1. Introduction
The structural characteristics of lignocellulosic materials form a strong native recalcitrance to enzymes, which results in the relatively low digestibility of raw lignocellulosic materials. An appropriate pretreatment method is required to overcome this recalcitrance and make polysaccharides easily available for enzyme digestion.1)
Since the raw lignocellulosic materials exhibit strong recalcitrance to enzymes, a suitable pretreatment process is required to facilitate the access of enzymes to the plant polysaccharides. Based on the lignocellulosic material’s structure, as well as the factors that restrain the enzymatic hydrolysis of lignocellulosic materials, a good pretreatment method should weaken the factors that restrain the enzymatic hydrolysis of lignocellulose as much as possible on the premise of economy. Moreover, alkali pretreatment is the most commonly used method to remove lignin and hemicelluloses from lignocellulosic materials and disperse bulk lignocellulosic materials into lignocellulosic fibers. During alkaline pretreatment, the ester linkages in hemicelluloses and lignin are easily broken down. The ether linkages in hemicelluloses and lignin can also be destroyed at a relatively high temperature. The cleavages of these linkages significantly promote the solubilization of hemicelluloses and lignin, lead to exposing the cellulose to enzymes. Alkali pretreatment can alter the structure of the lignocellulosic matrix via two approaches: (1) swelling cellulose; and (2) dissolving lignin and hemicelluloses. After swelling, the crystalline structure of cellulose is interrupted, leading to the significant decrease in lignocellulosic biomass crystallinity. Alkali pretreatment at a high temperature breaks down lignin and hemicelluloses into soluble fragments and exposes the cellulose to enzymes.2)
The enzymatic hydrolysis of cellulose to glucose has received increased interest over the last decades, and a growing demand for economically sustainable biofuels indicates an urgent need for cost reduction during their production. Cellulose is hydrolyzed to glucose via the synergistic action of several enzymes. Endoglucanases (EC 3.2.1.4) break down cellulose chains at random positions within the chains, whereas exoglucanases (i.e., cellobiohydrolases, EC 3.2.1.91) cleave off cellobiose specifically from the chain ends recessively.3) Cellobiose is subsequently converted into glucose by β-glucosidase (EC 3.2.1.21).4) The exo-endo synergism is explained by the fact that endoglucanases provide more chain ends for cellobiohydrolases to act upon.5) After an initial phase of adsorption of cellulases on cellulose, which is fast compared to hydrolysis,6) the enzymes cleave off cellobiose and move along the same chain, hydrolyzing glycosidic bonds until an event occurs that terminates cleavage. As the reaction proceeds to intermediate degrees of conversion, the rate of the reaction decreases dramatically, and the final part of cellulose hydrolysis requires an inordinate fraction of the overall total reaction time.7) Several factors, both substrate- and enzyme-related, are suggested to be responsible for this slowdown of the reaction rate, but so far, no mechanistic explanation of the slowdown has been validated. The substrate characteristics often implied in the slowdown of the reaction rate include surface area, porosity, the degree of polymerization, crystallinity, and the overall composition (complex substrates, such as lignocellulosic materials versus pure cellulose). The initial degree of cellulose crystallinity plays a major role in determining the hydrolysis reaction rate. A completely amorphous sample is hydrolyzed much faster than a partially crystalline cellulose,8) and this concept led to the idea that amorphous domains in a partially crystalline cellulose sample are hydrolyzed first, leaving crystalline parts to be hydrolyzed at the end. Then it results in an increased crystallinity index (CrI), which explains the dramatic drop in rate at higher degrees of conversion.9) Additionally, the difference in reactivity between crystalline and an amorphous celluloses was found to be related to the adsorption capacity of endoglucanases on both types of substrates.10) Furthermore, the degree of cellulose crystallinity influences adsorption at a given protein loading, and the maximum adsorption constant was shown to be greatly enhanced at low-crystallinity indices.11)
