1. Introduction
Cellulose is a natural polymer and is thus a biodegradable, biocompatible, and renewable material that can be synthesized from various natural sources, such as plants, algae, bacteria, fungi, and marine animals. Nanocellulose, which is known as a cellulose nanofibril (CNF)—also called microfibrillated cellulose, cellulose nanocrystal, and bacterial cellulose—is an interesting nanomaterial derived from native cellulose. It has unique biological, physical, and chemical properties and thus has many potential applications in various commercial products and services, such as hydrogels, food additives, cosmetics, tissue engineering, composites (coatings, films, and foams), biomedicals, drug delivery, coating materials, rheology modification, and other functional materials.1-6)
A CNF is nanostructure cellulose with a diameter of less than 100 nm and a length of 0.76 μm to ≥4 μm; the sizes of the fibrils vary depending on pretreatment, plant sources, and the fibrillation process. 7) There are several ways to produce CNFs through a high mechanical delamination process using grinding, high-pressure homogenization, micro-fluidization, and a conventional high-speed blender.8,9) However, mechanical fibrillation requires high energy consumption. Therefore, chemical pre-treatments, such as tempo-mediated oxidation,10) carboxymethylation,11) oxidative sulfonation, 12) and quaternization13) are required to help reduce the energy consumed during the fibrillation process. By subjecting CNFs to chemical pre-treatment, the cohesion between microfibrils due to hydrogen bonding can be reduced by introducing charge groups that generate electrostatic repulsion between fibrils.14)
The rheological properties of CNF suspensions have been previously studied.15-20) For example, CNF suspensions exhibit high viscosity with shear-thinning behavior. Moreover, increasing CNF suspensions to between 0.3% and 2.6% in turn increases the storage moduli and loss moduli.21) Furthermore, an investigation on the impact of divalent and trivalent cations on carboxylated CNFs shows that the valency of the metal cations and the strength with which they bind to carboxylate play an important role in the storage moduli of hydrogels.22) It is worth noting that the addition of cations into the cellulose suspension significantly affects hydrogel strength.
In this study, the rheological properties (shear viscosity, strain amplitude, and frequency) of CNFs with a monovalent salt addition were examined. Only a limited number of publications have focused on the rheology of CNFs with added monovalent salts, specifically 0-100 mM sodium chloride (NaCl).21,23) To the author’s knowledge, the rheology of CNFs reacted with monovalent salts NaCl, potassium chloride (KCl), and lithium chloride (LiCl), particularly in concentrations of 500-10,000 ppm, has not been reported.
2. Materials and Methods
2.1 Materials
The material used for CNF preparation was dried kraft pulp obtained from M. Company. Chemicals were purchased from various suppliers: Ethanol (EtOH) from Samchun Pure Chemical Co., Ltd., Korea; sodium hydroxide (NaOH) from OCI Company Ltd., Korea; monochloroacetic acid (MCA) from Denak Co., Ltd., Japan; NaCl from Junsei Chemical Co., Ltd., Japan; anhydrous LiCl from Samchun Pure Chemical Co., Ltd., Korea; and KCl from Duksan Pharmaceutical Co., Ltd., Korea. Distilled water was obtained from a water purification system in the Department of Wood and Paper Science at the Chungbuk National University.
2.2 Preparation of CNF
Dried kraft pulp (100 g) was soaked in ethanol containing NaOH (14.11 g) and stored at room temperature for 1 h. MCA (28.9 g) was dissolved in EtOH corresponding to 0.6 mmol/g cellulose then added slowly to the mixture; the reaction was conducted at 75℃ for 2 h. To obtain cellulose fibers, the reaction mixture was then washed and filtered several times with distilled water until neutral. The cellulose fiber was then mixed with distilled water to create 2% w/w CNF.
