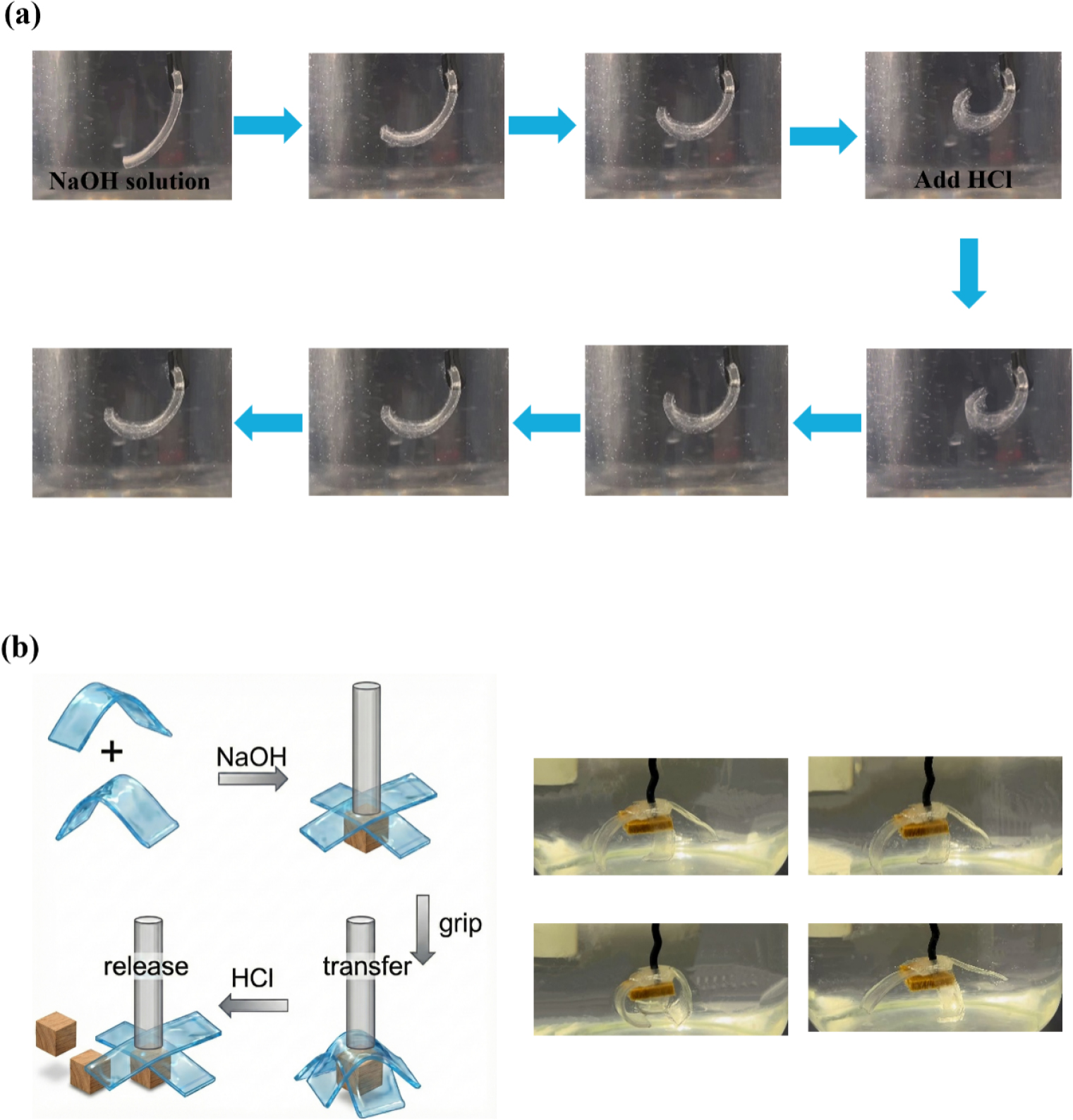
Fig. 5.
Programmable actuation and soft robotic applications. (a) Optical images demonstrating the reversible bending/unbending actuation of a hydrogel strip triggered by alternating immersion in NaOH (pH 12) and HCl (pH 2) solutions. (b) Demonstration of a pH-responsive soft micro-gripper: schematic illustration (left) and real-time snapshots (right) showing the capture, transport, and release of a cargo.
As a practical demonstration, two anisotropic strips were assembled into a pH-responsive soft micro-gripper (Fig. 5b). The alignment direction of each strip was designed such that both arms bent inward during swelling, enabling chemically triggered grasping and release. Under alkaline conditions, the gripper arms curl inward to capture a lightweight spherical object. The closed gripper can then transport the cargo, and subsequent exposure to acidic solution reversed the swelling and reopened the arms, enabling controlled release. Owing to the intrinsic compliance of the hydrogel, the gripper can adapt more effectively to irregular and delicate objects than rigid gripping systems.
Overall, compared with previously reported anisotropic hydrogels prepared by directional freezing, external-field alignment, or multi-material assembly, the present shear-assisted pre-stretching strategy provides a relatively simple and scalable route to construct oriented cellulose-based hydrogel networks. Existing studies have demonstrated that anisotropic structures can improve directional mechanics, sensing, or shape-morphing performance, but fewer reports have coupled straightforward mechanical processing with reversible pH-responsive actuation in a single cellulose-reinforced system. In the present work, shear-induced fibril alignment and post-stretch reorganization work together to enhance mechanical anisotropy, swelling asymmetry, and cyclic actuation stability. This combination makes the material distinct from conventional isotropic pH-responsive hydrogels and highlights the potential of mechanically programmed nanocellulose architectures for soft actuator design.
4. Conclusions
In summary, we developed a scalable and practical strategy for engineering anisotropic pH-responsive hydrogels by combining shear-induced alignment with programmed mechanical pre-stretching. This integrated approach enabled anisotropic alignment of CMCNFs within the PAA network and generated an anisotropic architecture with enhanced directional mechanical reinforcement and swelling asymmetry. The resulting hydrogels showed rapid pH-triggered actuation, with an initial response observed within 0.5 s and a stable curled state reached within about 15–20 s, while retaining about 92% of the initial bending amplitude after 10 switching cycles. A soft micro-gripper was further constructed to demonstrate the functional potential of the material in reversible, chemically controlled cargo manipulation. Owing to its simple fabrication, scalability, and compatibility with complex geometries, this strategy offers a versatile platform for cellulose-based soft actuators, adaptive devices, and related biomimetic systems.