1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Preparation of MC/PULL and MC/PULL/MMT films
2.3 Characterizations
3. Results and Discussion
3.1 Mechanical properties of as-prepared MC/PULL blend films
3.2 Morphological properties of the MC/PULL/MMT nanocomposite films
3.3 FTIR analysis of MC/PULL/MMT nanocomposite films
3.4 XRD of the MC/PULL/MMT nanocomposite films
3.5 Thermal properties of MC/PULL/MMT nanocomposite films
3.6 Contact angle analysis
3.7 Mechanical properties of MC/PULL/MMT nanocomposite films
4. Conclusions
1. Introduction
Food packaging plays a crucial role in preserving the sensory attributes and quality of food, ensuring safety, and extending shelf life. Through packaging, food produced in one part of the world can be safely transported and consumed in another region [1]. Currently, the food packaging industry relies heavily on petroleum-based synthetic plastics to provide mechanical, chemical, and microbiological protection [2]. Over recent decades, the production and use of such plastics have risen exponentially, yet their persistence in the environment poses serious ecological concerns due to their non-biodegradable nature [3]. To address this challenge, researchers have increasingly focused on biodegradable alternatives, particularly natural biopolymers such as proteins, lipids, and polysaccharides [4]. Among these, polysaccharides are considered especially promising, as they can form robust, continuous networks suitable for coatings and film fabrication [5]. However, since individual polysaccharides rarely possess all the desired attributes, blending different biopolymers with complementary properties has emerged as a strategy to develop packaging films with synergistic performance [6,7]. Achieving optimal film properties requires careful selection of constituent polymers and precise control of their mixing ratios, as blend behavior strongly depends on composition. To be effective for product protection, eco-friendly packaging films should also be sustainable, biodegradable, and have good mechanical and barrier qualities like high strength, clarity, and resistance to moisture, printability, heat-salability, and chemical inertness.
The macromolecular biopolymer pullulan (PULL) is produced by Aureobasidium pullulans through starch and sugar cultures. Structurally, it consists of a linear chain formed by polymerization via α-(1→6) and α-(1→4) glycosidic linkages, as reported by Shao et al. [8]. This unique linkage configuration imparts distinctive physical properties to PULL films. Moreover, PULL exhibits excellent film-forming ability and is transparent, odorless, tasteless, heat-sealable, biodegradable, and soluble in both hot and cold water. It also inhibits fungal growth in packaged products and provides outstanding oxygen barrier properties [9,10]. Owing to these characteristics, PULL has been widely explored as a polysaccharide that can be blended with other biopolymers to develop films, coatings, and food preservation materials [4,11].
Methyl cellulose (MC) is one of the most prevalent biopolymers in nature, derived as a modified form of cellulose and recognized for its biodegradability. Its abundant availability, low cost, and ease of processing make MC a widely utilized material for environmentally friendly applications, particularly as coatings and films [12]. In addition, MC is extensively employed across diverse industries, including food and pharmaceuticals, as well as in the production of construction and painting materials.
The demand for renewable and bio-based food packaging materials has risen with growing consumer awareness of environmental concerns and food safety [13]. Such materials, derived from natural sources such as proteins and polysaccharides, offer advantages including biodegradability, renewability, and in some cases, edibility. However, their commercial application remains limited due to relatively poor mechanical, thermal, and gas barrier properties compared to synthetic polymers [14]. To overcome these drawbacks, the incorporation of nanofillers such as clays has been widely explored, as they can significantly improve the mechanical, thermal, and barrier performance of biodegradable polymers [15,16]. Among these, montmorillonite (MMT) is the most studied layered silicate, owing to its natural abundance, high modulus, large surface area, and strong reactivity with polymer matrices [17]. When incorporated into polymers, MMT can form phase-separated, intercalated, or exfoliated structures, and such nanocomposites are commonly applied in packaging and pharmaceutical fields [18,19].
In addition, polymer blending has attracted increasing attention due to its economic advantages and the superior properties of blends compared to individual polymers [20]. Numerous studies have attempted to blend different natural polymers to tailor properties for specific applications [21,22,23], with improvements largely depending on polymer interactions and chemical structure [24].
