1. Introduction
The most important advantage of paper is its recyclability.1) Paper is recycled as a raw material for the manufacture of other paper products, and recycled wastepaper is classified into many groups such as old newspapers (ONPs), old corrugated containers (OCCs), old magazines (OMGs), and mixed office waste (MOW). Paperboard-grade products, including linerboard and white-lined chipboard, have been manufactured from recycled wastepaper, and the production of paperboard grade products accounts for approximately 50% of the paper products produced in South Korea.2) Therefore, recycled wastepaper is a very important resource for the Korean paper industry.
Among all wastepaper, the white varieties, including OMGs, MOW, and white ledger paper, are widely used to produce white-lined chipboard, which is used for food packaging, pharmaceuticals, detergents, and other products.3) White wastepaper contains fluorescent whitening agents (FWAs), which are subject to safety regulation in chipboard mills.4) In addition, costumers worry about the toxicity of FWAs in recycled paper products. Because it is inevitable to use white wastepaper to manufacture paperboard in the Republic of Korea, it is necessary to develop a method to remove or control FWAs originating from white wastepaper.
The FWAs used in the paper industry are classified into three groups according to the level of sulfonic acid present: disulfo-FWA (D-FWA), tetrasulfo-FWA (T-FWA), and hexasulfo-FWA (H-FWA).5,6) FWAs are used in the papermaking process because FWAs have their own affinity to cellulosic fibers and wide-ranging solubility in aqueous solutions.5) Therefore, papermakers should analyze the type of FWA for understanding FWA detachment and distribution change during the recycling process.7)
In this study, the effect of recycling on FWA detachment from white wastepaper and the distribution change of FWAs in the sheet was investigated. Three types of papers were prepared, dyed with various FWAs, disintegrated, and again made into handsheets. The recycling rounds were repeated up to ten times for D-FWA and five times for T-FWA and H-FWA. After each round of recycling, the CIE whiteness, ISO brightness, and fluorescence index of the handsheet were measured, and the fluorescence reduction was calculated as an indicator, which represents the FWA detachment from wastepaper, using the fluorescence indices before recycling and after each round of recycling. Finally, the distribution of FWAs in the cross section of the handsheet was observed using a confocal laser scanning microscope (CLSM).
2. Materials and Methods
2.1 Materials
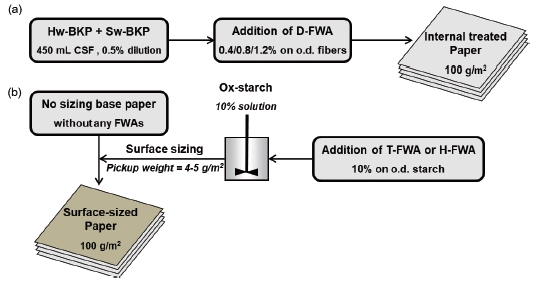
Three types of FWAs, including disulfo-FWA (D-FWA), tetrasulfo-FWA (T-FWA), and hexasulfo-FWA (H-FWA) are commercially available products. Bleached softwood kraft pulp (Sw-BKP) and bleached hardwood kraft pulp (Hw-BKP) were provided by Moorim Paper (Jinju, Republic of Korea), and used to make handsheets that were dyed with D-FWA. Non-sized fine paper with a grammage of 95 g/m2 was used as the base paper for surface sizing with T-FWA or H-FWA. Oxidized starch, which was provided by Daesang (Icheon, Republic of Korea), was used for surface sizing.
