1. Introduction
Cellulose fibers have several distinctive advantages over other materials: abundant in nature, biodegradable, non-toxic, eco-friendly, sustainable, easy to fabricate, hydrophilic, and cost-effective. Cellulose fibers have been used to produce paper products, hygiene paper (e.g., tissue and towel), disposable absorbent products (e.g., diapers and sanitary pads), and as a raw material to produce a novel material such as nano-cellulose. The sources of the raw material are diverse. At present, wood has been most commonly used as the raw material. A recycled pulp from used products has been also used.
Recently, several significant changes have forced the pulp and paper industry to rethink their approach. First, due to digital technology, there has been a steady decline in the need for pulp and paper products. The digital industry has continuously replaced printed products such as books, newspapers, and magazines. Second, there has been a trend initiated by developed countries to limit the use of wood as the raw material for the sake of environmental protection. This forces the industry to find a more efficient use of wood pulp as well as finding alternative, non-wood sources. Third, as an individual becomes wealthier and more conscious of health-care, the quality of a product becomes more important than the cost. Thus, the pulp and paper industry has been under pressure to produce a superior product at a competitive cost. These changes have forced the pulp & industry to make the paradigm shift from being a low-cost producer to being a high quality producer.
The objective of this paper is to review the current status of the pulp and paper industry and to suggest the ways of engineering cellulose fibers from the raw materials to produce high-value added products.
2. Background
2.1 Raw materials for cellulose fibers
The major compositions of the raw materials are lignin, cellulose and hemicellulose. The raw material for producing a cellulose fiber, as known as a pulp, can either be a wood or non-wood. Woods have been the dominant raw materials for producing pulp. Recently, however, non-wood materials have been increasingly examined as alternatives to wood materials to protect forests and the environment.1-3) In particular, the biomass (i.e., solid waste) from oil palm trees and sugar cane bagasse has been used as the raw material.4-8)Table 1 shows the source of the raw materials for producing cellulose fibers.
2.2 Pulping
Pulping is a process of removing lignin for the raw materials. To date, wood has been predominantly used as the raw material. For chemical pulping, Kraft pulping and Sulfite pulping have been mainly used though the use of the latter has steadily declined due to environmental issues.1)
In pulping, the term ‘yield’, is defined as the amount of the pulp obtained from the raw material, and it is expressed in %. As more non-cellulose components are removed from the raw material, the yield would decrease and would mean a lesser amount of pulp, but higher cellulose content would be obtained. With a higher content of the lignin, more vigorous pulping conditions (i.e., high concentration of pulping liquor, temperature, cooking time, etc.) would be required.
3. The structure of cellulose fibers: Multilamellar model
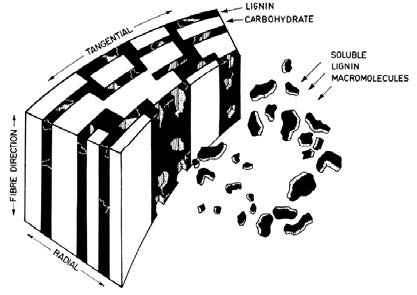
Bailey first published photomicrographs which showed that the cell wall of wood had a lamellar structure in 1983.9) Since then, the multilamellar model has been generally accepted as the pore geometry of the cell wall proposed by Stone and Scallan.10) Goring has speculated that the components (i.e., cellulose, hemicellulose, and lignin) are distributed in a multilamellar fashion as shown in Fig. 1.11)
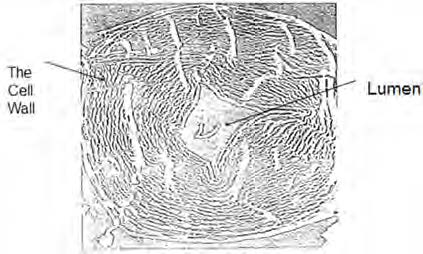
Then, as the lignin and the hemicellulose are progressively removed by a pulping process, pores are created where such components were previously located, as shown in Fig. 2. Thus, a pulp becomes highly porous after a complete removal of the lignin and its porosity should depend on the lignin and the hemicellulose contents. A never-been dried pulp after pulping becomes highly porous. The pores created in the cell wall are referred to as the intra-pores (or intra-capillaries). 10,11) The water retention value has been used to determine the pore volume in the cell wall. Fig. 3. shows the cross-section of the cell wall of a spruce pulp fiber (as illustrated). It shows many pores in the cell wall, as well as the lumen.12,13)
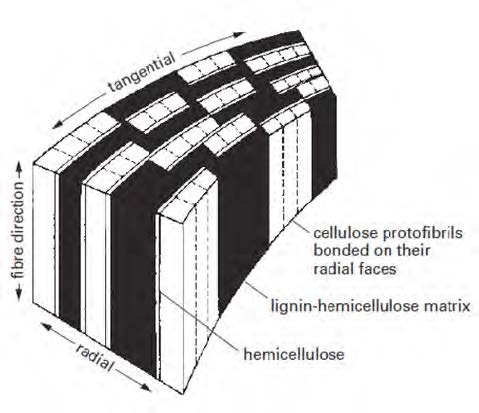
Fig. 1.