CNF was prepared from the fiber suspension by being passed through a GEA Homogenizer processor (Panda PLUS 2000, GEA, Italy). First, the fiber suspension at a concentration of 2% w/w was ground in two passes. Then, the fiber suspension was homogenized in two passes at a pressure of 600-800 bar. To obtain 1.5% CNF for further experiments, 2% CNF was diluted. Overall procedure for experiment was summarized in Fig. 1.
2.3 Preparation of CNF gels
CNF hydrogels were prepared by adding monovalent salt solutions of NaCl, KCl, and LiCl to the CNF suspension to obtain concentrations of Na+, K+, and Li+ ranging from 500 to 10,000 ppm. Approximately 2% CNF was diluted with a metal chloride solution to reach 1.5% CNF, and the mixtures were homogenized by shaking. The descriptions and compositions of the samples are listed in Table 1.
Table 1.
Descriptions and compositions of cations added to the gel
| Cations | Cation charge number | Cation radius | Cation concentration (mg/L) | |||
|---|---|---|---|---|---|---|
| 10,000 ppm | 5,000 ppm | 1,000 ppm | 500 ppm | |||
| Na+ | 1+ | 1.0 | 25,000 | 12,500 | 2,500 | 1,300 |
| K+ | 1+ | 1.4 | 19,000 | 9,500 | 1,900 | 1,000 |
| Li+ | 1+ | 0.6 | 48,000 | 24,000 | 4,800 | 2,400 |
Ionic radii were taken from the article of Luo et al.24)
2.4 Rheological characterization
Rheological measurements were conducted using the rheometer (MCR 102, Anton Paar, Austria) with a parallel plate (25 mm diameter; 1 mm gap). All the measurements were conducted at 25℃. Detailed descriptions of rheological measurements can be found in other studies.19) The rheometer was used to measure the viscosity, amplitude sweep, and frequency sweep of the samples. The apparent viscosity was measured by decreasing the shear rate from 100 to 1 s-1. Amplitude sweep was performed to determine the linear viscoelastic (LVE) region at a frequency of 10 rad/s (shear strain ranging from 0.01% to 100%). Following this experiment, the frequency sweep was performed at 0.5% strain at 0.1-100 rad/s.
3. Results and Discussion
CNFs gels can be obtained with different compositions of treatments using cations, as described in Table 1. Table 1 also presents the charge numbers and cation radii of the added salts. The rheological properties were an important parameter to assess the application of the CNF product. As such, the rheological properties (in particular, the shear rate and apparent viscosity) of CNFs with added cation solutions were examined.
The variation of the viscosities and shear rates of 1.5% CNFs with cations are summarized in Figs. 2-4. These figures show that the addition of cations increased the viscosity of the hydrogel. The apparent viscosities of CNFs with added salt were higher than those of CNFs without added salt.
Fig. 2 demonstrates the effect of various concentrations of added sodium on viscosity. As is evident, the viscosity increased significantly as the concentration of sodium increased up to 10,000 ppm. Similarly, increasing the concentration of NaCl resulted in an increased viscosity.25) The sodium in the MFC suspension influenced the viscosity because the fibril-fibril interaction associated with hydrogen bonds increased.16) However, the opposite effect was observed when the concentration of NaCl was increased (NaCl>1 mM, ≈23 ppm): The viscosity decreased significantly.21) In this study, the addition of high concentrations of sodium (1,000 to 10,000 ppm) prevented the gel form network from coalescing well; thus, the gel structures became stiffer. These conditions caused the water release from the plate during flow behavior measurement as the shear rate increased. This is evidence that the sodium concentration significantly affected gel strength. Furthermore, Tandjawa et al., who investigated the rheological properties of MFC suspensions, proposed increasing the viscosity by increasing the cellulose concentration.25)
Viscosity as a function of shear rate of 1.5% CNF with K+ at various concentrations is shown in Fig. 3. The potassium concentration had an effect similar to that of sodium: Increasing the potassium concentration resulted in increased viscosity. The viscosity increased from 34.4 Pa·s at a low concentration (500 ppm) to 189.2 Pa·s at a high concentration (10,000 ppm). The gel strength of the CNF clearly increased because of the addition of sodium and potassium. However, samples containing potassium showed lower gel strength than those containing sodium.