In this study, films based on MC and PULL at different ratios were prepared, and the optimal blend ratio (MC/PULL = 60/40) was determined based on mechanical performance. MMT (1, 3, and 5 wt.%) was subsequently incorporated into the MC/PULL (60/40) matrix. The mechanical, barrier, thermal, and physicochemical properties of the resulting MC/PULL/MMT nanocomposite films were systematically investigated, with particular focus on the influence of MMT concentration.
2. Materials and Methods
2.1 Materials
Food-grade PULL (Mw ≈ 20,000 g·mol-1, purity >90%, PF-20 grade) was obtained from Hayashibara Biochemical Laboratories Inc. (Okayama, Japan). MC (Mw ≈ 400,000 g·mol-1, analytical grade, degree of substitution (DS) ≈ 1.5) was purchased from LOTTE Fine Chemical Co., Ltd. (Ulsan, Korea) and used without further purification. MMT (Powder form) was supplied by Kunimine Industries Co., Ltd. (Tokyo, Japan).
2.2 Preparation of MC/PULL and MC/PULL/MMT films
Films were prepared following the procedure reported in reference [25]. MMT powder was first dispersed in double-distilled water and magnetically stirred at 300 rpm for 1 h at room temperature. Subsequently, PULL powder was added and stirred at 600 rpm for 24 h until fully dissolved. MC powder was then introduced into the solution, followed by heating at 50°C and stirring at 600 rpm for an additional 24 h. Blend solutions with different MC/PULL weight ratios (90/10, 80/20, 70/30, 60/40, and 50/50) were prepared using the same procedure. The overall polymer concentration was maintained at 0.7 wt.%, while MMT was incorporated at 1, 3, and 5 wt.% relative to the polymer content. For nanocomposite films, MMT (0–5 wt.%) was added specifically to the MC/PULL 60/40 blend solution. The resulting solutions were cast into iron molds with smooth bottoms (20 cm × 10 cm) and dried in an oven at 40°C for 24 h. The target film weight is 56.25 g/m2. After complete drying, the films were carefully removed from the molds and stored in plastic bags for subsequent characterization.
2.3 Characterizations
The thickness of the films was measured using a digital thickness gauge (IP65, Mitutoyo, Takatsu-Ku, Japan). Tensile testing was performed with a universal testing machine (Instron 5567, Korea) in accordance with ASTM D638-96 (type II). A crosshead speed of 80 mm min-1 was applied, using a 500 kgf load cell. Rectangular specimens (2 cm × 8 cm, gauge length 20 mm) were prepared, and three replicates were tested for each condition; the average values were reported.
The surface morphology of the films was observed by field-emission scanning electron microscopy (FE-SEM; SU8220, Hitachi, Japan). Hydrophilic properties were evaluated through static water contact angle measurements using a goniometer (Dino-Lite, AM703MZT, NARAE PLUS Co., Ltd., Seoul, Korea). Structural characterization was carried out using X-ray diffraction (XRD; D/Max-2500, Rigaku, Tokyo, Japan) to investigate crystalline features, and Fourier-transform infrared spectroscopy (FTIR; Frontier, PerkinElmer, Waltham, MA, USA) to identify functional group interactions. Thermal behavior and stability were analyzed with thermogravimetric analysis (TGA; Q-50, TA Instruments, Seoul, Republic of Korea).
3. Results and Discussion
3.1 Mechanical properties of as-prepared MC/PULL blend films
Table 1 presents the tensile properties of pristine MC and MC/PULL blend films prepared at different ratios. Compared with pure MC, which exhibited a breaking stress of 2.1 ± 0.1 MPa and a breaking strain of 9.2 ± 0.5%, all MC/PULL blends demonstrated noticeable improvements. The incorporation of PULL into MC enhanced both tensile strength and ductility, suggesting that the presence of PULL chains promotes flexibility while simultaneously reinforcing the MC matrix through hydrogen bonding interactions. Adding PULL to MC increases hydrogen bonding primarily due to synergistic intermolecular interactions between the hydroxyl-rich PULL molecules and the MC chains, which form a denser and more compact network than in the individual polymers. Among the compositions tested, the MC/PULL 60/40 blend exhibited the highest performance, with a breaking stress of 11.7 ± 0.6 MPa and strain of 25.6 ± 1.3%. This represents an approximate 179% increase in elongation at break compared with pristine MC. The synergistic effect observed at this ratio indicates that the balance between MC and PULL enables effective load transfer, improved chain mobility, and the development of a more ductile and tougher material.