Arrangements of cellulose, hemicellulose, and lignin in the wood cell wall prior to pulping.11)
3.1 The multilamellar model for the pores in the cell wall
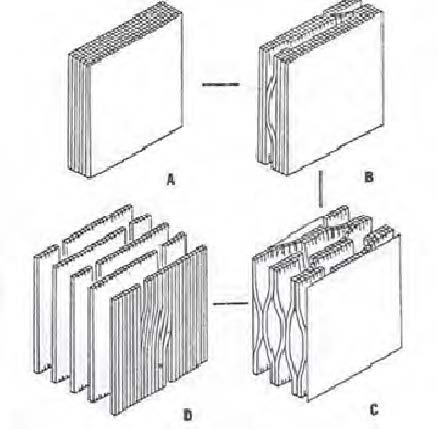
Stone and Scallan have proposed a laminar model of the pore in the cell wall.14-17) As discussed earlier, the cell wall of a never-been dried pulp is highly porous, consisting of many (up to several hundred) lamellae. They are gradually collapsed during drying, resulting in the loss of the pores between them. Thus, the dried cell walls essentially become non-porous.
The collapsed cell wall may be reopened by subsequent reswelling in water. Fig. 4 shows the model of the swelling of the cell wall suggested by Stone and Scallan. It shows that the fully swollen cell wall should have a multilamellar pattern. It is important to realize that they would never be fully re-opened, resulting in the permanent collapse of some pores after drying. Namely, drying and reswelling of a pulp should be an irreversible process.
3.2 Pore structure
3.2.1 The surface tension theory (Campbell forces)
Campbell suggested that the surface tension force of water should be primarily responsible for the structural collapse of cellulose fibers.18,19) The surface tension force is referred to as the Campbell force (or Campbell effect). The Campbell coefficient can be explained using the Laplace equation (Eq. 1).20)
where, ΔPc = the capillary pressure,
τ = the surface tension of liquid (i.e., water),
θ = the contact angle, and
w = the pore width.
Eq. 1 shows that △Pc should be proportional to the surface tension (τ) of a liquid. So, to reduce the surface tension of a drying liquid, several approaches have been taken.
A hydrocarbon liquid has much lower surface tension than water. It is, however, not miscible with water. Therefore, a direct replacement of water by a hydrocarbon solvent may not be possible. Merchant has developed the solvent exchange drying method (SED). For this method, a solvent which is miscible with both water and a hydrocarbon solvent is used as an intermediate. A polar solvent such as methanol or ethanol was used.21)
Freeze-drying from water has also tried to eliminate the surface tension force completely.15,21) As another way of eliminating the surface tension force completely, critical point drying has been also tried with CO₂ as the final replacement solvent. 22,23) Since liquid CO₂ is not miscible with water, however, an intermediate solvent which is miscible with the two has been employed. As is the case with Merchant’s solvent-exchange method, methanol or ethanol is used to this end.
The surface areas of the solvent-exchange dried pulps have been determined from the nitrogen adsorption experiments using the Brunauer-Emmett-Teller (BET) method.24,25)
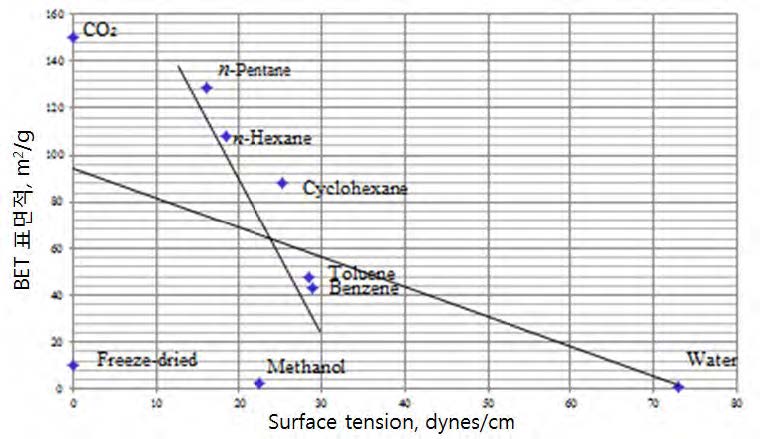
The results are summarized in Table 2 with the surface tension value of the final replacement solvent.
Table 2.
The BET surface area with the surface tension value of the final replacement solvent21)
Fig. 5 is the plot of the BET surface area vs. the surface tension value of the final solvent from Table 2. Fig. 5 shows several interesting points. First, the surface areas of the samples of the freeze-dried, from methanol, and from water, are very low. Second, the surface area of the critical point dried with CO₂ is largest, at about 150 m²/g. Third, the surface areas dried from a series of hydrocarbon liquids have a very high correlation with the surface tension values.
From these observations the following conclusions can be made regarding the solvent exchange methods.
1. The polarity of the final involved liquid is far more important than its surface tension value in determining a degree of the pore collapse.
2. The surface tension theory may be applicable to non-polar liquids which have different surface tension values. In this case, there seems to be high correlation between the BET area and the surface tension value as shown in Fig. 5.