Hemicelluloses and lignin are closely associated with cellulose through non-covalent and covalent linkages, and the content and distribution of hemicelluloses and lignin can also inhibit the enzymatic hydrolysis of lignocellulose.12) The hemicelluloses and lignin largely prevents enzymatic contact with cellulose, resulting in the very low enzymatic hydrolysis rate of raw materials. Furthermore, the non-productive binding of enzymes to lignin also restricts cellulose hydrolysis.13) Removing lignin and hemicelluloses can increase the pore size and accessible surface of the lignocellulosic materials and reduce the nonproductive adsorption of enzymes to lignin. Therefore, the removal of hemicelluloses and lignin can significantly improve the digestibility of lignocellulosic materials. Several pretreatment processes are aimed to reduce the recalcitrance of lignocellulosic materials by removing hemicelluloses and/or lignin.14)
This study evaluates the effects of glucose yield as a function of delignification under various alkaline pretreatment conditions. Finally, alkaline pretreatment has been employed to investigate the effects of delignification on the final glucose yield in the enzymatic conversion of Eucalyptus lignocellulose. This study may provide an insight about hardwood substrates and how to effectively use hardwoods as a source of bioethanol production.
2. Materials and Methods
2.1 Materials and chemicals
Physically pretreated Eucalyptus pellita that was twin-extruded at Korea Research Institute of Science and Technology (KRIST) were used as a substrate in the experiments. Eucalyptus wood chips were used after extrusion using a twin-screw extruder (Baker Perkins, MPC30, L / D = 13). After adding dry biomass and distilled water in a ratio of 1:10, twin-screw extrusion treatment was performed. Then, the same amount of distilled water was added and dehydrated using a filter press for 15 minutes. The recovered sample was used as a sample for pretreatment and saccharification without special drying. The water content of the sample subjected to twin-screw extrusion was 56.3%. The chemicals were purchased from Sigma- Aldrich. Furthermore, the 18.2 M Ω deionized water of the highest grade available in the laboratory was used in the experiment. All enzymatic hydrolysis experiments were performed using commercial enzyme solutions from Novozymes Chemical Company (Denmark), namely Cellic® CTec2 and HTec2, which were special gifts from KRIST.
2.2 Alkaline pretreatment
A combination of different pretreatments, such as physical (twin extrusion) and chemical (sodium carbonate and calcium hydroxide) pretreatments, have been made. Different concentrations of Na2CO3 (1.2 and 0.6 M) and Ca(OH)2 (1 and 0.5 M) per gram of biomass were examined. For the preparation of 1.2 M Na2CO3 solution, 63 g of Na2CO3 was dissolved in 500 mL water, and 10 mL of this mixture was combined with 0.74 g (1 M) of calcium hydroxide and 10 mL of distilled water. The total concentration of Na2CO3 turns to 0.6 M, and the total concentration of calcium hydroxide turns to 0.5 M. In preparing the 0.6 M Na2CO3 solution, 31.5 g of Na2CO3 was dissolved in 500 mL water, and 10 mL of this mixture was combined with 0.37 g (0.5 M) of calcium hydroxide and 10 mL of distilled water. The total concentration of Na2CO3 turns to 0.3 M, and the total concentration of calcium hydroxide turns to 0.25 M. Twin-extruded pretreated samples were pretreated at a liquid (L):lignocellulosic material (W) ratio of 20:1 in the closed reactor for 0.5-1 hour at 160℃, 180℃, and 200℃ under constant stirring (150 rpm). After the alkaline treatment, the samples were neutralized using hydrochloric acid (2% (w/v)) before washing with distilled water.
