1. Introduction
2. Experimental
2.1 Materials
2.2 Preparation of cellulose-lignin composite films
2.3 Characterization
3. Results and Discussion
3.1 Preparation and characterization of cellulose-lignin composite films
3.2 Strength properties of the composite film
3.3 Surface morphology analysis
3.4 FTIR and UV shielding and thermal stability properties of the composite film
3.5 Swelling properties and hydrophobic of the composite film
3.6 Rheological performance analysis
3.7 Biodegradability
4. Conclusion
1. Introduction
Cellulose is the most abundant renewable resource on Earth [1,2]. The mechanical properties, thermal properties, biocompatible degradability, and sustainability of cellulose have aroused the interest of researchers [3,4]. Cellulose has been used in various fields such as batteries [5,6], biomedicine [7,8], makeup products [9,10], etc. The widespread use of cellulose films has attracted the attention of researchers. Cellulose can be used for the preparation of flexible and transparent films, which are widely used in packaging, medical, and electronic applications [11]. However, cellulose films still have the disadvantages of low mechanical properties, and water instability, and the methods used to solve these problems are usually complex and costly [12].
Lignin is an inexpensive and renewable polymer. The key role of lignin in the structures and functions of the cell wall is to maintain the strength and stiffness of the cell wall, acting as a binder [13,14]. In natural wood fiber materials, lignin and carbohydrates are chemically bonded to each other [15]. Most of the lignin in black liquor was burned as fuel, and only 2% of lignin was used commercially as a dispersant and binder [16]. Lignin contains abundant hydroxyl and ether groups that can contribute to hydrogen bonding, which can increase intermolecular interactions [17], break the chemical connections of lignin and expand the hybrid use of lignin together with other natural and compatible polysaccharides (e.g., cellulose) [18]. Lignin can be used as a reinforcing agent or as a filler in combination with cellulose, which can be used to improve the mechanical properties and thermal stability of film materials.
In recent years, multi-component lignocellulose composite films have received great attention. Sadeghifar et al. [11] prepared cellulose-lignin film from the surface azide of microcrystalline cellulose and propargylated lignin by click reaction, and the mechanical properties of the film were 90 MPa. Liu [19] prepared a high-strength composite film by carboxymethylated cellulose nanofiber (CNF) and then mixed it thoroughly with pH-adjusted lignin and homogenized it with a homogenizer. Ji et al. [20] prepared a composite film by calendering ramie fiber, lignin, and chitosan after dissolution, and the mechanical properties of the film were 57.8 MPa. High-density and high-strength films were prepared by adding lignin as a small molecule filler to effectively improve the internal structure of the films [15,21,22]. Most of the studies prepared composites by modifying cellulose and lignin. Therefore, it is important to prepare cellulose-lignin composite films with good mechanical properties and thermal stability for various applications using a simple process.
Lignin, as a natural binder, holds together cellulose fibers to form a strong and tough wood matrix as the tree grows [23]. Inspired by the structure of trees, we propose the simple green method of codissolution-precipitation. More specifically, Eucalyptus dissolving pulp was codissolved with kraft lignin in 1-allyl-3-methylimidazolium chloride (AmimCl) and the cellulose-lignin composite film was prepared by precipitating in water. Hydrogen bonds can be formed between lignin and cellulose after wet film drying, which increases the mechanical and thermal stability of the composite film. Moreover, the composite film has other good properties such as UV shielding and water resistance, due to the incorporation of lignin.
2. Experimental
2.1 Materials
The ionic liquid 1-allyl-3-methylimidazolium chloride (AmimCl) was obtained from Aladdin. Eucalyptus dissolving pulp with a degree of polymerization of 646 and α-cellulose content of 97.58% was supplied by a paper mill in Shandong, China. Kraft lignin (Mw = 505.01 g/mol, density = 1.3 g/mL) was purchased from Aladdin. All materials were used directly without further purification, and the water used in the experiments was deionized water.