2.2 Stock preparation and manufacture of paper dyed with FWA
Sw-BKP and Hw-BKP were beaten to 450±5 mL CSF using a laboratory valley beater (Daeil Machinery Co. Ltd., Daejeon, Republic of Korea). The pulp samples were mixed in a ratio of 2:8 (Sw-BKP:Hw-BKP). The mixed pulp slurry was then diluted to a 0.5% consistency for handsheet preparation. Handsheets with a grammage of 100±5 g/m2 were produced according to TAPPI T205 sp-06 “Forming handsheets for physical tests of pulp” after D-FWA was added to the pulp slurry and mixed for 2 min at 600 rpm. The addition percentages of D-FWA were kept at 0.4%, 0.8%, and 1.2% of the oven-dried fibers. The handsheets were wet pressed at 345 kPa for 5 min and then dried at 120℃ using a laboratory wet press (model 326; Wintree Corporation, Japan) and a cylinder dryer (Daeil Machinery Co. Ltd., South Korea), respectively. The prepared handsheets were conditioned at 23℃ and 50% RH, and then analyzed.
2.3 Surface sizing of paper with a starch solution that included FWAs
Preparation of the starch solution and surface sizing were conducted in the same manner as described in our previous studies.8,9) The 10% starch solution was made by cooking oxidized starch at 90℃ to 95℃ for 30 min. After the temperature of the solution reached approximately 50℃, T-FWA or H-FWA was added, and the resulting solution was mixed for 30 min. The addition of the FWAs was kept at 10% and 20% based on the oven-dried starch.
The surface-sizing process was conducted using a rod coater (HT-BC-ST, Hantech, Republic of Korea). The topside of a never-sized sheet of paper was surface-sized, and the paper was then dried in an air dryer for 150 s. To prevent the surface-sized paper from curling, it was passed through a cylinder dryer twice at 120℃. After the topside of the base paper was surface-sized, the bottom side of the paper underwent the same treatment. The final pickup weight was controlled at 2.0 to 2.5 g/m2 for each side. The surface-sized paper was conditioned at 23℃ and 50% RH before analysis.
Figure 1 shows a flow diagram of the preparation of model paper containing FWAs.
2.4 Simulation of the recycling number and measurement of optical properties of the model paper treated with FWAs
The first round of recycling was performed by the disintegration of paper dyed with FWAs followed by subsequent handsheet preparation.7) The disintegration of paper dyed with FWAs was carried out at a 2% level using a standard disintegrator; subsequently, handsheets with a grammage of 100±5 g/m2 were made from the disintegrated pulp according to TAPPI T205 sp-06 “Forming handsheets for physical tests of pulp”. The handsheets were wet pressed at 345 kPa for 5 min and dried at 120℃ in a laboratory wet press and in a cylinder dryer, respectively. After each round of recycling, the handsheets were conditioned at 23℃ and 50% RH; and then, their CIE whiteness, ISO brightness, and fluorescence indices were measured. Here, the fluorescence index was determined from the CIE whiteness measurements under a D65 illuminant. The fluorescence reduction from the paper dyed with FWAs was calculated as an indicator, which represents the FWA detachment from paper, using the fluorescence indices before recycling and after each round of recycling.7) The distribution of FWAs in the cross section of the paper was observed using a CLSM (JP/FW 1000, Olympus, Japan).9)
where FIbefore is the initial fluorescence index induced by CIE whiteness under D65 illumination before recycling, and FIafter is the fluorescence index induced by CIE whiteness under D65 illumination after each round of recycling.
3. Results and Discussion
3.1 Effect of recycling on optical properties of paper treated with FWAs
Three types of papers were prepared using D-FWA, T-FWA, and H-FWA solutions. The D-FWA was added into the stock mix itself, whereas T-FWA and H-FWA were used on a laboratory coater for surface-sizing with oxidized starch. Fig. 2 shows the CIE whiteness and the ISO brightness values of the paper containing D-FWA as a function of the number of recycling rounds.
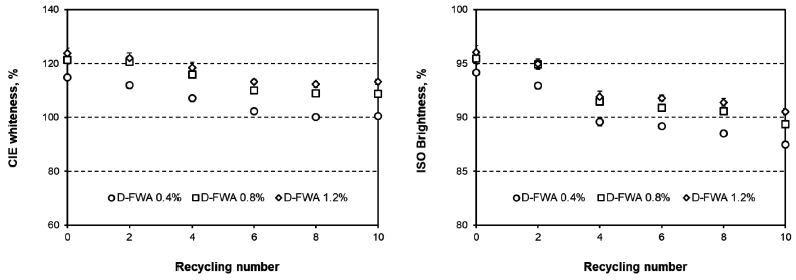
Fig. 2.
