1. Introduction
In pulp bleaching, the use of enzyme has known as a promising approach for environmentally benign process. First of all, xylanase was first reported to be useful enzyme for pulp bleaching (1). Xylanase partially removes the xylan in pulp so that lignin can be more accessible to the reagent and easily removed in subsequent bleaching stages. Thereby, the use of xylanase in pulp bleaching was demonstrated to save up to 25% of bleaching chemicals required to reach a desired brightness (2, 3). Meanwhile, since the effect of xylanase on delignification of pulp is limited and indirect, the use of ligninolytic enzymes such as lignin peroxidase (LiP), manganese peroxidase (MnP) and laccase may be a more efficient way to delignify and bleach pulp. Of the ligninolytic enzymes, laccase alone is not able to degrade non-phenolic lignin substructures that account for about 90% of lignin because of its low redox potential. However, laccase has proved to be effective in oxidizing non-phenolic fractions of lignin model compound and reducing the pulp kappa number in the presence of redox mediators of low molecular weight (4). Thereafter, many studies have focused on the role of mediator in biodegradation of lignin and the search for new effective laccase mediators. The effective mediators for pulp bleaching reported so far are those that contain N-OH group such as 1-hydroxybenzotriazole (1-HBT), N-hydroxy acetanilide (NHA) and violuric acid etc. Though laccase mediator system seems to be very effective in an environmentally benign pulp bleaching, some problems such as the toxicity of mediators, the inactivation of enzyme, and high cost have to be solved for application of laccasemediator system in pulp mill.
To enhance the effect of enzymatic bleaching, two or more enzymes have been applied jointly in pulp bleaching. Niku-Paavola et al. (5) reported the effect of pulp bleaching using MnP and xylanase together. Bermeck et al. (6) also demonstrated the synergistic effect of the combination of MnP and xylanase. Viikari et al. (7) presented the optimal condition in pulp bleaching by using of xylanase and laccase-mediator system. Valls et al. (8) gave a report that xylanase pretreatment increases the accessibility of residual lignin and facilitates the removal of hexenuronic acid in subsequent laccase-mediator system. And Martin- Samperdro et al. (9) postulated that XLEP sequence results in an improvement of optical properties and a reduction of the kappa number.
In previous work (10), we found that N-hydroxy- 2-pyridone analogue (NHP) containing N-OH group is effective on the decrease of kappa number as a mediator of Trichophyton sp. LKY-7 laccase (TrL). In the present work, we evaluated the individual and the synergistic effects of xylanase and laccase-mediator system in enzymatic bleaching of hardwood kraft pulp (HwKP). And the effects of subsequent treatment with alkaline extraction or alkaline/hydrogen peroxide treatment were also examined.
2. Materials and Methods
2.1 Source of pulp, enzymes, and chemical
Kraft pulp for the bleaching experiment was manufactured with 20-year old hardwood (Queracus variabilis) in laboratory digester (cap. 4 liter) with the conditions shown in Table 1. After cooking, the pulp was washed with water and sodium acetate buffer (50 mM, pH 4.5). The initial brightness and kappa number of washed pulp were 32.2% ISO and 15.5, respectively.
Table 1
Kraft pulping conditions
| Pulping conditions | |
|---|---|
| Active alkali (%, as Na2O) | 18 |
| Sulfidity (%, as Na2O) | 25 |
| Wood to liquor ratio | 4:1 |
| Max. temp. (℃) | 170 |
| Time to max. temp.(min.) | 90 |
| Time at max. temp.(min.) | 120 |
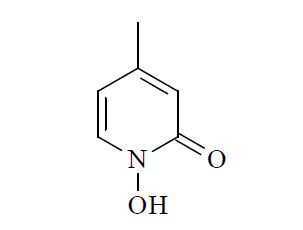
Laccase used in this experiment was produced from Trichophyton sp. LKY-7 (Tr. LKY-7) which was isolated and characterized in previous work (11). Tr. LKY-7 was grown in glucose-peptone broth supplemented with wood meal (Quercus variabilis) of 1% (W/W) as a laccase inducer at 29℃ for 14 days. And then laccase was acquired by single-step purification of Tr. LKY-7 cultures in Q-sepharose column (Pharmacia co.) as described in previous work (11). Partially purified xylanase derived from Aureobasidium pullulans was kindly provided by Dr. Hoon Kim (College of Pharmacy, Sunchon National Uni.). N-hydroxy-2-pyridone analogue (Fig. 1) as a laccase mediator was kindly supplied by Dr. Kaichang Li (Oregon State Uni., USA).
