1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Organosolv pulping of bark residues
2.3 Lignin recovery from black liquor
2.4 Micronization of bark-derived pulp and film preparation
2.5 Chemical composition analysis of bark residues
2.6 Yield and chemical composition analysis of pulps and recovered lignin
3. Results and Discussion
3.1 Chemical composition of oak bark residues
3.2 Yield and chemical composition of pulps
3.3 Recovery yield and chemical composition of lignin isolated from black liquor
3.4 Micronization behavior and film properties of microfibers
4. Conclusions
1. Introduction
Bark, a significant by-product of the wood industry, accounts for approximately 10–20% of the total log mass and is generated in large quantities during sawing and pulp production [1,2]. However, compared with ordinary woody tissues, bark contains higher levels of ash and lignin but lower cellulose content, which limits its direct use as a raw material for papermaking or pulp production [3]. Consequently, most bark is currently utilized as boiler fuel, horticultural compost, or treated as industrial waste [4]. Nevertheless, bark still contains abundant high-value constituents such as pectin, suberin, starch, fatty acids, resin acids, tannins, and polysaccharide biopolymers, including cellulose and hemicellulose [1,5]. Therefore, with appropriate pretreatment and pulping processes, bark can be converted into high-value bio-based materials such as plastic fillers (bio-fillers), reinforcing fibers for biocomposites, and functional bio-films.
Song et al. [6] extracted fibers from willow bark through alkali treatment, producing bark-derived fibers with high thermal stability. Similarly, Dou et al. [7] reported that willow bark fibers exhibited excellent compatibility with polymer matrices such as polylactic acid (PLA) due to their high lignin content and inherent hydrophobicity. However, most previous studies have utilized raw bark as-is, and there has been little research on a stepwise biorefinery approach using bark residues remaining after tannin extraction.
The residue remaining after tannin extraction is free of water-soluble low-molecular-weight organic compounds, resulting in a simpler and more stable chemical composition. Therefore, it is considered an ideal precursor for separating macromolecules (cellulose, hemicellulose, and lignin) through pulping [8]. Consequently, it is considered an ideal precursor for fractionating macromolecules such as cellulose, hemicellulose, and lignin via pulping processes. Based on this potential, our previous study [9] investigated the extraction yield and quality characteristics of polyphenols and tannins from oak bark according to particle size and solvent composition, and the remaining bark residues were collected for subsequent utilization.
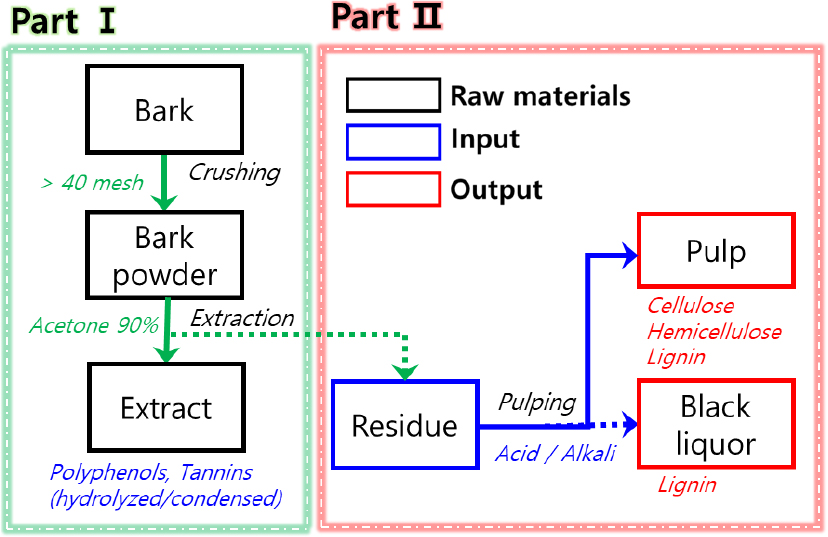
Organosolv pulping, compared with conventional kraft or sulfite pulping, is regarded as an environmentally friendly process that operates under relatively mild (low-temperature and low-pressure) conditions without using sulfur, while allowing for the recovery of high-purity lignin [10,11]. Therefore, this study (Part II) aimed to establish a stepwise resource utilization system for complete valorization of bark by applying organosolv pulping to tannin-extracted bark residues and fractionating microfibers and lignin. To this end, tannin-extracted bark residues prepared from Part I were subjected to acid- and alkali-catalyzed organosolv pulping, and the pulping yield and chemical composition were comparatively analyzed. Furthermore, lignin was isolated from the resulting black liquor under each condition, and its recovery yield and purity were evaluated.