The apparent viscosity of 1.5% CNF with various concentrations of Li+ is presented in Fig. 4. The viscosity of all samples gradually increased as the lithium concentration increased. Compared with the other samples, samples containing Li+ had higher viscosities regardless of concentration. This indicates that the gel strength of CNF with lithium is much higher than that with sodium or potassium (Li+>Na+>K+). This is maybe due to the denser aggregation of surfactant molecules in the membranes. It was reported that increasing the salt concentration induced stronger gelation, which thus increases viscosity.26) All samples exhibited low shear-thinning behavior with a monotonically decreased viscosity and an increased shear rate, which is similar to the results reported by Li et al.27) The results in this study indicated that the viscosity of the CNFs with added cation is significantly dependent on shear rate, cation addition, and cation concentration.
Amplitude sweep was performed to determine the LVE region of the hydrogel and evaluate gel strength. Gel strength was evaluated by comparing the values of the storage and loss moduli as a function of strain amplitude. The amplitude measurements of CNFs with various concentrations of cations are shown in Figs. 5-7. The storage and loss moduli as functions of strain amplitude for 1.5% CNFs with Na+ at various concentrations are presented in Fig. 5. The storage and loss moduli of CNFs increased from 1,505.8 Pa to 6,497.1 Pa as concentrations of sodium ions increased from 500 to 10,000 ppm, respectively. The amount of cation used as a cross-linker has a significant effect in that it increases the elastic moduli, which means a stronger fibrous network is formed.28) Similar behavior was reported by Maestri et al.23): The storage modulus of the carboxylated CNC increased when the concentration of NaCl increased from 10 to 100 mM. Contrary to this study, Naderi et al.21) reported an increase in the storage modulus when the concentration of NaCl decreased from 0.1 to 10 mM. Fig. 6 illustrates the effect of various concentrations of potassium on gels. Similar to the effect of sodium, increasing the potassium concentration increased the storage and loss moduli, indicating that the strength of the gels increases as the concentration increases. As shown in Fig. 7, the storage and loss moduli of CNFs with added lithium follow trends identical to those of CNFs with sodium and potassium ions. Variations of the elastic modulus of 1.5% CNF as a function of cation concentration also revealed decreased strength at low concentrations and increased strength at high concentrations.
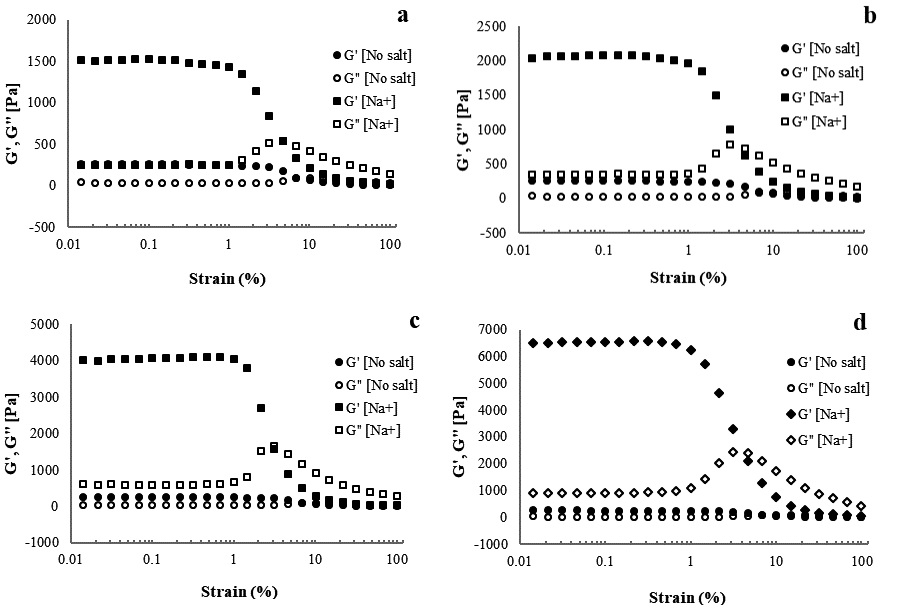
Fig. 5.