2.3 Enzymatic hydrolysis
The alkaline pretreated samples were selected for enzymatic hydrolysis. Cellic® CTec2 and HTec2 were used for enzymatic hydrolysis. The pretreated fibers (1 g O.D.) were put in a 2.5 mL sodium citrate buffer at pH 5.5 and 1.3 mL sodium azide 1% solution (biocide); then, 0.1 mL of the mixture of Cellic® CTec2 and HTec2 (6.480:0.725) were added to this solution, and the final amount was adjusted to 40 g using deionized water. The final pH of the suspension was adjusted at 5.5. The mixture was reacted in a shaking incubator (150 rpm) at 50℃ for 72 hours. Sampling was done every 3 hours by putting 0.5 mL of the sample in a microtube, and the reaction was stopped by placing the microtube on a hot plate at 100℃ for 5 minutes. The mixture was vacuum filtered, and the glucose concentration was analyzed.
2.4 Chemical composition analysis
The acid-insoluble lignin content of raw materials was measured according to TAPPI T 222 om-02 test method. For acid-insoluble lignin evaluation, the filtrate of the primary and secondary acid-hydrolyzed samples showed five types of neutral sugars (glucose, arabinose, galactose, xylose, and mannose), which were then analyzed using Bio-LC (ICS-3000, Dionex, USA) system according to NREL/TP-510-42618 (determination of structural carbohydrates and lignin in biomass).
3. Results and Discussion
3.1 The effect of alkaline pretreatment
In this study, mild alkaline conditions for Eucalyptus pretreatment were examined (0.3-0.6 M Na2CO3 per gram of biomass and 0.25-0.5 M Ca(OH)2 per gram of biomass). As shown in (Fig. 1(a)), the experiment was done at different temperatures (160℃, 180℃, and 200℃) for 30 and 60 minutes. By increasing the alkaline concentration of sodium carbonate from 0.3 M to 0.6 M and calcium hydroxide from 0.25 M to 0.5 M, respectively, the amount of holocellulose that was extracted after the alkaline pretreatment increased from 65.78% to 73.80%, and delignification increased by 26.6% after the alkaline molar concentration was increased, which means that pretreatment effectively removed lignin from the samples and made the cellulose accessible for further enzymatic hydrolysis. The results show that these temperatures and alkaline molarities produced a sufficient alkalinity that is required for the cleavage of the acetyl groups, thus can produces high sugar content. According to Fig. 1, the glucose yield decreased by approximately 7% for every 10% increase in delignification rate. Increased delignification improves enzyme accessibility but leads to a decreased glucose yield. The effectiveness of alkaline pretreatment primarily relies on reducing the lignin content in the lignocellulosic biomass, which in turn increases the rate and yield of enzymatic hydrolysis of carbohydrates.15)
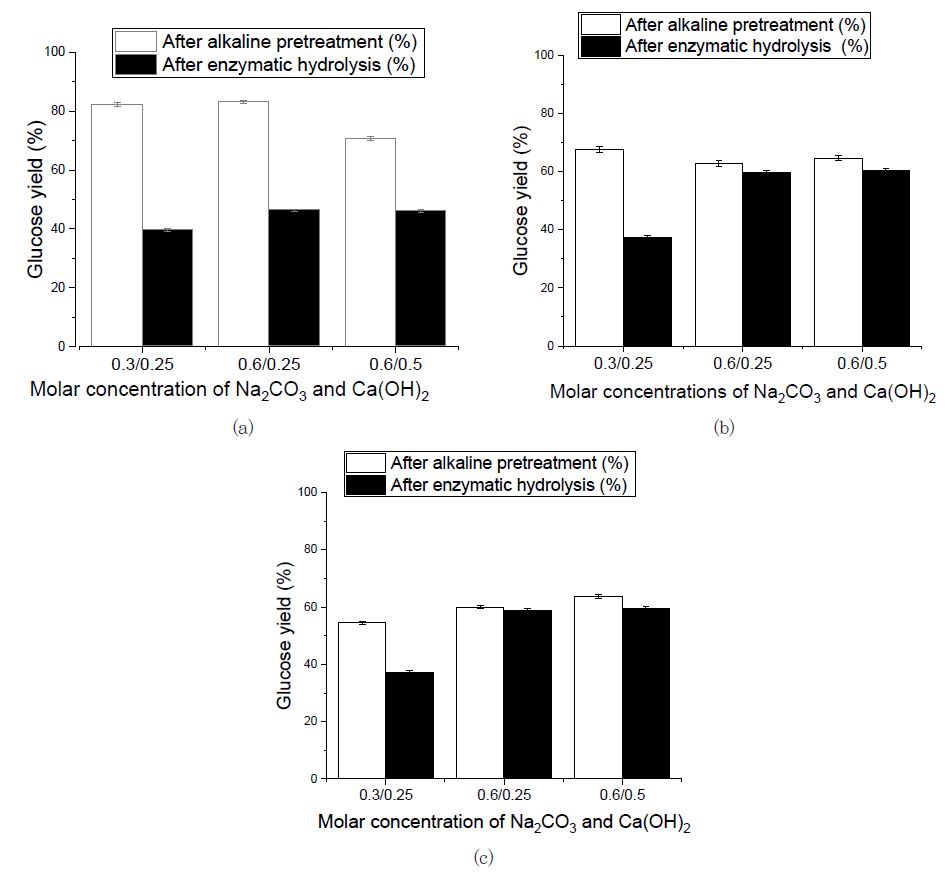
Fig. 1.
Comparison between glucose content after alkaline pretreatment and glucose yield after enzymatic hydrolysis of the pretreated Eucalyptus samples. The sugar content and yield are converted based on the initial sugar content before treatments (a) 160℃/60 min alkaline pretreatment, (b) 180℃/30 min alkaline pretreatment, and (c) 200℃/30 min alkaline pretreatment.