Table 1.
Mechanical properties of MC and MC/PULL composites at different ratios
However, when the PULL content increased further to 50/50, the tensile properties decreased to 10.1 ± 0.5 MPa and 24.1 ± 1.2%, respectively. This reduction can be attributed molecular weight difference between MC and PULL at higher concentrations, which disrupts miscibility, reduces intermolecular hydrogen bonding, and ultimately lowers the reinforcing efficiency. As a result, failure occurs at lower stress levels despite the relatively high ductility of the film. High tensile qualities are frequently the main objective when optimizing a film, particularly for uses like packaging where handling and tearing resistance is essential.
3.2 Morphological properties of the MC/PULL/MMT nanocomposite films
FE-SEM analysis revealed distinct morphological changes in the MC/PULL/MMT films as a function of MMT content (Fig. 1). The neat MC/PULL film exhibited a smooth and uniform surface, reflecting a compact structure and strong polymer–polymer miscibility. When MMT was incorporated, partial disruption of surface uniformity occurred because the silicate layers interacted with the polymer chains. At 1 wt.% MMT, this interaction was mild, producing only slight irregularities. As the concentration increased to 3 wt.%, the stronger interaction between clay platelets and the polymer matrix promoted microstructural roughness. At 5 wt.% MMT, however, excess clay particles aggregated due to limited polymer–filler compatibility, leading to the formation of distinct surface clusters. These structural changes directly affect the film properties: the progressive loss of surface uniformity compromises the compact morphology of the matrix.
3.3 FTIR analysis of MC/PULL/MMT nanocomposite films
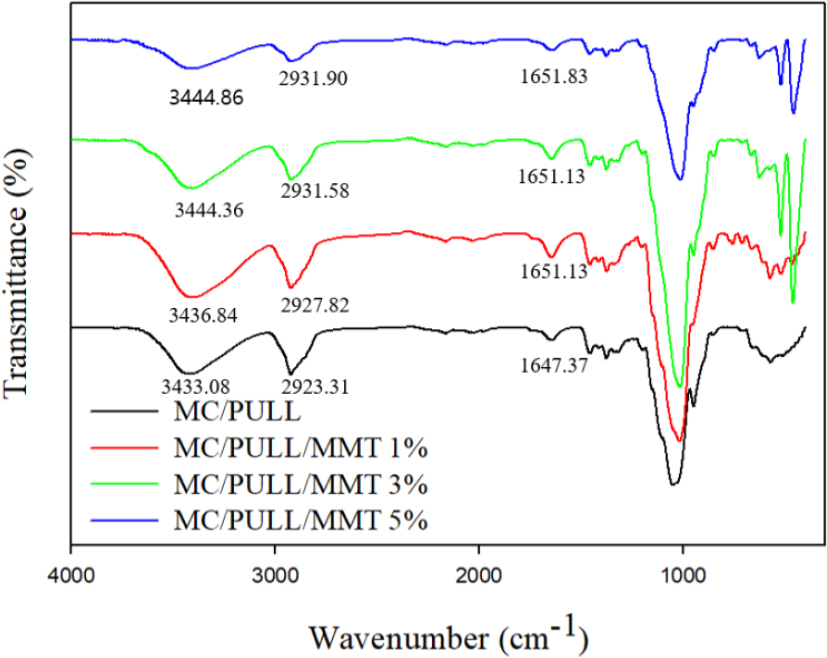
FTIR spectroscopy was applied to elucidate the structural features of MC/PULL blend films and their interactions with MMT in the wavenumber range of 400–4,000 cm-1 (Fig. 2). The neat MC/PULL film exhibited characteristic absorption bands corresponding to the functional groups of cellulose and PULL: O–H stretching at ~3,433 cm-1, C–H stretching at ~2,923 cm-1. Additional peaks included δ(C–H) vibrations at ~1,374 and 1,455 cm-1, a C–O–C stretching mode at ~1,048 cm-1, and OCH3 vibrations at ~946 cm-1[12,26,27]. These assignments confirm the presence of the principal polysaccharide