CIE whiteness (left) and ISO brightness (right) of the paper containing D-FWA as a function of the number of recycling rounds.
The initial CIE whiteness and ISO brightness values of the paper were proportional to the addition level of D-FWA.10) When the addition of D-FWA was more than 0.8%, the CIE whiteness of the paper was more than 120% and the ISO brightness of the paper was more than 95%. As the recycling was carried out, the optical properties of the handsheets exponentially decreased.
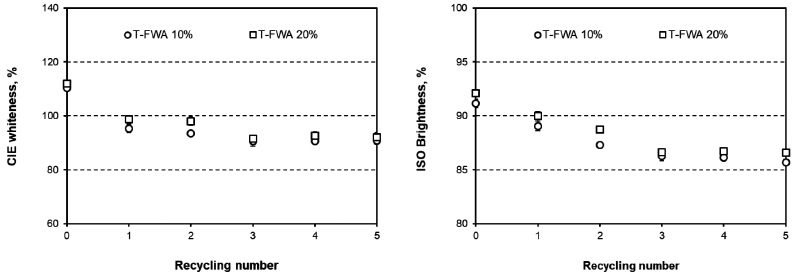
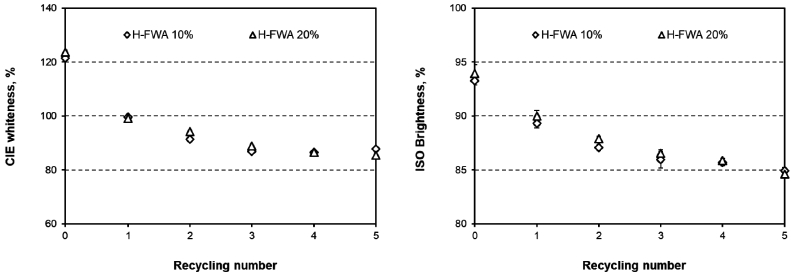
Figs. 3 and 4 show the CIE whiteness and the ISO brightness values of the paper that was surface-sized with T-FWA or H-FWA. Before recycling, the paper surface-sized with H-FWA showed higher optical properties than the paper that was surface-sized with T-FWA.9) As the recycling was performed, the CIE whiteness and ISO brightness of the handsheet exponentially decreased. Compared to D-FWA, T-FWA, and H-FWA showed a higher reduction in the optical properties irrespective of the lower number of recycling rounds. In addition, H-FWA showed the highest reduction in the optical properties among the FWAs used in this study.
3.2 Effect of recycling on the fluorescence index and fluorescence reduction in the paper treated with FWAs
FWA addition improves the optical properties of paper by absorbing more UV light and emitting fluorescence.11) The fluorescence emission can be determined by measuring the fluorescence indices of whiteness or brightness.12) In this study, the fluorescence index of whiteness under the D65 illuminant was measured to identify the existence of FWAs in the handsheets. The fluorescence reduction was calculated using the fluorescence indices before recycling and after each round of recycling.
Fig. 5 shows the fluorescence index and fluorescence reduction for paper dyed with D-FWA. The initial fluorescence index of the paper dyed with D-FWA was proportional to the addition level of D-FWA. After recycling, the fluorescence index of the handsheet decreased, which means that D-FWA was desorbed from the paper. The fluorescence reduction of D-FWA logarithmically increased as a function of the number of recycling rounds. However, the fluorescence reduction of D-FWA did not exceed 25% even though the recycling processes were performed ten times.