2.2 Enzyme assays
Xylanase activity was measured by the dinitrosalicyclic acid (DNS) method using birchwood xylan (Sigma Co.). One unit xylanase was defined as an amount of the enzyme required to product 1 μ mol of xylose from birch wood xylan per minute under the assay conditions(40℃, pH 4.5). Laccase activity was determined by measuring the oxidation of 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) in a sodium acetate buffer (50 mM, pH 4.5) at 420 nm (ε max=3.6x104M-1cm-1). One unit (U) of laccase activity was defined as 1.0 μ mol of product formed per minute under the assay conditions.
2.3 Xylanase (X) bleaching
The treatment of X was performed with 5 g of HwKP based on the oven-dry weight pulp (odp) at 2% consistency in sodium acetate buffer (50 mM, pH 4.5) in the 500 ml conical flask. The dose of xylanase was 5 U per gram of odp. The pulp suspension was incubated periodically in rotary shaker (150 rpm) at 5 0℃ for 16 hrs. After X treatment, the pulp was filtered and the filtrate was collected for measuring of the residual xylanase activity. The treated pulp was washed thoroughly and then was subjected to alkaline extraction (E) and alkaline/ hydrogen peroxide treatment (P), respectively. For a comparison, HwKP was treated with E stage and P stage, respectively, without X treatment. Residual xylanase activities were measured from the filtrates in the absence and in thepresence of pulp.
2.4 Laccase-mediator system (LM) bleaching
The treatment of LM was performed with 5 g of HwKP (odp) at 2% consistency in sodium acetate buffer (50 mM, pH 4.5) in 500ml flask. The doses of TrL and NHP were 15 U and 10 mg per gram of odp, respectively. The pulp suspension was incubated periodically in rotary shaker (150 rpm) at 50℃ for 16 hrs. After LM treatment, the pulp was filtered and the filtrate was collected for measuring of the residual laccase activity. The treated pulp was washed thoroughly and then subjected to E stage and P stage, respectively. For a comparison, HwKP was treated with E stage and P stage, respectively, without LM treatment. Residual laccase activities were measured from the filtrates in the presence and in the absence of pulp.
2.5 Xylanase and laccase-mediator system bleaching
HwKP was treated with X followed by LM sequentially (XLM) and with X and LM simultaneously (X+LM). For the XLM treatment, HwKP adjusted to 2% consistency in sodium acetate buffer (50 mM, pH 4.5) was treated with 5 U of xylanase per gram of odp at 5 0℃ for 4 hrs. After X treatment, the pulp was washed with distilled water and then treated with 15 U of TrL per gram of odp together with 10 mg of NHP per gram of odp at 50℃ for 4 hrs. Also, the pulp was treated with XLM stage at 5% and 10% pulp consistency. In case of 5% or 10% consistency, the treatment was performed in plastic ziploc bag of water bath with kneading the bag occasionally during the treatment. For the X+LM treatment, HwKP adjusted to 2% consistency in sodium acetate buffer (50 mM, pH 4.5) was treated with 5 U of xylanase, 15 U of TrL, and 10 mg of NHP per gram of odp at 50℃ for 4 hrs. Residual xylanase and laccase activities were measured with the sample aliquot taken from the reaction mixture at hourly intervals during the X+LM treatment in the presence and in the absence of pulp. After all the treatment, pulps were filtered and washed thoroughly, and then subjected to P stage.