Meanwhile, bark-derived fibers have smaller particle sizes and shorter fiber lengths than those obtained from wood tissues [12,13]. Typically, the production of nanocellulose requires complex pretreatment processes such as mechanical refining (beating and grinding) or chemical pretreatments, including enzymatic hydrolysis, alkaline treatment, or TEMPO-mediated oxidation [10,14,15,16]. However, bark-based fibers are advantageous for fibrillation because their inherently fine structure allows efficient fibrillation without additional pretreatment steps. Therefore, this study prepared microfibers from the pulped residues by high-pressure homogenization without any chemical pretreatment. Such lignin-containing microfibers are expected to exhibit superior compatibility with hydrophobic polymers, such as PLA and polybutylene succinate (PBS), compared to conventional bleached cellulose nanofiber (CNF). Their potential application as eco-friendly film materials derived from bark was also investigated. This study aims to provide foundational knowledge for upgrading underutilized forest biomass to high-value material applications.
2. Materials and Methods
2.1 Materials
The oak bark used in this study was obtained by applying the optimal extraction conditions (>40 mesh particle size and 90% acetone) established in the previous study (Part I) [9] for tannin extraction. The residual samples after extraction were used as raw materials for the pulping and lignin recovery experiments. The residues were repeatedly washed with 70% ethanol and distilled water to remove residual solvents and extractives, and then dried at 60°C until a constant weight was achieved. Ethanol (94.5%), acetone (99.5%), and glycol ether (GE, 98.0%) used in the experiments were purchased from Daejung Chemicals & Metals Co., Ltd. (Korea). Sulfuric acid (H2SO4, 95%) and sodium hydroxide (NaOH, 98%) were used as catalysts for the acidic and alkaline pulping reactions, respectively. All chemicals used were of extra pure grade and were employed without further purification.
2.2 Organosolv pulping of bark residues
After tannin extraction, Organosolv pulping was conducted to utilize the cellulose, hemicellulose, and lignin remaining in the bark residues. The pulping process was divided into two types according to the catalyst system (acid- or alkali-catalyzed methods) (Fig. 1). For the acid-catalyzed pulping, GE containing 3 wt% sulfuric acid was used as the pulping liquor without the addition of water. In the alkali-catalyzed pulping process, GE and distilled water were mixed in a 1:1 (v/v) ratio, and sodium hydroxide (20 wt%) was added to prepare the alkaline pulping liquor. The bark samples were mixed with the prepared pulping liquor at a 1:3 (w/w) ratio and allowed to soak for over 6 h to ensure complete impregnation. The pulping reaction was carried out using an autoclave (JSAT-105, JSR, Korea) under the temperature and time conditions summarized in Table 1. After the reaction was completed, the resulting pulp slurry was vacuum-filtered to separate the black liquor. The solid pulp was repeatedly washed with distilled water to remove residual solvents and insoluble materials, and then oven-dried at 60°C to constant weight for subsequent analyses.
Table 1.
Conditions for acid- and alkali-catalyzed organosolv pulping
| Catalyst type | Wood chips and liquor ratio | Liquor condition | Temperature (°C) | Time (min) |
| Alkali (NaOH) | 1:3 | Water : GE (50:50), NaOH 20% | 120 | 90 |
| Acid (H2SO4) | 1:3 | GE : H2SO4 (97:3) | 120 | 60/90/120 |
2.3 Lignin recovery from black liquor
Lignin was isolated from the recovered black liquor to utilize the by-products generated during the pulping process. For the acid-catalyzed pulping system, the black liquor was mixed with distilled water at a ratio of 1:4 (v/v) and stirred for 30 minutes. Meanwhile, the black liquor obtained from the alkali-catalyzed pulping (initial pH: 14) was acidified to a pH below 3 by adding sulfuric acid and subsequently stirred at 60°C for 1 hour. The resulting precipitates were separated by vacuum filtration, and the residues were repeatedly washed with distilled water until the filtrate became neutral. The purified lignin was dried at 60°C for more than 24 hours.
