1. Introduction
Surface sizing refers to the application of a sizing agent to the surface of paper and paper board, which can increase the liquid wetting resistance, improve surface properties and enhance certain physical properties of paper sheet.1,2) Because surface sizing increases the moisture content of the paper web, it requires extra drying energy in the after-drying section to reduce the moisture content of the web.3,4) To achieve this purpose efficiently many starch manufacturers have developed new products which can provide desirable properties to the final paper product at lower pickup weight and consequently allow the paper makers to use less amount of drying energy in the after-dryer section. Drying energy demand in the after-drying section can be reduced by using high solids sizing solutions or use more effective sizing agents.3,5,6) Lower energy requirement in the after-drying section could increase production speed especially for paper machines which are restricted by their afterdrying capacity. Moreover, less web breaks and higher paper machine efficiency can be achieved as a result of lower absorbed moisture at high solids surface sizing.4) Almost half of the total energy including both electricity and heat energies is used for drying of paper. And this indicates that most thorough investigation and researches are required if anyone wishes to save the energy in his or her papermaking processes.
Starch is the most frequently used agent in surface sizing. However, native starch has a high viscosity for surface sizing1) which is a limiting factor for solids content increment. Moreover, native starch solutions are not stable and have a tendency to retrograde, which is reassociation of dispersed amylose and linear segments of amylopectin during cooling of starch. The retrogradation results in the formation of three dimensional starch networks when the starch solution is held in the temperature range of 67°C to 89°C, and formation of a gel at temperatures below 55°C.1,7) The retrogradation limits the usage of starch in papermaking industry by increasing the viscosity of starch solution. Furthermore, it can restrict the storage time of cooked starch.8) This is the reason why paper industry usually uses modified starches to overcome the viscosity and retrogradation problems of native starch.
Various modified starches with a wide range of viscosities for desired application properties on the size press and proper starch penetration into base paper are available in the market. In addition modification of starch could retard, delay or eliminate retrogradation, which improve the stability and applicability in surface sizing process. Oxidized starches, cationic starches, starch ether, starch esters and grafted starches are the most frequently used modified starches for surface sizing. 1,7,9) Among these, graft copolymerization of starch by different monomers seems to be a new and promising method for modification of starch. In starch graft-copolymer, functional groups are attached to the main chain of starch along with their backbone monomers. Hence, graft copolymerization of starch improves the starch properties because not only functional groups but also various monomers with desirable characteristics can be added to starch molecules. Acrylic acid, acrylamide, methacrylic acid and other acrylic/vinyl monomers are usually used for grafting onto starch.10,11)
Pasting and rheological properties of starch solution might be controlled by the addition of desirable functional groups. Presence of carboxyl groups with negative charges in starch solution could prevent the retrogradation of starch molecules due to their repulsive forces. In addition carboxyl groups are more voluminous than the hydroxyl groups and tend to keep the amylose chains separated.12-14) Addition of same ionic compound to starch solution could reduce the peak and setback viscosities as well due to the repelling forces between starch and functional groups.
Starch graft copolymers which are new classes of modified starches have been introduced in paper making industry. Grafted starches with cationic charge are used as retention aids.7,12) Use of starch graft copolymer for moisture barrier coating and film-coated offset, recycled offset and LWC grades in pigmented size press formulation has been reported.15-17) These starch products are usually supplied in liquid form and ready to use.7) It has been shown that the production of high gloss and smoothness coated paper with high ink receptivity and porosity is possible with grafted starches with 1, 3-buradiene and styrene.18,19) Starch graft copolymers could be used as an alternative to the blending product of starch and synthetic binder. Applying starch graft copolymers as a sole coating binder, however, has not been commercially practiced.20)
Although use of starch graft copolymers in paper coating has been described in some literatures, there is limited information about how to use grafted starches in surface sizing of paper and board. Since starch copolymer provides benefits that we can obtain from natural polymer and synthetic monomers it could be used as an effective tool to control the retrogradation and rheological properties of sizing solutions. In this work a dextrin-graft poly(acrylic acid) was made and used as an additive for surface sizing of linerboard, and its effect on viscosity and pasting properties of sizing solution were investigated.