Storage and loss moduli as functions of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with Na+ at concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
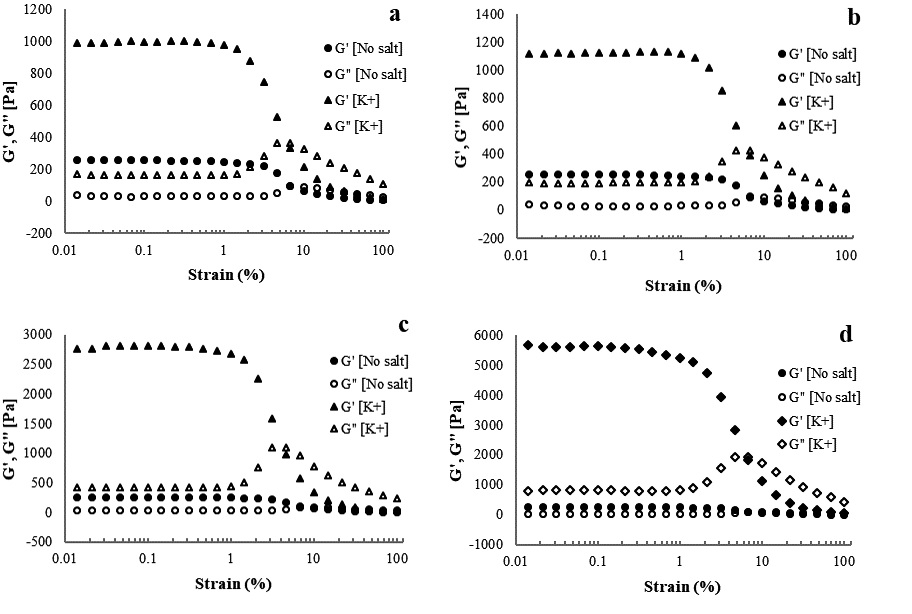
Fig. 6.
Storage and loss moduli as functions of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with K+ at concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
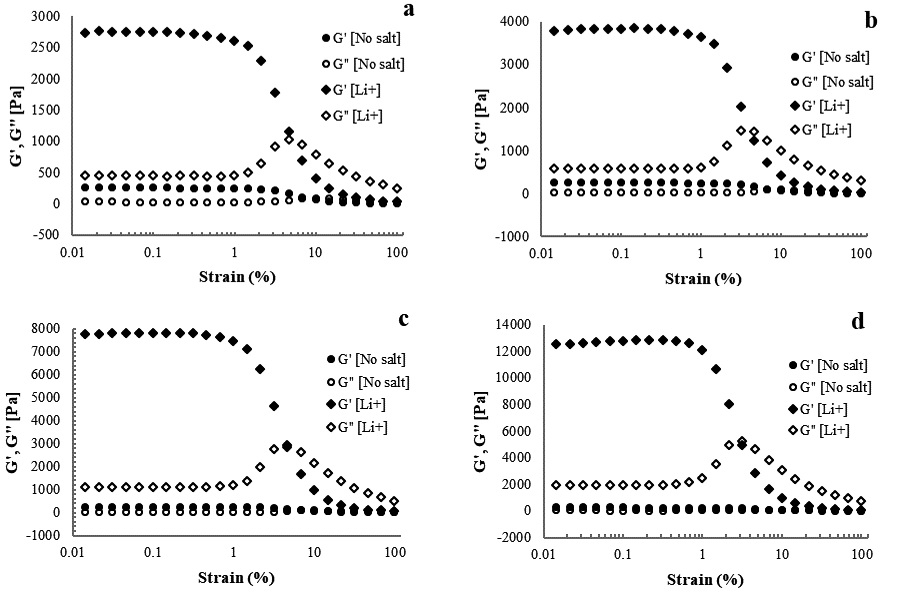
Fig. 7.