2.4 Micronization of bark-derived pulp and film preparation
Bark-derived pulp fibers obtained after organosolv pulping were micronization using a laboratory-scale high-pressure homogenizer (Panda PLUS Lab homogenizer 2000, GEA, Germany). The dried pulp was dispersed in distilled water to prepare a 0.5 wt% slurry, and then it was passed through the homogenizer at a pressure of 1,000 bar for 1, 5, 10, and 20 passes to produce microfibers. The resulting microfiber slurry was vacuum-filtered to form wet films with a basis weight of 30 g/m2, which were subsequently dried at 60°C using a gel dryer (SE 1160, Hoefer, USA) to obtain the final bark-based microfiber films.
2.5 Chemical composition analysis of bark residues
To analyze the chemical composition of the bark residues obtained after tannin extraction, the contents of acid-insoluble lignin (Klason lignin, KL) and acid-soluble lignin (ASL) were determined according to the TAPPI T222 om-02 and TAPPI UM250 standard methods. The hydrolysate obtained during the KL analysis was analyzed using high-performance liquid chromatography (HPLC; RID-20A, Shimadzu, Japan) to quantify the cellulose (glucose) and hemicellulose (xylose) contents. A Shodex Sugar SP0810 column (Resonac, Shūnan, Japan) was employed to separate and detect carbohydrate components.
2.6 Yield and chemical composition analysis of pulps and recovered lignin
The yield of the pulps obtained after the pulping reactions was determined, and the chemical compositions of both the pulps and the lignin recovered from the black liquor were analyzed based on the contents of KL, ASL, and total sugars. The analytical methods followed were identical to those described in Section 2.5. Based on the compositional analysis of the isolated lignin, the recovery yield and purity of lignin were compared and evaluated under various pulping conditions (acid- and alkali-catalyzed systems).
3. Results and Discussion
3.1 Chemical composition of oak bark residues
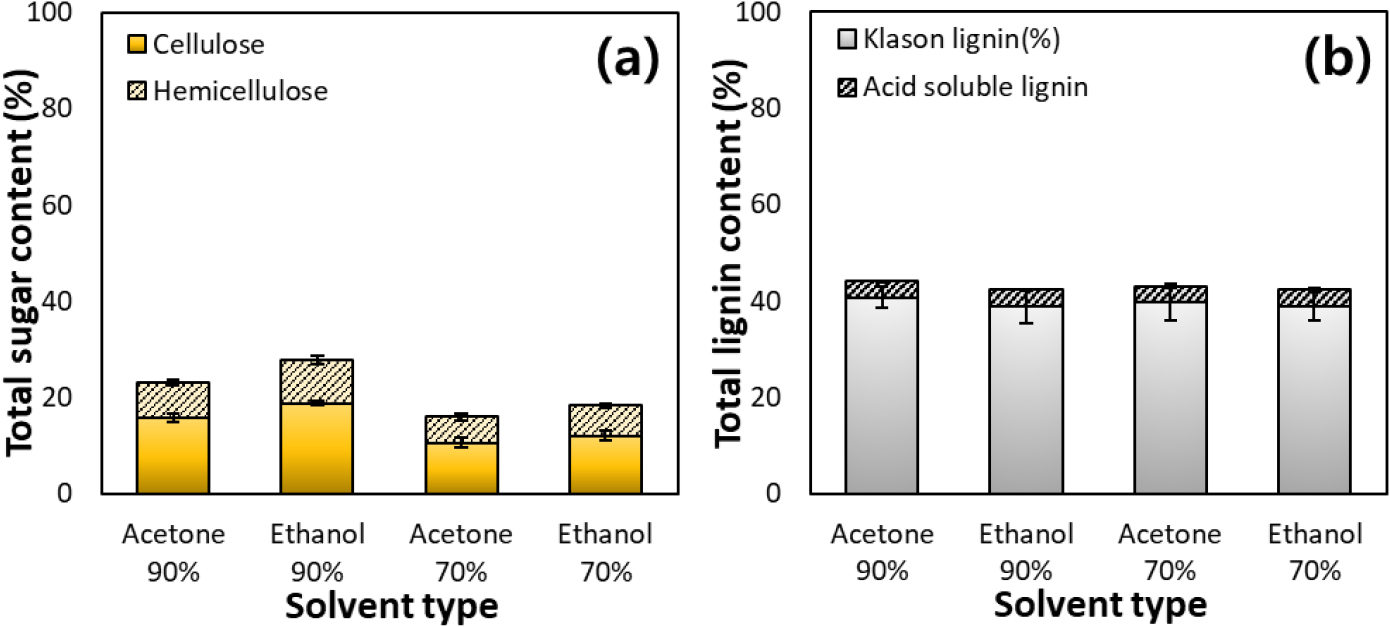
The chemical composition of the oak bark residues obtained after tannin extraction was analyzed in terms of sugar and lignin contents, as shown in Fig. 2. Regardless of the extraction solvent used, the total carbohydrate (cellulose and hemicellulose) content of the residues ranged from 16.0 to 27.8%, with cellulose and hemicellulose contents of 10.6–18.8% and 5.4–9.0%, respectively (Fig. 2a). When 90% acetone and 90% ethanol were used as solvents, higher carbohydrate contents were observed compared to those extracted with 70% solvents. This is attributed to the lower extraction efficiency of polyphenols and tannins in 70% solvents, resulting in the retention of extractable compounds within the residues and, consequently, lower apparent carbohydrate contents.