functional groups. Upon the incorporation of MMT, distinct spectral modifications were observed. The O–H stretching band shifted to higher wavenumbers (3,433 → 3,444 cm-1), which indicates the formation of new hydrogen bonds between hydroxyl groups of MC/PULL and silicate layers of MMT [12,26]. Similarly, the C–H (~2,923 cm-1) and C=O (~1,642 cm-1) bands showed slight upshifts, further supporting polymer–filler interactions. With increasing MMT content (1–5 wt.%), the intensity of the O–H and C–H bands progressively decreased, suggesting that free hydroxyl groups were consumed during hydrogen bonding and that chain mobility was increasingly restricted. These changes provide molecular-level evidence of strong polymer–clay interactions. The reduction in intensity of the carbonyl band at ~1,642 cm-1 with higher MMT loading reflects the reduced availability of free polar groups, which is consistent with the establishment of an interconnected hydrogen-bonding network within the nanocomposite. Such behavior has also been reported in chitosan/MMT and carboxymethyl cellulose/MMT systems [28], where the silicate platelets were shown to act as hydrogen-bond acceptors, thereby strengthening polymer–filler compatibility. The significance of these spectral features extends beyond chemical interaction. The formation of stronger hydrogen-bonding networks explains the improved thermal stability of the films at moderate filler contents and supports the exfoliated morphology identified by XRD. Moreover, the reduction in free hydroxyl groups correlates with the increased water contact angle, as fewer hydrophilic sites are available for water interaction. The FTIR results confirm that MMT is not merely physically dispersed but chemically interacts with MC/PULL chains, thereby altering the molecular organization and providing a mechanistic basis for the changes in structural, thermal, and barrier properties observed in subsequent analyses [26,27,28].
3.4 XRD of the MC/PULL/MMT nanocomposite films
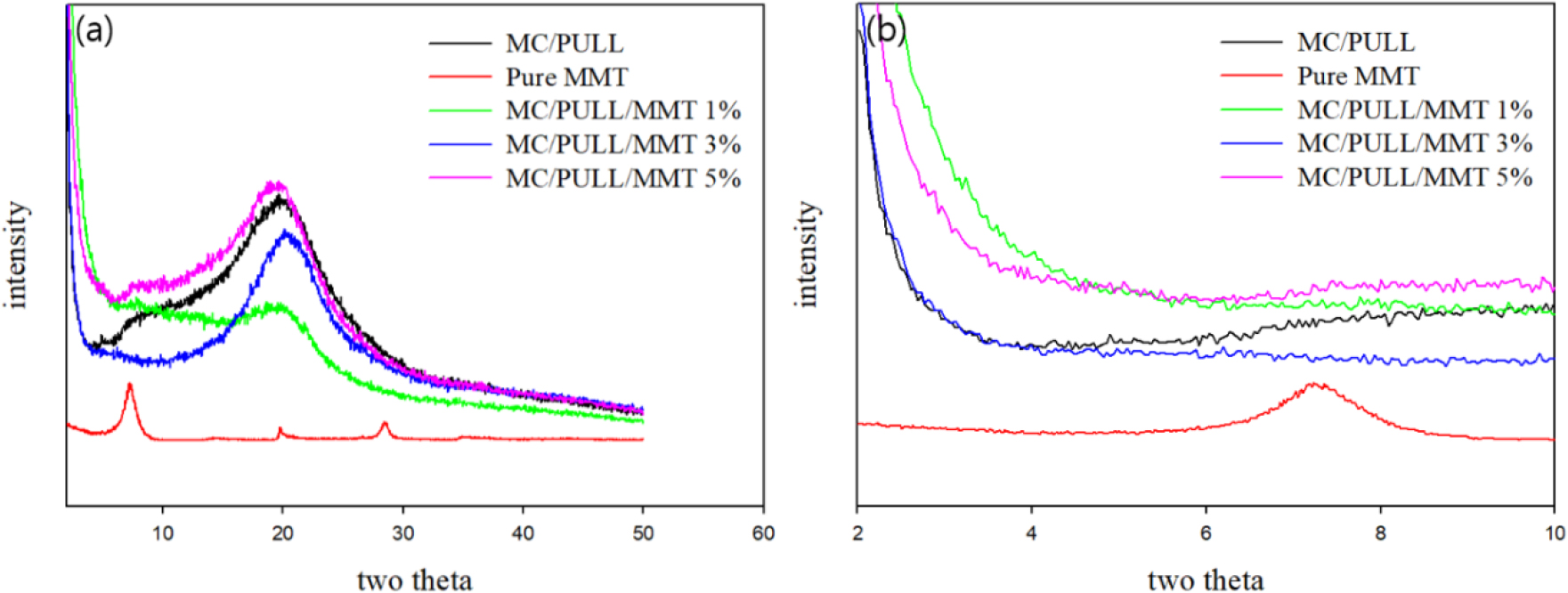