3. The reasons that the critical point drying with CO₂ are two-fold. One is that liquid CO₂ is non-polar and it is dried at zerosurface tension value. So, it eliminates both the polarity and the Campbell forces simultaneously.
3.2.2 Solute exclusion technique
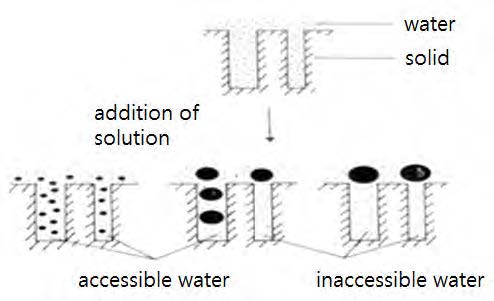
Stone and Scallan have developed the “solute-exclusion technique” to characterize the pores in the cell wall directly without drying.10,16,17,26) The principle of the solute-exclusion technique is shown in Fig. 6. When a never-dried pulp is put in a polymer solution, polymer molecules will penetrate into the pores, depending on their molecular sizes. Then, some of the water in the pores will be replaced by such polymer molecules due to penetration. In this case, the water replaced is called the accessible water to the polymer molecules; otherwise, it is the inaccessible water to the polymer molecules.
The accessible (or replaceable) water will dilute the polymer solution. Therefore, by measuring concentration changes in an initially known polymer solution, using a series of homologous polymers of different sizes, the amount of the accessible (or inaccessible) water can be determined. As polymers, a series of dextran polymers was most frequently used for this technique.
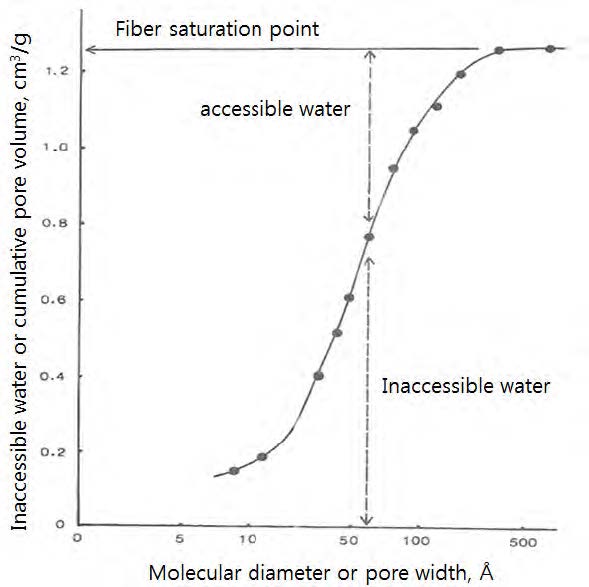
Fig. 7 shows a plot of the inaccessible water vs. molecular diameter (pore width) for a pulp fiber, obtained by the solute exclusion technique. Notice in the Fig. 7 that molecular diameter is plotted as log-scale.27)
In the Fig. 7, the inaccessible water corresponds to the cumulative pore volume, while molecular diameter is equivalent to the pore width. Further, the fiber saturation point (FSP) is the total inaccessible water by the biggest polymer molecules. This FSP is empirically highly-correlated with the WRV (water retention volume of water) determined by the standard centrifugation method.28) This indicates that the WRV can be used as a measure of the total pore volume in the cell walls in the swollen state.
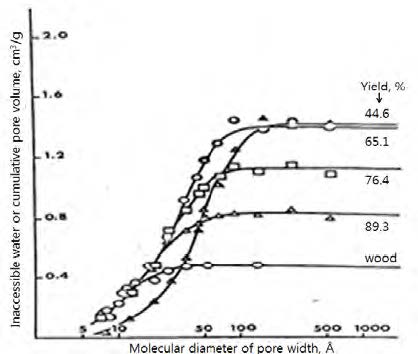
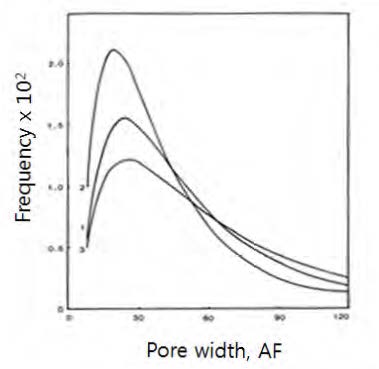
Fig. 8 shows the cumulative pore volume (Vp) of various wood pulps having different yields. It shows Vp increases as the yield decreases (i.e. , more lignin is removed) as expected. It shows also that the cell wall pores are quite broad in sizes ranging from about 10 Å to a several hundred Å.