The lignin analysis revealed that the KL content ranged from 38.9 to 40.7%, and ASL accounted for approximately 3.3–3.5%, resulting in a total lignin content of 42.4–44.1% (Fig. 2b). No significant difference in lignin content was observed among residues extracted with different solvents. This consistency indicates that structural lignin remains tightly bound within the bark cell wall matrix, and its relative proportion is not significantly affected even after removing low-molecular-weight phenolics during extraction [17]. These findings confirm that bark contains a higher lignin content and lower polysaccharide fraction than woody xylem, suggesting the potential formation of lignin-rich pulps during subsequent pulping and micronization processes. In particular, the residue extracted with 90% ethanol exhibited the highest glucose content. This can be attributed to the enhanced removal of non-polar extractives (resins, waxes, and phenolics) by high ethanol concentrations, leading to a relative enrichment of polysaccharides. Therefore, oak bark residues retain substantial lignin and stable carbohydrate fractions even after tannin extraction, indicating their suitability as precursors for organosolv pulping and lignin isolation processes.
3.2 Yield and chemical composition of pulps
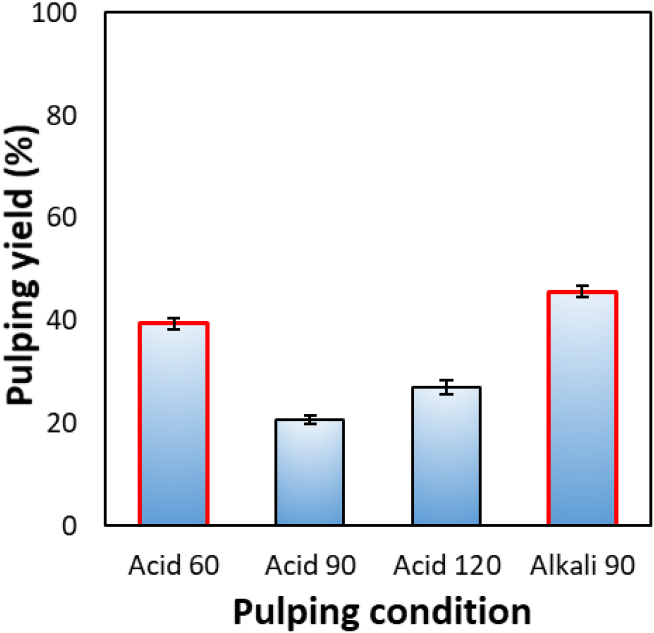
The pulping yield of the oak bark residues after tannin extraction is presented in Fig. 3. Under acidic catalytic conditions (Acid 90 and Acid 120), the pulping yields were 20.8% and 27.0%, respectively, which were relatively low. In contrast, the yields under Acid 60 and Alkali 90 conditions were notably higher, reaching 39.4% and 45.6%, respectively.