As the Fig. 1 shows, the highest glucose yield (73.86%) after alkaline pretreatment was produced in the combination of 0.6 M Na2CO3 with 0.5 M Ca(OH)2 per gram of biomass at 160℃ after 60 minutes of pretreatment. The rate of delignification improved with increasing reaction temperature but resulted in decreased glucose yield. According to Table 1, high temperature in associated with alkaline concentration produced the highest delignification rate (93.1%), but glucose yield was decreased because high temperature treatment can induce degradation of hemicelluloses with poorer thermal stability, leading to a significantly decreased number of free hydroxyl groups on the fiber surface, decreased surface polarity, increased surface roughness, and increased crystallinity.16) Cellulose with a high content of amorphous regions is more easily digested by enzymes. This may be the reason for the low glucose content at a high temperature.17)
Table 1.
Comparison of delignification and neutral sugar content after alkaline treatment in different conditions
| Temperature (℃) | Molar concentration of Na2CO3:Ca(OH)2 (M) | Reaction time (min) | Delignification (%) | Glucose* (%) | Acid insoluble lignin (%) |
|---|---|---|---|---|---|
| 160 | 0.6/0.50 | 60 | 79.4 | 70.8 | 07.1 |
| 0.6/0.25 | 50.7 | 83.2 | 17.2 | ||
| 0.3/0.25 | 52.8 | 82.4 | 16.4 | ||
| 180 | 0.6/0.50 | 30 | 87.4 | 64.2 | 03.8 |
| 0.6/0.25 | 79.1 | 59.8 | 07.3 | ||
| 0.3/0.25 | 78.1 | 74.6 | 07.6 | ||
| 200 | 0.6/0.50 | 92.6 | 50.0 | 02.6 | |
| 0.6/0.25 | 91.4 | 54.6 | 03.0 | ||
| 0.3/0.25 | 93.1 | 53.5 | 02.4 |
3.2 Effect of enzymatic hydrolysis
Alkaline pretreatment overcomes both chemical and physical barriers and makes polysaccharides easily available for enzyme digestion. Cellulose accessibility can be affected by many factors, such as cellulose crystallinity, contents and distributions of lignin and hemicelluloses, and available surface area.18,19) Enzymatic hydrolysis was performed to investigate the optimum condition for the pretreated Eucalyptus. The glucose concentration data from the enzymatic hydrolysis of the pretreated samples presented in Fig. 1 clearly shows that the highest glucose concentration (60.3%) after alkaline pretreatment was from the alkaline-pretreated Eucalyptus with the combination of 0.6 M Na2CO3 by 0.5 M Ca(OH)2/g of biomass for 180℃/30 min. This is due to the effects of the alkaline pretreatment, such as dissolving the wood structure, effectively removing the lignin and hemicellulose, and making the cellulose structure more accessible for enzymes to digest. Finally, the maximum sugar yield of 60.3% by enzyme hydrolysis (Fig. 1(b)) was achieved. The glucose content was also decreased during alkaline pretreatment as the delignification increased; however, glucose conversion by the enzymatic hydrolysis was increased by about 14% for each 10% increase in the delignification rate (Fig. 2).