3.2.3 Log-normal distribution model
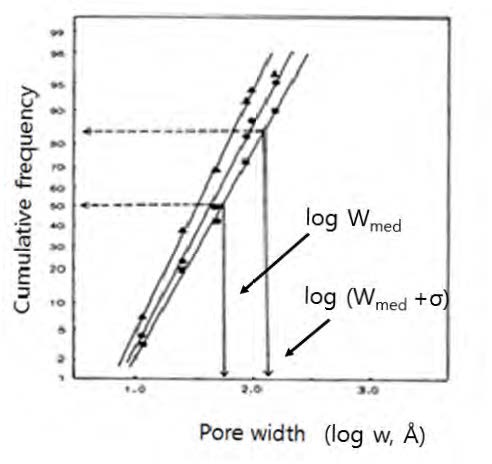
It has been confirmed that the cumulative pore size distribution curves as shown in Fig. 8 should follow a log-normal frequency distribution mode by demonstrating the linearity between the two on a log-probability paper.29-31)Fig. 9 are such plots of the curves in Fig. 8. In the plot, Wmed is the pore width corresponding to the 50% of the cumulative pore volume, and σ is the standard deviation of the pore width where 84% of the cumulative pore size distribution is at the pore size of Wmed+σ.32) Here, a cumulative frequency is the cumulative percentage of the pore volume normalized by the total pore volume, i.e., fiber saturation point.
Log-normal frequency distribution is expressed mathematically as follows.32)
Eq. 2 shows that the two parameters of the median pore width (Wmed) and the standard deviation (σ) are necessary to construct the frequency distribution curves as a function of pore width (w).
Fig. 10 shows the pore width frequency distribution constructed from Fig. 9. Eq. 2 indicates that the pore size distribution should be proportional to the pore width. Meanwhile, the actual pore size distribution follows a log-normal distribution, as shown in Fig. 11.30)
Allan and Ko have reasoned that a log-normal frequency distribution of the cell wall pores should be the combined effect of the forces involved during drying: 1) long-range van der Waals attraction forces to pull the laminates of the cell wall together, 2) electrical repulsion due to the overlap of the electrical double layers to push the laminates, and 3) the surface tension forces. Their theory is analogous to the DLVO (Deragin-Landau-Verway-Overbeek) theory of the stability of colloidal.20)
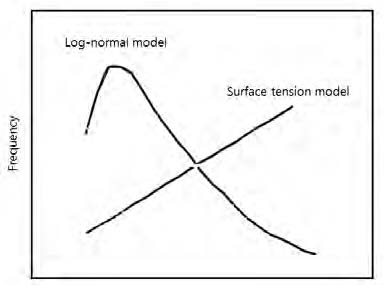
Fig. 11.
Comparison of the pore size distribution between the surface tension model and log-normal model.30)
4. Engineering cellulose fibers for high value-added products
In this section, bulk fibers, carboxyl methyl cellulose (CMC) attachment technology, inclusion cellulose, nano and micro-cellulose, and drying of nano-cellulose will be discussed.
4.1 Bulk fibers
Bulk is defined as the volume per unit mass and has the unit of cm³/g. Fiber bulk is inversely related to the coarseness which is the linear mass, expressed as miligrams per 100 m.12) Thus, a higher coarseness means the heavier fiber. Bulk determines the strength, softness and absorbency. A high bulk may not be desirable for strength, but it is highly desirable for softness and absorbency of hygiene paper such as paper towels and disposable absorbent products like baby diapers and sanitary pads.
A high yield pulp is bulkier due to the lignin within. As the lignin is progressively removed, a pulp loses the bulk. Thus, a high yield pulp such as TMP (thermo-mechanical pulp) and CTMP (chemical thermo-mechanical pulp) has a much higher bulk than chemical pulp such as kraft or sulfate pulp.
For a good absorbency, a product requires a high absorbency capacity at a fast speed. To have a fast speed, cellulose fibers should have good wettability. A high yield pulp has, however, poor wettability due to the presence of lignin.33) Accordingly, a highly bleached chemical pulp such as Southern softwood kraft pulp (SSK) has been used for highly absorbent products.
4.2 Chemically cross-linked fibers
The morphology of chemical pulp such as Softwood kraft pulp has a 2-dimensional flat morphology and has many internal pores which are fragile under the load. The fragility increases with the WRV.
So, there have been efforts to reduce the fragility of a pulp by permanently interlocking the pores by use of cross-linking chemicals. After the internal pores are cross-linked, the fiber will become bulky and stiff. For example, the Buckeye Cellulose Corp. developed the curly fibers by chemical cross-linking of a Foley pulp (Southern softwood kraft).34-38)
Chemical cross-linking in the cell walls will prevent the cell wall from swelling with water, resulting in a decrease of the WRV. Thus, an intensity of the chemical cross-linking may be estimated from the decrease in the WRV of the crosslinked fibers from that of the uncross-linked fibers by 20 - 60% reduction.34)
One of the main issues in this chemical process is that chemical crosslinking occurs not only within the individual fibers, but also between the fibers to form the entangled forms. So, the chemical crosslinked fibers must be mechanically defibrated into individualized fibers known as “fluff”. For defibration, a Waring blender or rotating disk refiner might be employed. A fluffing device may be also used such as one described elsewhere.34-38)
After chemical treatment, flash drying has been employed. It is observed that both dried and wetfibers are twisted and curly. The average dry fiber twist count is in a range of 2.5 - 3.0 counts per millimeter whereas the average wet fiber twist count is at least approximately 1.5 - 2.0 counts per mm, less than the dry fiber twist count by 1.0 counts per mm.34)
After flash drying, additional heat-treatment is required to cure the cross-linking, i.e., to react with the cellulosic fibers. The rate and degree of cross-linking depends upon dryness of the fiber, temperature, pH, amount and type of catalyst and cross-linking agent and the method utilized for heating and/or drying the fibers while cross-linking is performed. Thus, the chemically crosslinked process invented by Buckeye Cellulose is a complicated process, dealing with cross-linking agents. Nonetheless, the fibers have been successfully used to produce disposable absorbent products such as baby diapers.