2. Materials and Methods
2.1 Materials
A commercial oxidized starch (C 3010) and a dextrin-graft-poly(acrylic acid) were prepared from Samyang Genex (Korea). The copolymer was a solution type and its solids content was 52%.
2.2 Methods
2.2.1 Viscosity measurement
A starch slurry at 16% concentration was cooked at a temperature of 95°C for 30 minutes. After cooling down to 65°C, starch paste was mixed with dextrin graft copolymer in conical tubes. The mixture was kept at constant temperature of 55°C. The viscosity measurement was carried out using a Brookfield viscometer (DV2T, Brookfield AMETEK, Massachusetts, USA). For better controlling of the temperature, an ultralow viscosity adaptor supported with a water jacket was used. All measurement of the viscosity was carried out at 50°C.
2.2.2 Fourier transform infrared (FTIR)
FT-IR spectra of the oxidized starch and dextrin-graft poly(acrylic acid) were obtained using a FT-IR spectrometer (Nicolet 6700, Thermo Electron Corporation, Madison, USA) in ATR mode with a range of 600-4,000 cm-1.
2.2.3 Pasting properties
Pasting properties of oxidized starch or oxidized starch-copolymer mixtures were determined using a Brabender Viscograph (Viscograph-E, GmbH & Co., Duisburg, Germany). The temperature and time condition were set as following manner. The slurry was heated from 30°C to 95°C at a rate of 3°C/ min and then held at 95°C for 30 min. It was subsequently cooled to 50°C at 3°C/min and then held at a temperature of 50°C for 180 min. Aqueous starch suspension at a solids level of 16% was used for pasting experiment with a Brabender Viscograph, and the viscosity in Brabender unit (BU) was recorded. The pasting temperature in °C, i.e. the temperature indicating an initial increase in viscosity, peak viscosity, i.e. the maximum viscosity during the heating, breakdown viscosity, i.e. peak viscosity minus final viscosity at 95°C, setback viscosity, i.e. the difference between the final viscosity at 50°C and minimum viscosity at 95°C, viscosity at the end of cooling period, and final viscosity were determined.
3. Results and Discussion
3.1 IR spectroscopy of starch copolymer
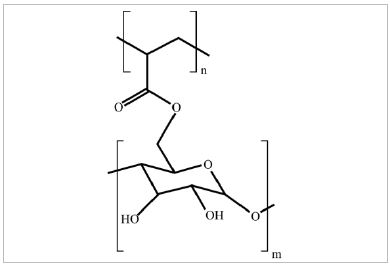
General structure of dextrin-acrylic acid copolymer is depicted in Fig. 1. As seen here, the acrylic acid polymer is covalently bonded on the hydroxyl group of starch molecules. The acrylic acid is polymerized after one monomer was reacted with a hydroxyl group via radical polymerization reaction. It is not clear, however, how many acrylic acid monomers were polymerized onto dextrin. Obviously, acrylic acid gave carboxylic functional groups to the dextrin-poly(acrylic acid) copolymer.
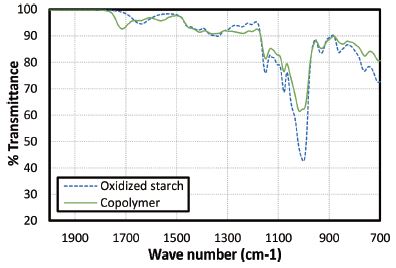
IR spectra of oxidized starch and dextrin-acrylic acid copolymer were obtained and shown in Fig. 2. Presence of functional groups like carbonyl and carboxyl groups may hinder retrogradation of starch sizing solutions.12,14) The FT-IR spectra of oxidized starch and dextrin-acrylic acid copolymer revealed that there was an extra absorbing peak at about 1700 cm-1 for copolymer, which showed the presence of carbonyl bonds in copolymer structure (Fig. 1). Obviously, the polyacrylic acid chain contained many carboxyl groups in the branch.