Fig. 3 shows the XRD patterns of neat MC/PULL, pure MMT powder, and MC/PULL/MMT nanocomposites with different MMT contents in the 2θ range of 2–50°. The pure MMT exhibited three characteristic peaks: a broad reflection at 2θ ≈ 6.7° (d-spacing ≈ 1.3 nm) corresponding to the interlayer distance of silicate platelets, and two sharp peaks at 2θ ≈ 19.8° and 29.9°. In contrast, no diffraction peak corresponding to the basal spacing of MMT (~1.3 nm) was detected in any of the MC/PULL/MMT nanocomposites, indicating partial exfoliation and random dispersion of MMT layers within the polymer matrix. This suggests a relatively high degree of miscibility between the filler and the MC/PULL blend [12]. The neat MC/PULL film displayed a broad halo centered at 2θ = 5–27°, which is attributed to the amorphous intermolecular packing of polysaccharide chains. Upon the addition of MMT, the intensity of this amorphous peak gradually decreased with increasing filler content, indicating a reduction in the overall crystallinity of the matrix. This decrease is likely due to heterogeneous nucleation caused by the dispersed MMT platelets, which disrupts chain ordering and reduces crystalline perfection. A similar decrease in crystallinity upon the incorporation of clay platelets has been reported for Nylon 66/MMT nanocomposites, where the crystallinity was reduced by approximately 10% even at 1 wt.% MMT loading [29]. Therefore, the XRD analysis confirms that MC/PULL/MMT nanocomposites possess an exfoliated morphology across the studied concentration range (1–5 wt.%). This morphology provides evidence of strong polymer–filler miscibility and explains the changes in structural ordering that contribute to the thermal and barrier performance discussed in subsequent sections.
3.5 Thermal properties of MC/PULL/MMT nanocomposite films
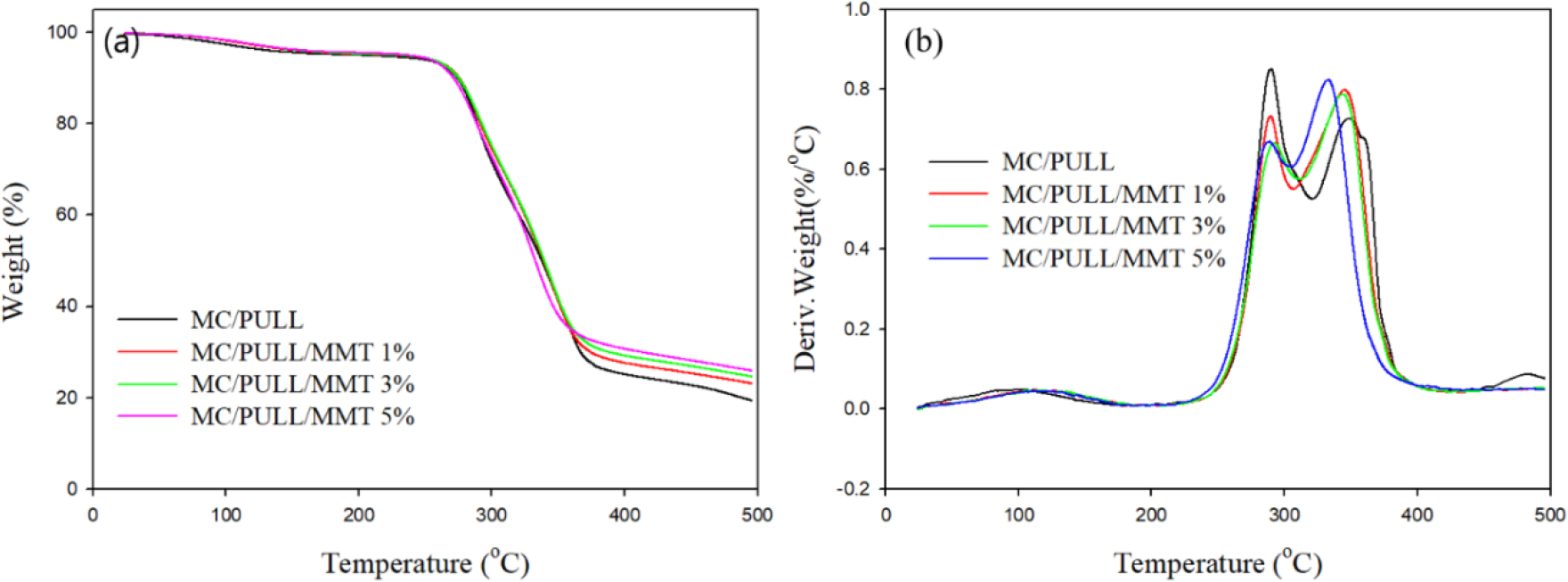
The thermal parameters obtained from TGA and DTG analysis are summarized in Fig. 4 and Table 2. Neat MC/PULL exhibited Tonset = 235.9°C, T10% = 273.2°C, and T50% = 337.8°C, with Tmax peaks at 290.2°C and 348.6°C, and a char yield of 19.8% at 500°C. Upon incorporation of MMT, several notable changes occurred. Tonset and T10% remained nearly unchanged at 1–3 wt.% MMT but slightly decreased at 5 wt.% (Tonset = 234.3°C, T10% = 270.6°C), suggesting that higher filler content facilitates earlier chain scission. In contrast, T50% increased from 337.8°C (MC/PULL) to 339.2°C at 3 wt.% MMT, indicating delayed mass loss and improved mid-range thermal stability. However, at 5 wt.% MMT, T50% decreased to 332.3°C, reflecting the negative influence of excessive filler loading. Tmax values generally decreased with MMT incorporation, especially at higher contents (332.5°C at 5 wt.