4.3 Twisted fibers by flash drying
Ko, et al. have developed a process of producing twisted fibers by the flash drying method without use of a chemical cross-linking agent.39,40) Flash drying is a well-known method for drying materials such as wood pulps, gypsum, starches, and inorganic fillers. It has been reported that flash drying should be more economical due to a lower capital cost and less manpower requirements than conventional drum drying.41)
In flash drying, a never-dried pulp is exposed to a very hot drying air (or gas) environment for a very short time (up to less than a few seconds). These flash drying conditions are likely to put the fibers in a non-equilibrium state, thus making them shrink non-uniformly. Consequently, the flash dried fibers are likely to be twisted or coiled. In addition, such flash drying conditions may provide little time for the internal pores to be collapsed. 39,40)
As an illustration, a never-been dried Kraft pulp has been flash-dried using a pilot flash dryer at the Barr-Rosin, Montreal, Canada, at the temperature of 232 - 327℃ (or 450 - 620℉) for about one second. This temperature is much higher and the drying time is much shorter than that of conventional thermal drying, such as Yankee drying. Fig. 12 shows the twisted, curly fiber produced by flash drying method by Ko et al.39)
Table 3 compares the chemical cross-linking method with the flash drying method. The WRV of a Foley fluff pulp is reported as about 1.0 g-water per g-pulp whereas its WRV after cross-linking reduces to around 0.2 - 0.4 g-water per g-pulp.34)
4.4 CMC attachment technology by the STFI (Now Innventia), Stockholm, Sweden
Cellulose fibers are negatively charged. The total charge is referred to as the bulk charge which includes the charge inside and on the surface. The latter charge is called the surface charge. It is the surface charge which is important in the wet-end chemistry and papermaking process. It influences strength (both dry and wet), retention of cationic additives such as dry strength resins, or debonders.
Unfortunately, however, the surface charge density of a pulp is too low for practical applications, so the effectiveness of such cationic additives is limited by the available surface charge density of the fiber. To compensate for this, for example, in paper towels, CMC (carboxyl methyl cellulose) is added to retain the wet strength agent which is cationic (Kymene–Hercules). To this end, an aqueous CMC solution and a cationic solution are added to a slurry of pulp.
A conventional way of increasing the negative charge density of a pulp has been by the means of chemical modifications such as carboxylation, methylation, and ethylation. All these chemical modifications are, however, costly and complex. It also can easily destroy the original structure. More importantly, however, such chemical modifications increase the total charge density (i.e., bulk charge density) with a marginal increase in the surface charge density.
In a way to increase the surface charge density of cellulose fibers, Tom Lindstrom et al., have developed a method of attaching CMC (carboxyl methyl cellulose) to the surface of cellulose fibers without chemical reactions. They have identified the key variables that determine the retention efficiency of CMC to the fiber surfaces: 1) molecular weight (MW) and 2) the degree of the substitution, 3) temperature, 4) electrolytes and concentrations. 42-45)
4.4.1 CMC specification
As discussed in the solute exclusion technique, a CMC of a high molecular weight (MW) should be more difficult to penetrate inside the fibers (i.e., the cell wall pores). So, a high MW CMC is preferable to retain the CMC on the surface to increase the surface charge density. It is reported that if the CMC has a MW higher than 1 million, it would not be able to penetrate into the cell wall pores resulting in a very little increase in the charge density.35) In addition, CMC should have a low degree of the substitution (DS = 0.52). For example, STFI has shown that, with a higher MW CMC (Finnfix WRH, MW = 1,000,000), the CMC retention was 96%, but with a lower MW CMC (Cekol FF2, MW = 280,000), the retention was only 59%.
4.4.2 Effect of temperature
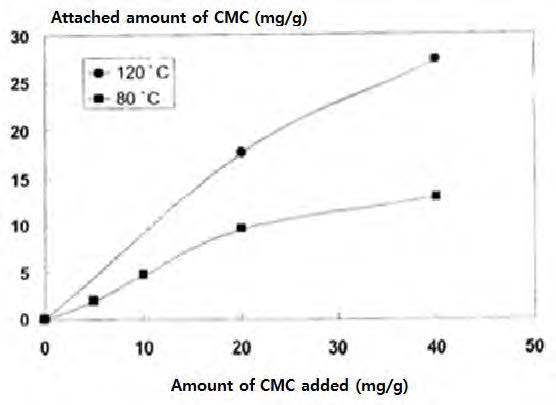
It is found that a higher temperature is preferable for the retention of CMC by a pulp.35)Fig. 13 shows the effect of temperature on the attachment of CMC to a pulp. It shows clearly that the retention of the CMC is much higher at 120℃ than at 80℃.