In the acid-catalyzed system, sulfuric acid promotes the hydrolytic degradation of cellulose and hemicellulose into low-molecular-weight sugars [18]. At 60°C for 60 min (Acid 60), the milder reaction conditions maintained a pulping yield of approximately 40%. However, As the reaction time extends beyond 90 min, excessive depolymerization occurs, leading to a marked reduction in pulp yield. Meanwhile, although Acid 120 represents a harsher reaction condition than Acid 90, it resulted in a higher pulping yield. This is attributable to the higher lignin and extractives content of bark compared with wood, which can lead to incomplete solubilization under strong acid treatment, followed by re-precipitation or the formation of acid-insoluble residues. Consequently, under the Acid 120 condition, lignin condensation reactions are likely accelerated, thereby increasing the amount of residual solids. Under alkaline conditions (Alkali 90), the pulping yield reached approximately 46%, which can be attributed to the selective cleavage of lignin–hemicellulose linkages by NaOH. This reaction promotes partial lignin dissolution while minimizing severe cellulose degradation [19,20]. A compositional analysis of the resulting fibers was performed to verify this interpretation. Overall, organosolv pulping of oak bark residues was most effective under the Acid 60 and Alkali 90 conditions; therefore, pulps produced under these two representative conditions were selected for subsequent compositional and morphological analyses.
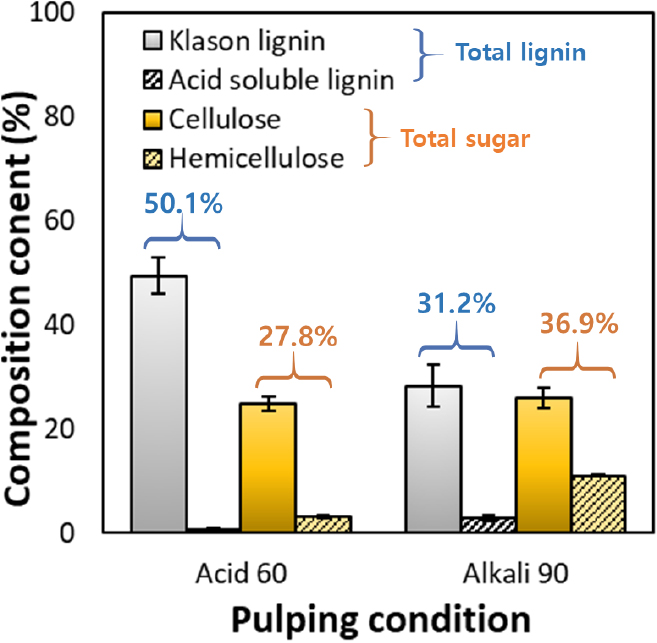
The chemical compositions of the pulps obtained under the Acid 60 and Alkali 90 conditions, which exhibited higher pulping yields, are presented in Fig. 4. Under the Acid 60 condition, the pulp contained 50.1% total lignin and 27.8% total sugars. This result indicates that the selective hydrolysis of hemicellulose by sulfuric acid in the acidic catalytic system reduced the polysaccharide fraction, thereby increasing the relative proportion of lignin and lowering the total carbohydrate content [18]. In contrast, the pulp produced under the Alkali 90 condition exhibited a total lignin content of 31.2% and a total sugar content of 36.9%, showing approximately 19% lower lignin content and 9% higher carbohydrate retention than the acid-treated pulp. This tendency is consistent with the pulping yield trends shown in Fig. 3.
Therefore, alkali-catalyzed organosolv pulping provided the most favorable balance between lignin removal and polysaccharide preservation. These compositional differences are expected to influence the potential end-use applications of the pulps. The Acid 60 pulp, being lignin-rich, may serve as a bio-based filler or reinforcing component in polymer composites, whereas the Alkali 90 pulp, characterized by higher carbohydrate content, is more suitable for fiber-based films or as a reinforcement in biopolymer matrices.
3.3 Recovery yield and chemical composition of lignin isolated from black liquor
The extraction yield of lignin from black liquor and its recovery ratio relative to the lignin content in the original feedstock (bark residue) are presented in Table 2. Both the lignin extraction yield and recovery ratio showed distinct differences depending on the pulping conditions, which were attributed to variations in lignin dissolution and solubilization behavior during pulping. The lignin extracted under the Acid 90 and Acid 120 conditions showed extraction yields of 35.1% and 30.5%, respectively, along with recovery ratios of 79.6% and 69.1%. In contrast, under the Acid 60 condition, the fractionation yield and recovery ratio were relatively low, at 16.5% and 37.3%, respectively. This is presumed to be due to incomplete lignin dissolution at the low reaction temperature (60°C) and short reaction time, causing partial lignin retention within the fiber. For the Alkali 90 condition, the isolated lignin yield and recovery ratio were 13.1% and 29.7%, respectively, significantly lower than those obtained under acidic conditions.