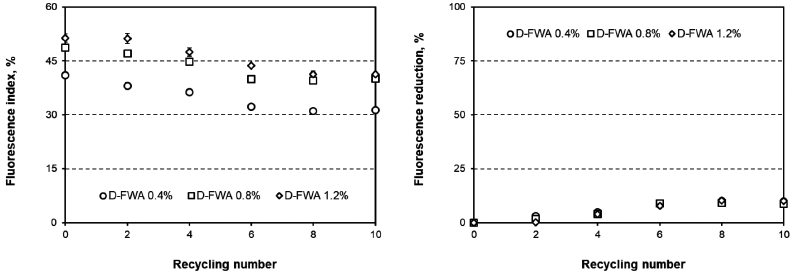
Fig. 5.
Fluorescence index (left) and fluorescence reduction (right) for the paper containing D-FWA as a function of the number of recycling rounds.
Figs. 6 and 7 show the fluorescence indices and the fluorescence reduction in the paper surface-sized with T-FWA or H-FWA. The fluorescence indices of the paper decreased as the number of recycling rounds increased. As the number of recycling processes increased to five, the T-FWA and H-FWA reduction logarithmically increased together. However, the maximum fluorescence reduction varied according to the type of FWA used. The fluorescence reduction of T-FWA showed almost 50% when the paper was surface-sized with T-FWA and recycled five times. H-FWA exhibited almost a 75% fluorescence reduction after five recycling processes. These results corresponded to the differences in the optical properties as a function of the number of recycling rounds.
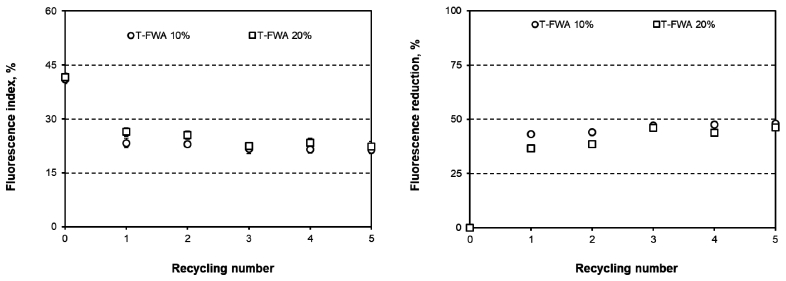
Fig. 6.
Fluorescence index (left) and fluorescence reduction (right) for the paper surface-sized with T-FWA as a function of the number of recycling rounds.
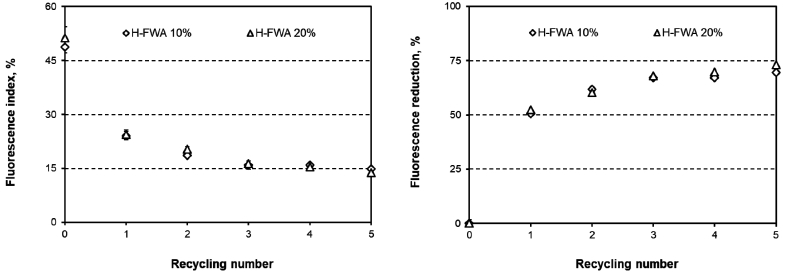
Fig. 7.
Fluorescence index (left) and fluorescence reduction (right) for the paper surface-sized with H-FWA as a function of the number of recycling rounds.
The D-FWA reagent adsorbs on the cellulosic fibers via hydrogen bonding.5) However, T-FWA and H-FWA are delivered to the surface of the paper by starch or other carrier chemicals, and there are no chemical bonds formed between T-FWA or H-FWA and the cellulosic fibers.8) When white wastepaper containing FWAs was recycled, the starch or carrier chemicals can be extracted from the wastepaper with T-FWA or H-FWA. However, D-FWA was not intentionally extracted from the paper because it was attached to the cellulose fibers more strongly than T-FWA and H-FWA. Therefore, we concluded that D-FWA showed a much lower fluorescence reduction than T-FWA and H-FWA.