4.4.3 Surface charge density of CMC-attached cellulose fibers
Table 4 shows a comparison of charge density of the CMC-attached pulp with that of the untreated pulp.42) It shows that the untreated pulp has 3 unit (surface charge density) among the 40 total charge, occupying only 7.5%. Meanwhile, the CMC attached pulp has the surface charge of 29 among the total 67 charge, occupying 41.4%.
Table 4.
Internal and surface charge density of CMC attached pulp
| CMC attached wood pulp * Furnish: Never-dried unbeaten ECF-bleached softwood kraft | |||
|---|---|---|---|
| Total | Surface | % of Surface charge | |
| Reference (No treatment) | 40 | 3 | 7.5 |
| CMC attached | 67 | 29 | 41.4 |
Source : T. Lindström, STFI, 2002.42)
4.4.4 Water retention value
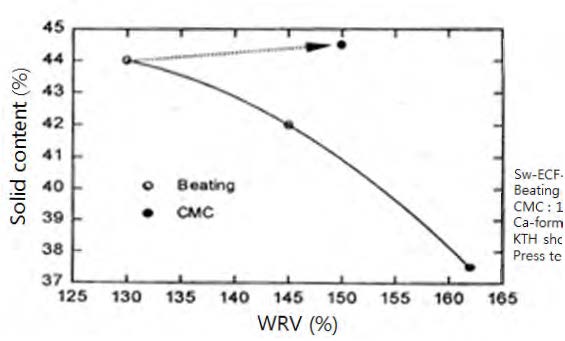
Beating (or refining) a pulp is commonly employed to increase the strength of a product made thereof. Beating increases the WRV (water retention value). The WRV has been determined using the method of SCAN-C 102XE, 15 min. at 3000 g.42)Fig. 14 shows the solid content vs. WRV of a beaten pulp and of CMC-attached pulp. In the Fig. 14, the WRV was determined by dividing the water content by the solid content. For example, if the solid content is 44%, then the water content would be 56%. So, the WRV would be 1.27 (56%/44%).
Fig. 14 shows that the WRV increases (solid content decreases) rapidly as the intensity of beating increases. Meanwhile, for the CMC attached pulp, the solid content remains constant though the WRV increases. This indicates the increase in the WRV primarily comes from the water held by the attached CMC, not from the swollen cell wall pores. Thus, the CMC would not contribute to swelling the cellulose fibers unlike the beating. It is suggested that an increase in WRV of the CMC-attached wood pulp will not negatively influence the drainage rate, dewatering and drying.
4.4.5 Wet strength enhancement by CMC attachment
A cellulose fiber has a very low surface charge density to interact with wet strength resin which is positively charged. For boxes or paper towels, PAE (Kymene) is commonly used with CMC.
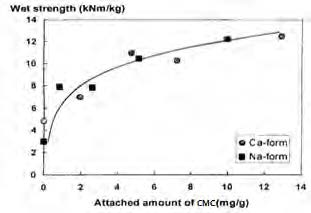
Fig. 15 shows that wet strength of the handset constantly increases as the attached amount of CMC increases. On the other hand, the wet strength of the unattached pulp (control) is relatively low. This suggests the control pulp would require external CMC to increase the wet strength. (PAE–Kymene) which had been practiced in paper industry. The CMC attached pulp eliminates such necessity of adding CMC to retain PAE (Kymene). For CMC attachment, both NaCl and CaCl₂ electrolytes have been used. The figure shows that the effect of the electrolyte on the wet strength is negligible.
4.4.6 Stress-strain curves of CMC attached fibers
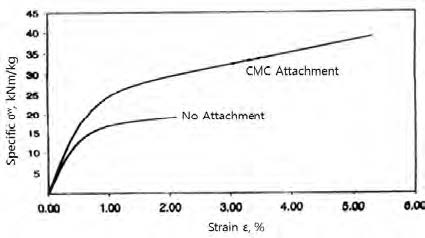
Fig. 16 shows the stress-strain curves of the handsheets made of the control (no CMC attachment) and those made of the CMC-attached pulp. It shows that the CMC-attachment provides much higher strength and the strain than the control (no attachment).
Beating is commonly used to increase the strength of a paper. Table 5 compares the effect on the stress-strain curves between beating and CMC attachment. It shows that the CMC-attached pulp may minimize beating process in increasing the strength of the product.
Table 5.
Comparison between beating and CMC attachment42)
| Tensile | Modules | Strain to failure | Bonding strength | |
|---|---|---|---|---|
| Beating | + + | + + | + | + |
| CMC | + + | (+) | + + | + + |
4.5 Inclusion cellulose
The internal pores in the lumen and the cell wall of cellulose fibers have been utilized as the storage for fluids and active ingredients.46-49) Inexpensive metal such as aluminum oxide (Al2O3) has been loaded into the lumen to increase the basis weight (BW) of a sheet to save more expensive cellulose fibers.46) Active ingredient such as perfume, herbicide, pesticide, pharmaceuticals, and fertilizer were put into the cell wall pores. Then they were entrapped by closing the pores during drying. The active ingredient entrapped cellulose is referred to as “inclusion cellulose”. Once the ingredient entrapped, it may be difficult to be released from the inclusion cellulose. The closed pores may be opened by moisture to release the ingredient. So, inclusion cellulose is a way of producing as low-release system.