The carboxyl groups carrying negative charges obviously are very effective in hindering the reassociation of amylose molecules because their structural difference. Also the presence of long acrylic side chains would prevent reassociation of the other starch molecules. Moreover, the carbonyl and carboxyl groups are more voluminous than the hydroxyl groups and tend to keep the amylose chains separated, thus delaying retrogradation. 13,14)
3.2 Pasting properties of sizing solution
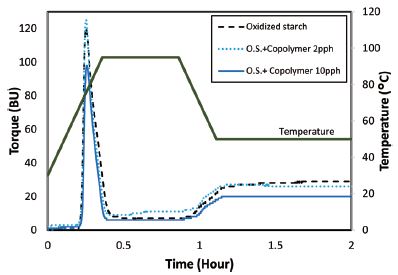
The pasting characteristics of oxidized starch without copolymer and with 2 parts and 10 parts of copolymer were determined with Brabender Viscoamylograph, and the results are depicted in Fig. 3. The general shape of the viscosity was all the same for starch and starch-poly(acrylic acid) copolymer mixture. Peak viscosity and retrogradation, however, were different depending on the starch samples.
Compare to oxidized starch solution, addition of copolymer resulted in significant reduction of peak, breakdown and setback viscosities. In the case of the addition of 2 parts copolymer, however, the peak viscosity increased. The results also revealed that as dosage of the copolymer increased the effects on peak and breakdown viscosities increased as well. And this could be attributed to the negative charges of both oxidized starch and copolymer. Chaisawang and Suphantharika studied the effect of the addition of negatively charged gums on pasting properties of oxidized starch, and reported that the repelling forces between the negatively charged groups on the anionic starch granules and the negative charges on the xanthan molecules could reduce the peak and setback viscosities.21) The results also showed addition of copolymer could reduce the viscosity after cooling period. In addition the final viscosity after 2 hours holding at 50°C decreased with the addition of copolymer and it was more pronounced for 10 parts of copolymer. Moreover Brabender viscosity for oxidized starch solution with copolymer during 1 hour holding period, showed more stable trend compare to oxidized starch solution without copolymer. Carboxylic groups of starch copolymer can hinder the reassociation of the starch molecules hence it can inhibit retrogradation of oxidized starch solution after cooling and during the holding period.12,22)
Table 1 shows the pasting properties of oxidized starch solution and starch-poly(acrylic acid) copolymer solutions. The addition of copolymer resulted in increased pasting temperature. The peak time (time to reach the peak viscosity) was unaffected by addition of copolymer. All of the viscosity data improved significantly when 10 parts of copolymer was used. Peak viscosity decreased from 120 BU for oxidized starch to 98 BU when 10 parts of copolymer was used. Most importantly, the final viscosity, which was 32 BU for the oxidized starch, was decreased to 27 BU and 21 BU when 2 parts and 10 parts of copolymer were used, respectively.
Table 1.
Pasting characteristics of starch solutions
3.3 Effect of copolymer dosage on viscosity of sizing solution
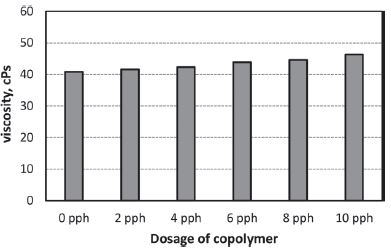
Viscosity of oxidized starch sizing solution with different dosage of copolymer was measured using a Brookfield viscometer and shown in Fig. 4. The viscosity increased as copolymer dosage increased. Since the addition of copolymer was made based on the weight of starch and the concentration of the copolymer was higher than starch solution, the increase of copolymer dosage resulted in higher solids content and consequently higher viscosity. Table 2 shows the solids content of oxidized starch solution with different dosage of copolymer and their relevant viscosities. The increase in solids content by 1% resulted in 15% increase of the low shear viscosity. Lipponen and coworkers reported that the viscosity increased by 25% with 3% increase in solids content of oxidized starch solution at 60°C.4) Moreover, retrogradation of dispersed starch during cooling and holding of the sizing solution at 50°C led to viscosity increase.