3.3 Effect of recycling on the FWA distribution from the cross section of the paper
The fluorescence emission images from the cross section of the paper were captured by CLSM to analyze the effect of recycling on the distributions of FWAs before and after recycling. Fig. 8 shows the D-FWA distribution before and after the recycling process. The D-FWA was distributed evenly in the cross sections of the paper before recycling, and the initial distribution of D-FWA was maintained until four rounds of recycling. Figs. 9 and 10 show T-FWA and H-FWA distributions before and after the recycling process. The T-FWA and H-FWA reagents were located on both surfaces of the paper, which means that T-FWA and H-FWA had a high level of surface holdout.8) However, T-FWA and H-FWA were considerably desorbed from the paper, as the recycling process was preceeded, as shown in Figs. 6 and 7. Both T-FWA and H-FWA showed an especially similar distribution with that of D-FWA after three rounds of recycling.
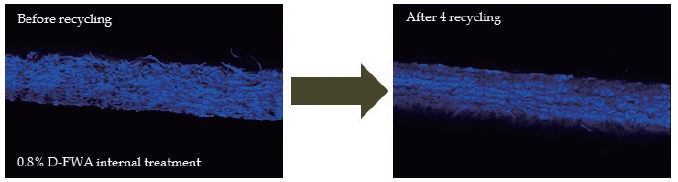
Fig. 8.
D-FWA distribution in the cross section of the paper containing 0.8% D-FWA before (left), and after (right) four rounds of recycling.
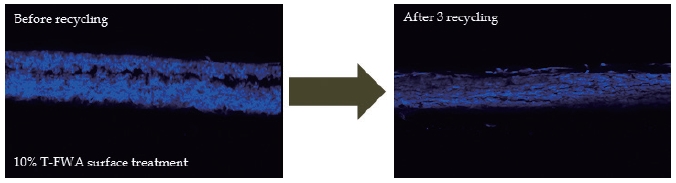
Fig. 9.
T-FWA distribution in the cross section of the paper surface-sized with 10% T-FWA before (left), and after (right) three rounds of recycling.
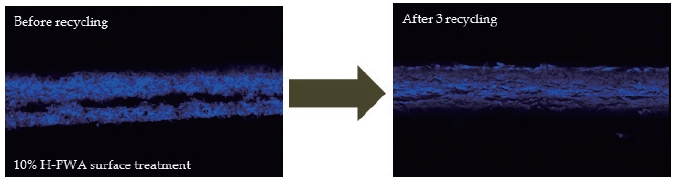
Fig. 10.
H-FWA distribution in the cross section of the paper surface-sized with 10% H-FWA before (left), and after (right) three rounds of recycling.
The distribution changes in T-FWA and H-FWA can be explained by re-adsorption. When T-FWA and H-FWA were extracted from the paper that had been surface-sized with them, the FWAs stayed soluble in the stock. As the contact time between FWAs and the cellulosic fibers increased, some of the detached FWAs re-adsorbed on the cellulosic fibers because of the sulfonic acids present on a FWA molecule.
4. Conclusions
D-FWA exhibited a much lower fluorescence reduction than T-FWA and H-FWA. The CIE whiteness and ISO brightness of the paper containing D-FWA decreased by less than those of the papers containing T-FWA and H-FWA reagents when the number of recycling processes increased.
The D-FWA was distributed evenly in the cross section of the paper before recycling, and the initial distribution of D-FWA was maintained until four rounds of recycling. However, T-FWA and H-FWA were significantly detached from the papers but showed a similar distribution as that of D-FWA when the recycling process was carried out because of re-adsorption.
The D-FWA adsorbed on the cellulosic fibers via hydrogen bonding. However, T-FWA and H-FWA were delivered to the surface of the paper by starch or other carrier chemicals, and there were no chemical bonds formed between T-FWA or H-FWA, and the cellulosic fibers. Therefore, the D-FWA was possibly not extracted from paper because it was attached to the cellulose fibers more strongly than T-FWA and H-FWA.
Finally, we concluded that surface-treating FWAs can be detached easily compared with internal FWA and some of the detached FWAs re-adsorbed on the cellulosic fibers during the recycling process.