Since the swollen cell wall has the extremely large surface area of about 1000 m²/g, it may be used as the carrier for a catalyst to make ‘catalyst-inclusion cellulose’. Table 6 lists the materials loaded within cellulose for the inclusion cellulose.
Table 6.
Materials loaded within cellulose (inclusion cellulose)42)
| Materials embedded within cellulose |
|---|
| • Inorganic pigments, white and colored |
| • Drugs, exemplified by aspirin |
| • Plant growth simulants |
| • Herbicides |
| • Fertilizers |
| • Simple sugars |
| • Glucose oligomers |
4.6 Nano-Cellulose
Recently, nano-cellulose has attracted much interest in many industrial applications. Though nano-size is 1000 times smaller than micro-size, they are often used interchangeably to indicate the size is very small. Sometimes, micro- is used to indicate the aggregates of nano-size objects.45)
Nanocellulose (NC) may be classified into:
1) Micro/Nano-crystalline cellulose (MCC/NCC)
2) Micro/Nano-fibrillated cellulose (MFC/MFC)
Table 7 lists some applications of nano-cellulose.
Table 7.
Applications of nanocellulose
4.7 Microcrystalline Cellulose (MCC)
As shown in Fig. 1, cellulose fibers are comprised of the microfibrils which are the aggregates of the elementary fibrils. The elementary fibrils are comprised of the crystalline region and the non-crystalline region.45,46)
In early 1960s, Battista invented on producing microcrystalline celluloses (MCC) by removing non-crystalline portions from the microfibrils in cellulose fibers. To this end, acid hydrolysis was employed to weaken the links between the crystalline portion and the non-crystalline portion. To liberate the crystalline portion from the acid-hydrolyzed pulp, a high shear force was applied.47-52) To this end, a Gauline homogenizer or Microfluidizer may be used.47,53) AvicelⓇ from FMC (Food Manufacturing Company) is the representative brand name of the commercial microcrystalline cellulose.
It is realized that chemical treatment should be required to break the links between the crystalline portion and the non-crystalline portion. Mechanical treatment alone without chemical treatment will not work. For chemical treatment, acid such as hydrochloric acid or sulfuric acid may be employed.54)
As acid treatment on the pulp proceeds, the pulp will be depolymerized until its degree of polymerization (DP) does not change any more, i.e., is leveled-off. Battista has defined that DP as level-off degree polymerization (LODP).48) A LODP is found to depend on the source of raw material and the acid treatment conditions. It is observed that a LODP is highly correlated with the length (or size) of the crystallites. The higher the LODP is, the longer the crystal would be.47,48)
It is, however, observed that the MCC in water forms aggregates and become very viscous even at a low consistency of 2 - 10%. To disintegrate the agglomerates into individual entities, ultrasonic treatment may be applied. The disintegrated crystallite aggregates has a wide distribution of sizes ranging from 40 to 250 or 300 microns and has a density of 1.2 to 1.57 g/cm³.47)
Battista’s work on MCC may be summarized as the following.
1. A mild acid-treatment step is necessary to eliminate the non-crystalline (amorphous) parts from, the elementary fibrils. Without the acid-treatment, it is impossible to produce nano-crystalline cellulose which has a high crystalline index. It is reported that the crystalline index of commercial MCC is about 78%.54)
2. The LODP can be used as a measure of the length of the crystalline cellulose (crystallites). It depends on the source of cellulose material. The higher the LODP that the cellulose has, the longer the crystallites would be.
3. An aqueous NCC sol is dried to produce nano-crystallites. Spray drying has been used to this end. The nano-crystallites are, however, agglomerated to form large particles, by losing the original fibrous morphology. Once dried, it would be extremely difficult, if not impossible, to disintegrate the agglomerates into individual entities having the fibrous morphology. This is why Battista refers to the NCCF as the aggregates of microcrystalline celluloses (MCC).
4.8 Nano/Micro-fibrillated cellulose (N/MFC)
Microfibrillated cellulose (MFC) was first coined by Turbak, et al., to describe a product as a gel type material by passing wood pulp through a Gaulin type milk homogenizer at high temperature and high pressure.53,55-59) It is to be noted that producing MFC does not involve an acid-treatment step to eliminate non-crystalline region in contrast with producing microcrystalline cellulose (MCC). Accordingly, MCC should have always a higher crystalline index than MFC. It is to be noted that producing both MCC and MFC should require a device which generates high searing forces such as a Gaulin homogenizer or microfluidizer.47,53)
5. Drying of nano-cellulose
5.1 Spray drying
Spray drying has been commonly used for an aqueous solution of hydrophilic particles. Battista et al., have employed the spray drying method for producing the aggregates of MCC. In spray drying, the size of the drops entering into the chamber plays an important role in determining the size of the product. Atomization is employed to produce smaller particles with minimal aggregation of the drops.49) In addition, to minimize the aggregation during drying, a surfactant, or a protective colloid might be added to an aqueous solution.47)
The surface area of Avicel (Commercial) has only 2 m²/g, indicating the MCC particles are agglomerated during drying to form the large particles. This value is not much different from the dried pulp from water or methanol as shown in Table 2.