2.6 Alkaline extraction (E) and alkaline/ hydrogen peroxide treatment (P)
The treated pulp was suspended at 5% consistency in the plastic ziploc bag and subjected to E and P stage, respectively. E stage was done with 2% NaOH per gram of odp at 90℃ for 2 hrs. P stage was done with 2% NaOH, 2.5% H2O2,1% DTPA (diethylenetriaminepentaacetic acid), and 0.05% MgSO4 per gram of odp at 90℃ for 4 hrs. Pulp was mixed occasionally by kneading the bag during the incubation.
2.7 Pulp properties
The bleached pulp was disintegrated for 30 seconds in a Waring blender and then formed into handsheet according to TAPPI test method T218 om-83. Brightness was determined using TAPPI test method T525 om-86 and kappa number was measured according to the micro kappa number method (12). For a comparison, delignification was calculated from the kappa number of initial pulp and treated pulp.
3. Results and Discussion
3.1 Xylanase bleaching of HwKP
Generally, it is known that the xylanases derived from strains of bacterial origin are the most effective between pH 6 and 9, while those derived from strains of fungal origin should be used within the pH range of 4-6 (13). Thus, to joint with laccase derived from wood-degrading fungus Tr. LKY-7 in enzymatic bleaching, we used the xylanase derived from the fungal strain, Aureobasidium pullulans. An optimal pH (4-5) and temperature (40-50℃) for xylanase were determined by our preliminary experiments (data not shown).
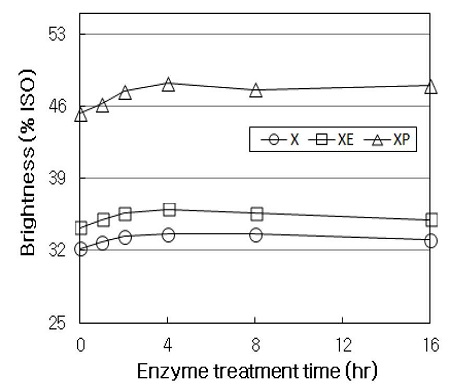
Fig. 2 shows the brightness of HwKP treated with X stage, XE sequence, and XP sequence. When the initial pulp was treated with E stage without enzymatic treatment (at time zero in XE), the brightness of pulp increased from 32.2% ISO to 34.2% ISO. By contrast, P stage leaded to so much higher increase of brightness than that obtained in E stage. Thus, P stage increased the brightness of the initial pulp from 32.2% ISO to 45.3% ISO (at time zero in XP). X stage alone did not significantly improve the brightness of pulp throughout the treatment, showing a slight increase of about 1.4% ISO in comparison with the initial pulp. This result is consistent with that presented by Nelson et al. (14), who indicated that there is no increase in brightness immediately after the X stage, while the treated pulp shows the increased brightness in subsequent E stage, suggesting that xylanase exhibits a bleach-boosting effect. In XE sequence, the brightness gradually increased from the beginning of the treatment to 4 hrs, and then a slight decrease up to 16 hrs. XE sequence increased the brightness of pulp from 34.2% ISO to 36% ISO, and it was about 2.4% ISO higher than that obtained in X stage throughout the treatment. In XP sequence, the brightness increased up to 4 hrs of treatment, and then levelled off. XP sequence increased the brightness of pulp from 45.3% ISO to 48.2% ISO, and it was about 14% ISO and 12% ISO higher than those obtained in X stage and XE sequence throughout the treatment, respectively. The subsequent treatment with E and P stage in combination with X stage was reported to improve the bleaching effect of pulp (15). That is, the subsequent treatments are suggested to facilitate the release of xylans containing chromophoric groups or the dissolution of lignin-carbohydrate fragments that is formed in pulp during kraft pulping. The relatively high brightening of pulp observed in XP sequence seems to be ascribed to hydrogen peroxide which is mainly acts by brightening pulp in the final bleaching stage. These results indicated that although the X stage alone does not significantly increase the brightness of pulp, the subsequent E stage or P stage enhance the brightness of pulp treated with X stage.