Table 2.
Fractionation and recovery yield of lignin isolated from black liquor
| Liquor | Fractionation yield (%) | Recovery rate (%) |
| Acid 60 | 16.5 ± 2.4 | 37.3 ± 4.1 |
| Acid 90 | 35.1 ± 3.1 | 79.6 ± 6.8 |
| Acid 120 | 30.5 ± 3.0 | 69.1 ± 5.3 |
| Alkali 90 | 13.1 ± 2.2 | 29.7 ± 1.7 |
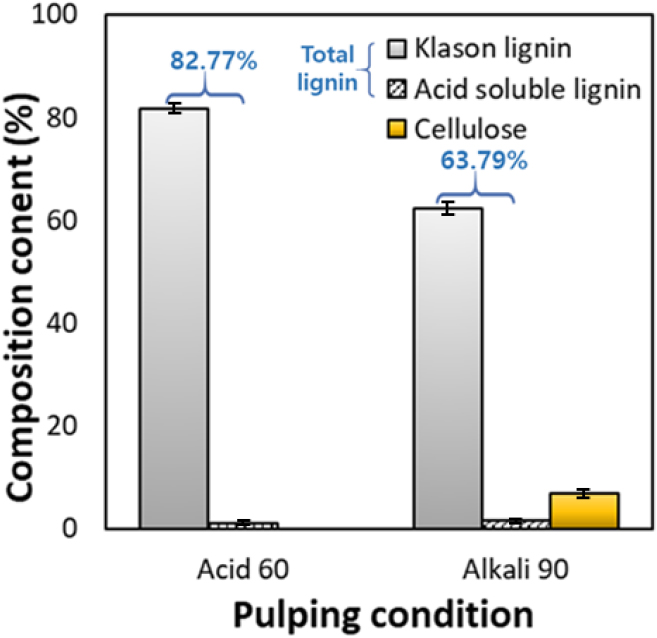
The chemical composition of lignin isolated from black liquor after pulping was analyzed, and clear differences in lignin purity and residual carbohydrate ratio were observed depending on the pulping conditions (Fig. 5). Lignin isolated under the Acid 60 condition consisted of 81.8% KL and 1.0% ASL, showing a high total lignin purity of 82.8%, and no sugar components were detected. Under acidic conditions, the acid hydrolysis of hemicellulose proceeds rapidly, and monosaccharides are released and therefore do not precipitate together with lignin [21]. In contrast, lignin recovered under the Alkali 90 condition showed 62.3% KL and 1.5% ASL, resulting in a total purity of 63.8%, which was significantly lower than that under acidic conditions. Approximately 6.8% glucose remained in the lignin, which is attributed to fine lignocellulosic particles generated during the washing stage after pulping, precipitating together with lignin and thereby reducing the relative purity of the lignin.
3.4 Micronization behavior and film properties of microfibers
Fig. 6 shows the optical microscopy (×60) images of the homogenized pulp samples prepared under the Acid 60 and Alkali 90 conditions, which were passed through the homogenizer 1, 5, 10, and 20 times, respectively. In both conditions, large agglomerated clusters were initially observed, but as the homogenization passes increased, the particles gradually became finer and more uniformly dispersed. In particular, the Acid 60 pulp exhibited a more homogeneous dispersion after 10 passes than the alkali-treated sample. Since the bark residue after tannin extraction has a smaller particle size and shorter fiber length than conventional wood pulp [12,13], microfibers could be produced solely by mechanical homogenization without additional refining or chemical pretreatment steps typically required for nanocellulose fabrication. These results indicate that organosolv pulped bark fibers are suitable for low-energy fibrillation processes.