3.4 Retrogradation of sizing solution
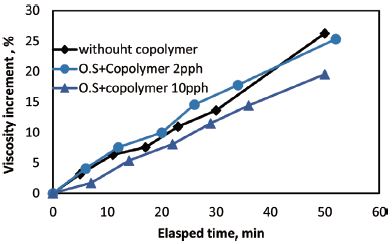
Starch retrogradation is usually accompanied by an increased viscosity due to the tendency of amylose polymers to orient in a parallel fashion with hydrogen bonds as starch solution cools. Fig. 5 reveals the viscosity of starch solution as a function of time after cooling to 50°C. As time went by, the viscosity of oxidized starch solution increased gradually as a result of retrogradation. Effect of retrogradation on viscosity of oxidized starch with 2 parts and 10 parts of copolymer were also depicted in Fig. 5 and the result showed that the viscosity of the sizing solutions with copolymer also increased with the increase of time indicating that more retrogradation proceeded at 50°C.
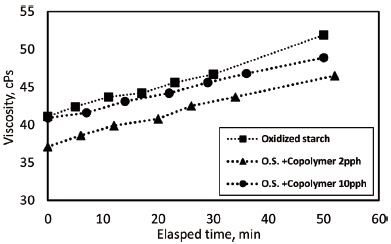
Fig. 5.
Viscosity change of the starch solutions at 50°C as a function of time for oxidized starch slurry, oxidized starch with 2 parts of copolymer, and oxidized starch with 10 parts of copolymer.
Because the initial viscosities of oxidized starch solution and oxidized starch solutions with 2 parts and 10 parts of copolymers were different, it was not possible to compare the final viscosity directly for the three different starch solutions. Hence, the percent increase in viscosity of three solutions was depicted in Fig. 6 to compare the effect of copolymer dosage on the viscosity of starch solution. Viscosity increment as a function of time for different sizing solutions are shown in Fig. 6. The result showed that addition of 10 parts of copolymer reduced the retrogradation of sizing solution and consequent viscosity increment after 50 min. But low addition level of copolymer did not show any significant influence on viscosity increment. Carboxylic groups of starch copolymer would hinder the reassociation of starch molecules by hydrogen bonding, leading to a lower tendency for retrogradation.12,22) Compare to oxidized starch sizing solution, Addition of 2 parts of copolymer did not have significant effect on viscosity at initial viscosity measurements. However, between 10 to 40 minutes the viscosity of oxidized starch with 2 parts of copolymer increased more. After 45 minutes the viscosity increment was lower for oxidized starch with 2 parts of copolymer.
As the amount of copolymer increased the viscosity increment reduced. When the viscosity increment for oxidized starch with 10 parts of copolymer with that for oxidized starch solution was compared, it is clearly shown that the increment of viscosity for sizing solution with 10 parts of copolymer is lower. Moreover, viscosity increment due to retrogradation for oxidized starch solution with 2 and 10 parts of copolymer showed a constant rate. However, for oxidized starch without copolymer the viscosity increment after 30 min was more pronounced than those of both oxidized starch solutions with copolymer.
4. Conclusions
Drying energy saving by reducing the steam consumption in the after-dryer section becomes more important because surface sizing gets more widely used for many grades of paper including linerboards.
Drying energy demand in the after-dryer for reducing the moisture of the sized paper is an important economic factor for paper manufacturing which could determine the production capacity. Using an efficient sizing solution with higher solids content and decreasing the pickup weight can reduced the after drying energy. In addition, pasting and rheological properties of starch sizing solution have a key role in penetration of sizing solution into paper. A dextrin poly(acrylic acid) copolymer was prepared and used as an additive for oxidized starch surface sizing agent. The effect of copolymer on rheological and pasting properties of starch sizing solution was investigated.
The results showed that addition of dextrin poly(acrylic acid) copolymer has a beneficial effect on the rheological and pasting properties of starch sizing solution. Starch graft copolymer could reduce the reassociation of starch molecules and hinder retrogradation after cooling and during the holding period.