5.2 Supercritical point drying
Recently there have been attempts to apply the supercrtical point drying method.60-63) Critical point drying has been commonly used to dehydrate biological tissue in the SEM (Scanning Electron Microscopy). The main purpose of critical point drying is to maintain the original morphology of the specimen by eliminating the surface tension of the fluid as discussed earlier as is the case with freeze drying. Freeze-drying has also been attempted to dry micro-fibrillated cellulose.64) Supercritical drying has also been widely used to dry inorganic gel systems to create aerogels.65)
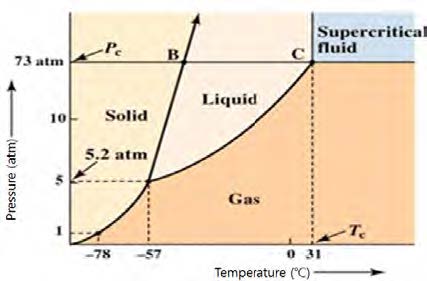
A liquid undergoes the three phases of the solid, the liquid, and the gas. At the critical conditions of the temperature and the pressure, however, a fluid has a state where the phase differences disappear. Such a state is referred to as the critical state, and the corresponding temperature and pressure is referred to as the critical temperature (Tc) and the critical pressure (Pc), respectively. A supercritical fluid (SCF) is any substance at a temperature and pressure above its critical point.
SCF is most widely used in extraction process such as the decaffeination of coffee and tea and extraction of flavors, fragrances, and essential oils from natural sources.66) Such a process of extracting one component from another using SPF as the extracting solvent is referred to as supercritical fluid extraction (SPE). Supercritical fluid extraction has been used in the food industry.62,66-67)
As a supercritical fluid, CO2 is most widely used since it is inexpensive, non-toxic and has favorable critical conditions of Tc=31℃ and Pc=74 Bars. Fig. 17 shows the phase diagram for carbon dioxide.
In the extraction process, solubility is an important factor. In order to increase the solubility of extractable ingredients in CO2, it is sometimes modified by co-solvents such as ethanol or methanol. 62,68)
Meanwhile, employing such co-solvents such as ethanol, methanol, or acetone can be detrimental for the supercritical point drying process. In other words, directly applying the supercritical extraction process for drying nano-crystalline cellulose fibers would work, but also affect adversely to produce NCCF as discussed in the following.
Brown et al., investigated the use of supercritical carbon dioxide (scCO2) for the removal of moisture from cylindrical pieces of carrot. They found that carrots dried in the supercritical fluid environment were seen to retain their shape much better than air-dried carrots which underwent shrinkage. Samples dried in ethanol-modified scCO2 possessed less dense structures and consequently displayed more favorable rehydrated textural properties than the air-dried equivalents.62)
A supercritical point drying with CO2 has been attempted for drying nano-cellulose. Since CO2 is, however, not miscible with water, direct replacement of the water in the specimen by liquid CO2 is impossible. Therefore, an intermediate fluid which is miscible with both water and liquid CO2 needs to be introduced. As an intermediate fluid, a polar solvent such as methanol, ethanol, and acetone is generally used62,64) as a SPE process which was discussed earlier.
A literature survey reveals that this important fact has been frequently overlooked for supercritical point drying.
For example, Peng, et al., tried to use a supercritical point drying method with CO2 in drying nano-fibrils.64,68) Ethanol was used as an intermediate fluid between water and liquid CO2. But, they abandoned this method since it is practically impossible to replace water with CO2 using ethanol as an intermediate fluid.
Peng’s work suggests that supercritical fluid such as ethanol or methanol used for the supercritical extraction process would not function well as an intermediate solvent for supercritical point drying. After giving up on using the supercritical point drying method, they reverted to the spray drying method as used by Battista, et al.
Thus, the use of polar liquid such as ethanol or methanol is a poor choice since it is practically impossible to replace water since the polar liquid and water are infinitely miscible. Besides, it should be noticed that such a polar solvent would not be much different from water in preventing the agglomeration of the particles as suggested in Table 2.
6. Conclusions
The pulp & paper industry has been considered as a mature industry whose market has been steadily declining. One characteristic of the mature industry has been to become a low-cost producer while less paying attention to developing a higher quality of the product. Consequently, there has not been much effort in engineering the raw material to produce a high-value added product or on developing the next generation of state-of-the art technology.
Recently, however, the pulp & paper industry has been forced to make the paradigm shift from the low-cost producer to the high-quality producer due to several reasons. First, the digital industry has penetrated into the pulp & paper industry by replacing paper products such as printed materials with online materials. Such replacement is expected to continue. Second, the woods have been the dominant raw material for the pulp & industry. The wood pulp producers, however, have faced governmental regulations on using the trees for environmental protection. This has forced the industry to develop non-wood pulp as an alternative.
Drying hydrophilic material such as cellulose fibers while maintaining its original structure still remains a challenging problem.