Storage and loss moduli as functions of strain amplitude at a frequency of 10 rad/s for 1.5% CNF with Li+ at concentrations of (a) 500, (b) 1,000, (c) 5,000, and (d) 10,000 ppm.
Oscillatory measurement was used to determine the LVE region and viscoelastic behavior. It also provides information regarding hydrogel characteristics and the degree of crosslinking.26) The G′ value at a lower frequency (0.1 rad/s) was used to compare the degree of cross-linked of different samples. We investigated the storage and loss moduli as functions of the angular frequency of CNFs with different cation concentrations, as shown in Table 2. Table 2 demonstrates that the increase of the ionic radii of the cations in CNF gels led to a decrease in the G′ and G″ (Na+<K+<Li+). This trend is different from that of CNC gels with added cations, in which the G′ and G″ increased as the ionic radii of cations increased.29) The higher the G′ value, the higher the degree of crosslinking, which will cause the material to stiffen. Furthermore, the ratio of G′ was much higher than that of G″, indicating the former’s solid-like structure, which means the CNFs with added cations have increased elasticity.
Table 2.
Rheological properties of 1.5% CNFs at different cations
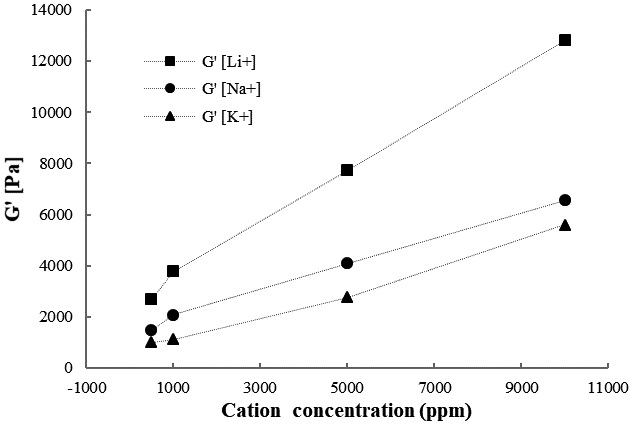
The storage modulus as a function of different cation concentrations is presented in Fig. 8. In all samples, the storage modulus increased as the cation concentration increased, indicating that the gel is stronger at higher concentrations. Assuming that cations induce fibril aggregation and interfibrillar bridging at higher concentrations, the network stiffens.30) We noticed that there were differences between the CNF gels after the addition of Li+, Na+, and K+. The suspension without cation was weaker compared with those with added cations. Furthermore, the storage moduli of the CNF gels follow the order of Li+>Na+>K+, indicating that Li+ increases gel strength. The difference in ionic radii among Li+, Na+, and K+ ions significantly affects hydrogel strength. Regarding the specific differences among Li+, Na+, and K+, our results demonstrate that, overall, low hydrogel strength occurs when cations with larger radii, such as K+ and Na+, are added compared with the addition of Li+, which has a smaller radius. Lithium has a very hard cation and thus lower in polarizability than sodium and potassium. However, soft cation, such as potassium has high polarizability, less ionic interaction, and more covalent in nature.31)
4. Conclusions
In this study, we examined the rheological properties of monovalent ions (LiCl, NaCl, and KCl) in 1.5% CNF at concentrations of 500, 1,000, 5,000, and 10,000 ppm. We found that cation type and concentration have a significant impact on the rheology of CNF gels. Adding Li+, Na+, and K+ at concentrations of 500, 1,000, 5,000, and 10,000 ppm gradually increased the viscosity, storage moduli, and loss moduli, which indicate an increased hydrogel strength. Regarding hydrogel strength, we found that Li+>Na+>K+ because of different cation radii and interfibrillar bridging on the hydrogel. Further research is necessary to determine the maximum strengths of hydrogels with added cations.