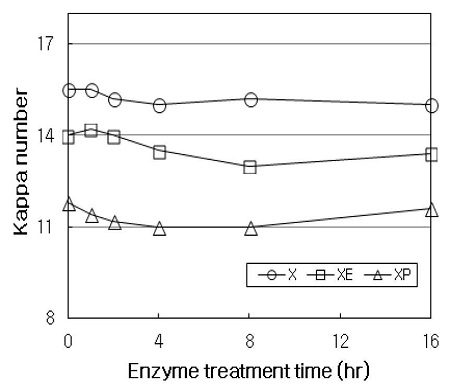
Fig. 3 shows the kappa number of treated HwKP. When the initial pulp was treated with E stage without enzymatic treatment (at time zero in XE), the kappa number of initial pulp decreased from 15.5 to 14, which is corresponded to about 9.7% delignification. When the initial pulp was treated with P stage without enzymatic treatment (at time zero in XP), the kappa number decreased from 15.5 to 11.8, corresponded to about 24% delignification. This result was thought to be, as with the brightness of the initial pulp, that E stage or P stage facilitates the dissolution of lignincarbohydrate fragments in pulp that are modified during kraft pulping process (15). In X stage, the kappa number of pulp decreased by about 0.5 (about 3.2% delignification) throughout the treatment, compared with the initial pulp. In XE sequence, the kappa number decreased by about 1 (about 7% delignification) throughout the treatment. In XP sequence, the kappa number decreased by about 0.8 after 4 hrs of treatment (about 7% delignification), and then rather increased slightly up to 16 hrs. These results indicated that X stage alone have little effect on decreasing the kappa number of pulp, whereas the subsequent treatment with E or P stage exhibits a little enhancing the delignification of pulp. At latter stage of treatment, the increase of kappa number might be associated with the relative greater lignin content due to the removal of hemicelluloses with treatment of X stage (16). An optimal treatment time for X stage was found to be 2-4 hrs, since there was no substantial improvement in pulp properties at longer time than 4 hrs,.
3.2 Laccase-mediator system bleaching of HwKP
Laccase-mediator system has enabled the development of alternative TCF bleaching sequences, whereas it also has the disadvantages that the mediator is expensive and potentially toxic. In the previous study (10), we found that NHP, N-hydroxy-2-pyridone analog, is able to enhance the delignification of TrL as a laccase mediator. Besides, NHP has the advantages which is a low molecular weight, cheap, and water- soluble, compared with other mediators.
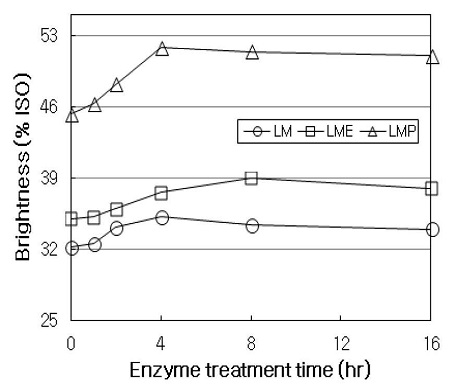
Fig. 4 shows the brightness of HwKP treated with LM stage, LME sequence, and LMP sequence, respectively. When HwKP was treated with LM stage, the brightness of pulp gradually increased by about 3% ISO in 4 hrs of treatment, and then slightly decreased. This decrease of brightness in the latter part of LM treatment may have resulted from the formation of chromophoric groups during the oxidation of lignin (17). No appreciable difference in brightness at an early stage of treatment (up to 4 hrs) between LM stage and LME sequence was observed, whereas at a latter stage (up to 16 hrs), the brightness in LME sequence was higher than that obtained in LM stage. In LME sequence, the brightness increased from 35% ISO to 39% ISO, and it was about 3.8% ISO higher than that in LM stage throughout the treatment. In LMP sequence, the brightness sharply increased up to 4 hrs treatment, and then levelled off. LMP sequence increased the brightness of pulp from 45.3% ISO to 51.8% ISO throughout the treatment, and it was about 16% ISO and 13% ISO higher than those obtained in LM stage and LME sequence, respectively. This could be explained by the fact that lignin structures are altered and modified by LM stage, and the modified lignin fractions are removed by the subsequent treatment hence increased brightness observed after E stage or P stage (18).