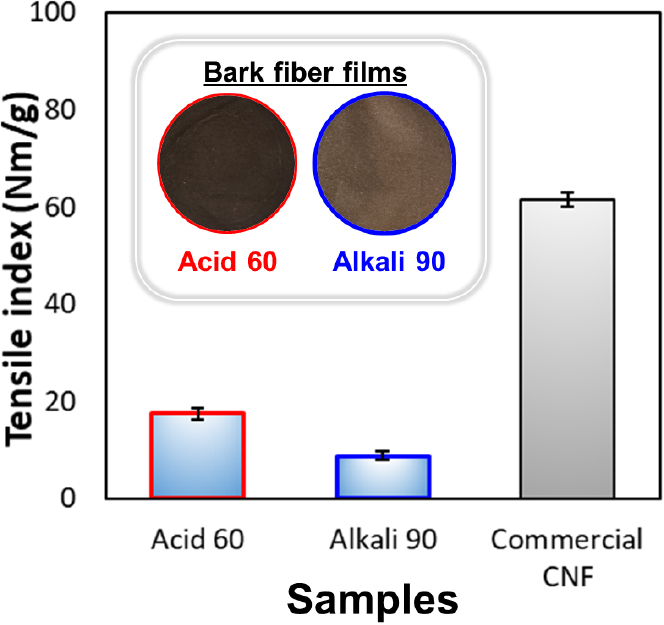
Fig. 7 shows the appearance and tensile index of films prepared from microfibers obtained by 20-pass homogenization of bark pulps produced under the Acid 60 and Alkali 90 conditions. Lignin is known to enhance ultraviolet (UV) shielding due to its intrinsic light-absorbing properties, but simultaneously reduce optical transparency [22]. Both films exhibited dark brown and opaque appearances owing to their high lignin content, and in particular, the Acid 60-based film appeared darker than the alkali-based film (KL: about 30%) due to its relatively higher lignin content (approximately 50%).
The tensile index of the bark-derived fiber films was lower than that of commercially available enzyme-pretreated CNF films (about 61.5 Nm/g). Although the fiber size difference between bark-derived fibers and commercial CNF may also influence mechanical performance, the markedly higher lignin content in bark is considered the dominant factor limiting inter-fiber hydrogen bonding and tensile strength. This is because lignin, as a nonpolar polymer, interferes with hydrogen bonding between cellulose hydroxyl groups, inducing micro-void formation within the film and thereby reducing its mechanical continuity [22,23]. Therefore, the low tensile strength of the bark-derived microfibers is attributed to the limited inter-fiber bonding caused by lignin.
The tensile indices of the Acid 60 and Alkali 90 samples were approximately 17.5 Nm/g and 8.8 Nm/g, respectively, with the acid-based film exhibiting nearly twice the strength. This is because more efficient micronzation and homogeneous dispersion occurred under the acidic condition, consistent with the microscopic observation shown in Fig. 6.
In conclusion, the relatively low tensile strength does not limit the applicability of bark-derived microfibers. The high lignin content enhances the hydrophobicity of the fiber surface, improving compatibility with polymers such as PLA or polypropylene (PP) [7,24]. Therefore, bark-based microfibers are more promising as eco-friendly bio-based fillers or compatibilizers for polymer composites rather than stand-alone materials. Compared with commercial bleached CNF, these fibers are expected to exhibit superior dispersibility and interfacial adhesion within polymer matrices without requiring chemical pretreatment or surface modification. Such characteristics suggest that lignin-containing microfibers can provide mechanical reinforcement and additional functional properties such as thermal stability and UV shielding, indicating their potential as functional bioplastic fillers. These findings demonstrate that bark residues can be utilized not only as pulp or lignin sources but also as a structural lignin–cellulose composite precursor for developing bioplastics.
4. Conclusions
In this study, the residual bark obtained after tannin extraction from oak bark was utilized as a raw material to evaluate its potential for high-value utilization through sequential valorization. Organosolv pulping was conducted using acidic (H2SO4) and alkaline (NaOH) catalysts, resulting in pulp yields of 39.4% and 45.6%, respectively, under the Acid 60 (120°C, 60 min) and Alkali 90 (120°C, 90 min) conditions. Under acidic conditions, partial hydrolysis of cellulose and hemicellulose occurred, reducing the total sugar content; whereas, under alkaline conditions, the preservation of polysaccharides was enhanced. The purity and recovery yield of lignin isolated from black liquor were 82.8% and 37.3%, respectively, under the Acid 60 condition, which were higher than those obtained under alkaline conditions.
Microfibers prepared from the Acid 60 and Alkali 90 pulps by 20-pass homogenization were successfully micronized without chemical pretreatment, exhibiting more uniform dispersion under acidic conditions. These findings suggest that bark residues are suitable precursors for producing lignin-containing composite microfibers, which can serve as eco-friendly bio-based fillers or compatibilizers for polymer composites. Overall, this study demonstrates the complete utilization of unused forest biomass by recycling tannin-extracted bark residues into pulp and lignin resources, providing fundamental data for establishing a sustainable biorefinery system.