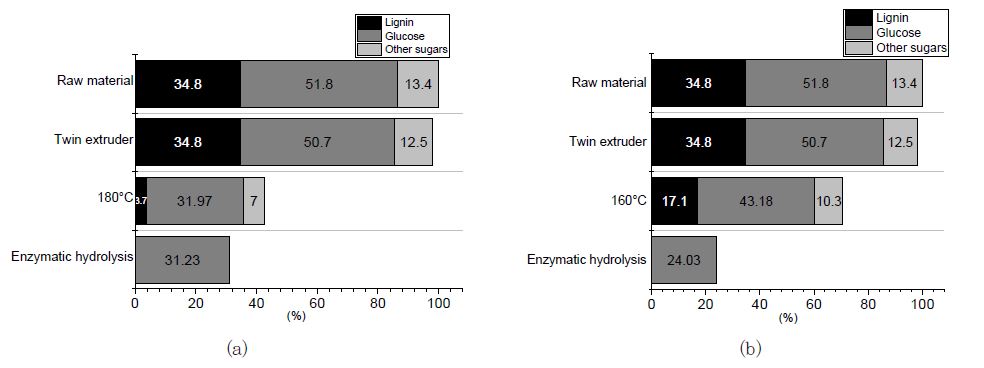
Fig. 2.
Changes in neutral sugar and lignin content from raw material to each process by enzymatic hydrolysis. (a) 0.6 M Na2CO3 in combination with 0.5 M Ca(OH)2 per g of biomass in 180℃ for 30 min; L:W, 20:1. (b) 0.6 M Na2CO3 in combination with 0.5 M Ca(OH)2 per g of biomass in 160℃ for 60 min; L:W, 20:1).
4. Conclusions
The researchers examined the effect of different pretreatment conditions to evaluate the effect of alkaline pretreatment on glucose yield. The variables were alkaline concentration and temperature. The effectiveness of alkaline concentration was higher than temperature, since increasing the temperature higher than 180℃ did not affect glucose yield. Furthermore, combining mild alkaline mixtures of Na2CO3 and calcium hydroxide perceptibly overcame the recalcitrance of Eucalyptus, removed 50-93% lignin, and produced 60.3% glucose yield. In alkaline pretreatment, glucose yield decreased by approximately 7% for every 10% increase in delignification rate. Enzymatic hydrolysis was performed under different pretreatment conditions, and its effectiveness was demonstrated by an increase of approximately 14% in glucose yield for every 10% increase in delignification. The optimum condition to reach the highest glucose yield in this experiment was pretreatment with the molar concentration of 0.6 M Na2CO3/0.5 M Ca(OH)2 per gram of biomass in 180℃ for 30 minutes.
The amount of peeling reaction and glucose loss in liquor after pretreatment to reach to a higher glucose yield need to be investigated in future researches. Also, further studies may be needed to investigate the effects of this kind of mild alkaline pretreatment on the degree of crystallinity of cellulose.