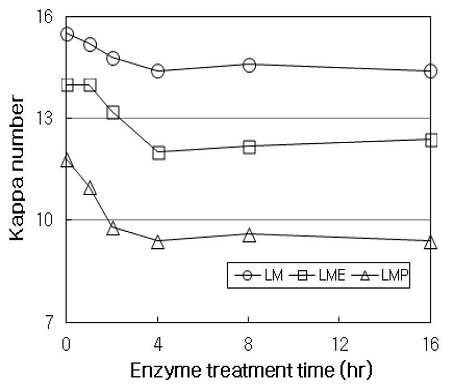
Fig. 5 shows the kappa number of treated HwKP. In LM stage, the kappa number decreased by about 1.1 (about 7% delignification) throughout the treatment with respect to the initial pulp. The lignification of LM stage was lower than that of the initial pulp after only E stage without enzymatic treatment. By contrast, in LME sequence, the kappa number decreased by 2 (about 14% delignification), and it was about 2.4 lower than that obtained in LM stage. This, as previously presented by Fillet et al. (18), suggested that LM stage alters and modifies lignin present on pulp in such a way that a modified fraction of lignin becomes soluble during the E stage. In LMP sequence, the kappa number significantly decreased up to 4 hrs, and then levelled off approximately. The kappa number decreased by 2.4 throughout the treatment (20% delignification), and it was about 5 and 2.6 smaller than those obtained in LM stage and LME sequence, respectively. It should be noted here that LMP sequence decreased the kappa number by 6.1 with respect to the initial pulp, which is corresponded to about 39% delignification. In general, it has known that in P stage, the alkaline pH used facilitates dissolution of lignin by causing the ionization of acid groups formed in previous stages, and also hydrogen peroxide oxidizes residual lignin in pulp and increase its brightness as a result (19). Therefore, it can be concluded from this results that P stage significantly enhance not only the increase of brightness but also the decrease of kappa number.
3.3 Xylanaseandlaccase-NHPbleaching
To boost the bleaching effect of pulp, HwKP was treated with XLM or X+LM, followed by P stage. Table 2 showes the effects of bleaching sequence on pulp properties. When HwKP was treated with XLM sequentially followed by P stage, the brightness of pulp increased to 56.3% ISO and the kappa number decreased to 8.2. In comparison with the initial pulp before and after P stage, the brightness of pulp increased by about 24% ISO and 11% ISO, respectively, and the kappa number decreased by about 7.3 (47% delignification) and 3.6 (30% delignification), respectively. This result suggested that the pretreatment with X stage enhanced the bleaching effects of pulp in LM stage, although X stage alone did not exhibited the distinct effectiveness of bleaching. That is, partial enzymatic hydrolysis of xylan in pulp is thought to render the pulp more permeable by increasing fiber swelling and break lignin-carbohydrate bonds, thereby boosting the effect of the laccase-mediator system in reducing the content of residual lignin (8, 9). By contrast, when HwKP was treated with X+LM simultaneously followed by P stage, the brightness resulted in 52.4% ISO and the kappa number resulted in 9.1. These results were approximately similar to that obtained in LMP sequence. This may be due to the inactivation of enzymes, especially in case of xylanase, leaded by the NHP, as revealed by the analysis of residual enzyme activity (Fig. 8). It is consistent with that observed by Valls et al (8), who presented that applying xylanase and laccase-mediator jointly in a single step is not effective due to the inactivation of xylanase. However, when the xylanase and laccase-mediator are applied in sequence, the bleaching effects are enhanced significantly. Also, this inactivation of xylanase in X+LM stage was supported by Martin-Samperdo et al. (9), who confirmed that when xylanase is applied jointly with LMS, the higher hemicellulose content is observed. Moreover, a lower removal of hexenuronic acid is observed after X+LM treatment, compared to that found in the sequential application of XLM.