% vs. 348.6°C for MC/PULL), suggesting altered degradation pathways. Char yield increased progressively with MMT content, from 19.8% (MC/PULL) to 26.6% (5 wt.% MMT), due to the inorganic and non-combustible nature of MMT. This trend is consistent with previous reports on chitosan/MMT nanocomposites, where silicate platelets acted as thermal insulators and enhanced char formation [7,28].
Overall, the data suggest a dual role of MMT in thermal degradation. At low-to-moderate loadings (1–3 wt.%), MMT provides a barrier effect that retards weight loss and improves char yield, thereby enhancing thermal stability. At higher loadings (5 wt.%), particle agglomeration reduces homogeneity, lowers Tonset and T50%, and compromises overall stability despite higher char residue.
Table 2.
Thermal properties of the prepared MC/PULL/MMT nanocomposite films (MC/PULL ratio: 60/40)
3.6 Contact angle analysis
The water contact angle was measured to assess the surface hydrophilicity of MC/PULL and MC/PULL/MMT nanocomposite films, and the results are summarized in Table 3. The neat MC/PULL film exhibited a contact angle of 61.4 ± 1.0°, reflecting the strong hydrophilic character imparted by abundant hydroxyl groups [30]. With increasing MMT content (0–5 wt.%), the contact angle gradually increased from 61.4° to 64.4°. At 1 wt.% MMT, the contact angle remained nearly unchanged (61.5 ± 1.0°), whereas higher MMT loadings caused a measurable increase: 63.3 ± 1.5° at 3 wt.% and 64.4 ± 1.5° at 5 wt.%. This increase is attributed to the presence of silicate layers, which hinder the spreading of water droplets on the surface, thereby reducing overall hydrophilicity. These findings are consistent with previous studies. Cellulose films incorporating MMT have shown higher contact angles compared to cellulose alone [9], and carboxymethyl cellulose films demonstrated an increase from 48.6° to 67.6° upon the addition of 10 wt.% MMT [31]. Such results confirm that the introduction of layered silicates decreases the availability of free hydroxyl sites on the surface and enhances surface hydrophobicity. Therefore, the incorporation of MMT modulates the wettability of MC/PULL films, with moderate increases in contact angle linked to nanoparticle-induced surface modification. This trend has direct implications for barrier properties, as reduced hydrophilicity is favorable for moisture resistance in packaging applications.