Table 2
Effect of the bleaching sequence on pulp properties
| Brightness (% ISO) | Kappa number | ||
|---|---|---|---|
| Initial pulp | Before alkaline/H2O2 | 32.2 | 15.5 |
| After alkaline/H2O2 | 45.3 | 11.8 | |
| Pulp treated with enzymes, followed by P stage | XP | 48.2 | 11.0 |
| LMP | 51.8 | 9.4 | |
| XLMP | 56.3 | 8.2 | |
| (X+LM)P | 52.4 | 9.1 | |
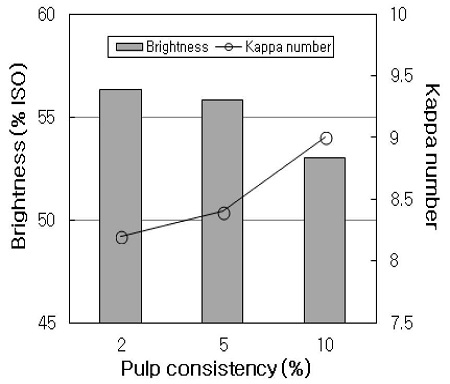
The pulp consistency in laccase-mediator bleaching is favorable as high as possible in order to stabilize laccase and to give better contact between the mediator and fibers, and to improve the bleaching effects as a consequence (19). When HwKP was treated with XLM stage at 2%, 5%, and 10% consistency, as shown in Fig. 6, no appreciable differences of the bleaching effect were observed between the 2% and 5% consistency, although the kappa number appeared to be a little lower at 2% consistency than that of at 5% consistency. However, at 10% consistency, the brightness decreased by about 2.3% ISO and the kappa number increased by about 0.8, compared with 2% consistency. This result is similar with that observed by Geng at al (20), who found that the pulp consistency negatively affects the bleaching efficacy of a laccase-mediator system when the pulp consistency is as high as 10%. Presumably, the diffusion rate or penetration rate of laccase and mediator would be lower at a higher pulp consistency. On the other hand, Moldes et al (19) indicated that no clear differences of the bleaching effects are observed between 5% and 10% consistency, therefore 10% is better choice for mill application as it substantially reduces water consumption. Possibly, the difference of these results can be ascribed to a different raw material used, a disparate lignin composition, and a different mediator.
3.4 Stability of enzymes during the incubation
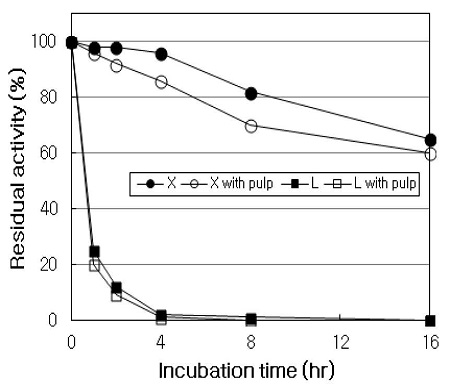
During the incubation of X stage or LM stage at 50℃ for 16 hrs, xylanase and laccase activities were monitored in the absence or in the presence of pulp. As shown in Fig. 7, in X stage, xylanase activity in the absence of pulp was relatively stable until 4 hrs of incubation, and then decreased to 65% of residual activity after 16 hrs. In the presence of pulp, however, xylanase activity was a little lower than those in the absence of pulp throughout the incubation. In general, it has known that the presence of pulp in enzymatic pulp bleaching acts a protective role of enzyme, thus lead to reduce the inactivation of enzyme (19, 21). The decrease of xylanase activity, here, might be due to the adsorption of enzyme to the pulp fiber which would give rise to a decrease in the activity of enzyme in solution (6). In LM stage, laccase activity in the absence of pulp so rapidly decreased from the beginning of incubation that the residual activity decreased to about 25% in the first 1 hr and below 1% after 4 hrs. Similarly, laccase in the presence of pulp was rapidly inactivated from the beginning of incubation, and no protective effect of pulp on laccase was observed.