Table 3.
Contact angle and thickness of the MC/PULL/MMT nanocomposite films (MC/PULL ratio: 60/40)
| Film sample | Contact angle (°) |
| MC/PULL | 61.4 ± 1.0 |
| MC/PULL/MMT 1% | 61.5 ± 1.0 |
| MC/PULL/MMT 3% | 63.3 ± 1.5 |
| MC/PULL/MMT 5% | 64.4 ± 1.5 |
3.7 Mechanical properties of MC/PULL/MMT nanocomposite films
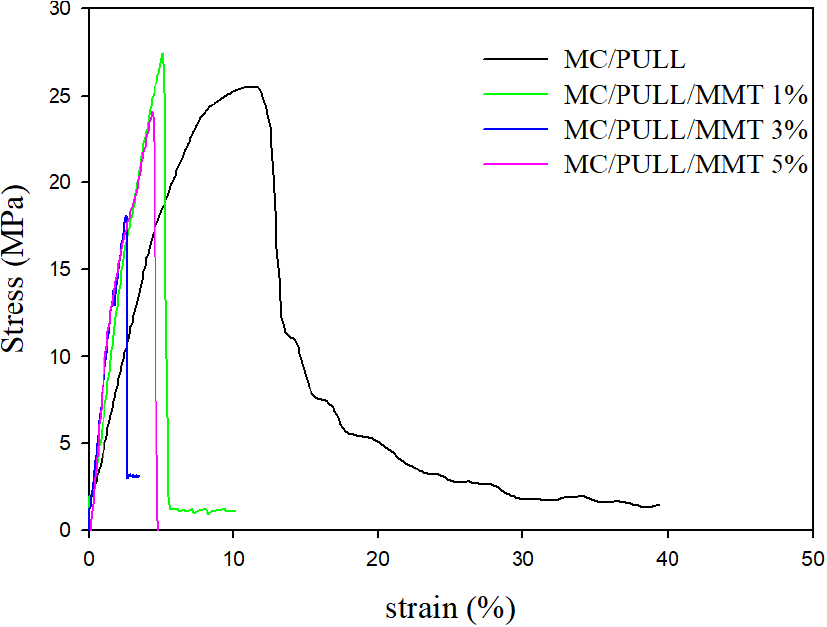
One of the most crucial aspects of food packaging materials is their mechanical strength, and films intended for use in packaging should possess this quality. Fig. 5 shows the effect of MMT clay loading on the mechanical characteristics of MC/PULL films. It was seen from Table 4 that stress at the break of MC/PULL increased after nanofiller loading of 1 wt.% however, when the filler concentration surpasses 1 wt.% the circumstances change. This is explained by the nanoclay particles’ micro-aggregation at higher MMT filler contents (>1% wt). The reason for this is that, the MMT particles have a tendency to bind together to form microsized particles that could function as defects and reduce the tensile strength. A decrease in the breaking strain values was observed after adding MMT in MC/PULL film. For example, the breaking strain reduced from 11.41% to 2.52% with the addition of 3 wt.% MMT into MC/PULL film matrix. The situation can be explained by MMT’s reinforcement effect. The mobility of the polymer chains gets restricted through MMT layers. Consequently, the elongation of the film sample is decreased when MMT is present in the film matrix.
4. Conclusions
In this study, MC/PULL/MMT nanocomposite films were successfully fabricated by solution casting and evaluated for their potential use in food packaging. Mechanical testing of different MC/PULL ratios identified 60/40 as the optimal blend composition, providing the highest tensile strength and strain. Incorporation of MMT further modified the structural and functional properties of the films. TGA revealed that moderate MMT loadings (1–3 wt.%) enhanced thermal stability, as reflected in increased T50% values., whereas excessive loading (5 wt.%) led to reduced stability reflecting the negative influence of excessive filler loading. Contact angle analysis indicated that hydrophobicity increased slightly with rising MMT content (61.4° to 64.4°), consistent with the presence of silicate layers at the surface. These results demonstrate that MC/PULL/MMT nanocomposite films combine improved mechanical performance with tunable thermal and surface properties, making them probable candidates for sustainable food packaging applications. Future studies could focus on tailoring filler dispersion strategies and exploring antimicrobial or barrier enhancements to further expand their practical utility.