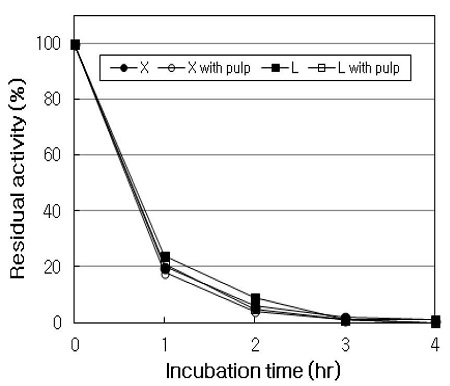
During the incubation of XLM at 50℃ for 4 hrs, xylanase and laccase activities were monitored periodically in the absence or in the presence of pulp. As shown in Fig. 8, xylanase and laccase were so strongly inactivated by NHP that the residual activities sharply decreased throughout the incubation. In the absence of pulp, the residual activities of xylanase and laccase decreased to the level of about 20% and 24% in the first 1 hr, respectively, while in the presence of pulp, it decreased to 18% and 21% in 1 hr, respectively. No protective effect of pulp on xylanase and laccase activities was observed throughout incubation, and the difference of residual activities between without and with pulp was not great. And the residual activities of xylanase and laccase were hardly detected after 3 hrs of incubation. Moldes et al. (19) presented that although the presence of pulp stabilizes enzyme to some extent against mediator, no protective effect is apparent after 2 hrs of incubation. On the other hand, Valls et al. (21) presented that the presence pulp reduces the laccase inactivation by 37% throughout incubation in the laccase-mediator system, and laccase activity is little varied during the first hour of treatment in the presence of pulp. From these results, it suggested that the extent of enzyme inactivation might be varied with the mediator, the temperature of incubation, and the pulp source.
4. Conclusions
To enhance the bleaching effect of laccase-mediator system, X derived from Aurreobasidiumpullulans and LM using TrL and NHP as new mediator were applied in enzymatic bleaching of HwKP. Also, the effects of subsequent treatment with E stage or P stage were examined. In X or LM stage alone, an appreciable bleaching effect was not observed, whereas subsequent E stage or P stage enhanced the increase of brightness and the decrease of kappa number. Especially, P stage significantly enhanced the bleaching effect in enzymatic bleaching of HwKP. Bleaching of HwKP with XLM sequentially gave significantly higher pulp brightness and lower kappa number than that obtained after the treatment of HwKP with X+LM simultaneously. When HwKP was sequentially treated with XLM followed by P stage, the brightness increased by 11% ISO and the kappa number decreased by 3.6 in comparison with the initial pulp. By contrast, when HwKP was treated with X+LM simultaneously followed by P stage, a distinct bleaching effect of pulp was not observed. Bleaching of HwKP with XLM performed better in low pulp consistency (2%) than in high consistency (10%). No apparent differences of the brightness and the kappa number of HwKP between the 2% and 5% consistency were observed. X and TrL were so strongly inactivated by NHP that the residual activities of xylanase and laccase were hardly detected after 3 hrs of incubation both in the absence and the presence of pulp.