2.2 Preparation of cellulose-lignin composite films
Cellulose was dissolved in the ionic liquid 1-butyl-3-methylimidazolium chloride (AmimCl) at a concentration of 2 wt %. Mixtures of lignin and cellulose at various mass ratios (lignin:cellulose = 0.025:1, 0.05:1, 0.125:1, 0.25:1, 0.5:1, and 1:1) were added to the AmimCl solution. The resulting suspension was stirred and heated under vacuum at 80°C until completely dissolved and homogeneous, then degassed to remove bubbles. The solution was cast into a polytetrafluoroethylene (PTFE) mold and immersed in a deionized water coagulation bath at room temperature to form the cellulose-lignin composite membrane. Finally, the membrane was thoroughly washed with deionized water. To verify the complete removal of AmimCl, silver nitrate solution was added to the filtrate until no further precipitation formed. After drying, the cellulose-lignin composite films were obtained. The water in the coagulation bath was removed by rotary evaporation to regenerate AmimCl. The cellulose-lignin composite films with different lignin contents were named as C-L1 (0.025:1), C-L2 (0.05:1), C-L3 (0.125:1), C-L4 (0.25:1), C-L5 (0.5:1), and C-L6 (1:1).
2.3 Characterization
An electronic universal testing machine (UTM2203, SUNS, China) with a test speed of 10 mm/min was used to test the tensile strength and elongation at break values at room temperature. The specimen size was dumbbell type 4 mm × 50 mm, adopted the international standard ASTM D882 for the test.
UV-visible spectrophotometer (TU-1950, PERSEE, China) was tested for UV-visible transmission spectra from 200–800 nm wavelength range.
A contact angle meter (CA200, HOKUTO, China) was used to determine the contact angle at room temperature. A drop of about 2 µL of distilled water falls onto the surface of the film, and the image was taken within 5 seconds of the moment the droplet made contact with the film surface.
The swelling properties of the film were measured using gravimetric analysis. The film was cut to 30 mm × 10 mm and dried in an oven until the weight was unchanged. After soaking the film for 24 hours in deionized water, remove water from the surface of the sample. The water absorption rate (A) is calculated by Eq. (1):
where, M1 is the weight of the sample after drying in the oven, M2 is the weight of the sample immersed under water for 24 h. Each sample was measured three times.
A Fourier transform micro-infrared spectrometer (Nicolet iS50, ThermoFisher, USA) measured the FTIR spectra of the samples. During the test, a total of 64 cumulative scans were performed in transmission mode at a resolution of 1 cm-1 across the frequency range of 4,000–500 cm-1.
Scanning electron microscopy (SEM) observations of cellulose-lignin composite films were performed on a scanning electron microscope (Apreo S HiVac, Thermo Scientific, USA). Samples were coated with gold via sputtering on both surfaces and sides, followed by observation at an acceleration voltage of 10 kV.
Atomic force microscopy (AFM) was used to test the surface morphology of the sample on an atomic force microscope (Multimode8, BRUKER, USA). Resolution: X, Y-axis precision 1 nm, Z-axis precision 0.2 nm.
Thermal stability was evaluated using a thermogravimetric analyzer (STA 6000, PerkinElmer, China) with a temperature range of 40–800°C, protected by nitrogen gas with 10 °C/min heating rate.
Steady-state and dynamic rheological measurements were performed on the cellulose/ionic liquid solution to characterize the properties of the cellulose solution. Measured using a rotational rheometer (AR2000ex, TA Instruments, USA) with 40 mm parallel plates.
In an experiment on soil degradation, the composite film was cut into a 27 × 21 mm size and buried in the soil of the school garden at a depth of 5–10 cm for the degradation experiment.
The crystal structure of the cellulose-lignin composite film was analyzed by X'Pert3 Powder. During the test, the scanning rate was 1 °/min, with a voltage of 40 kV and a current of 40 mA. The scattering angle range was 5–55°, and the test was conducted under the Cu K radiation with a wavelength of 0.154 nm.
3. Results and Discussion
3.1 Preparation and characterization of cellulose-lignin composite films
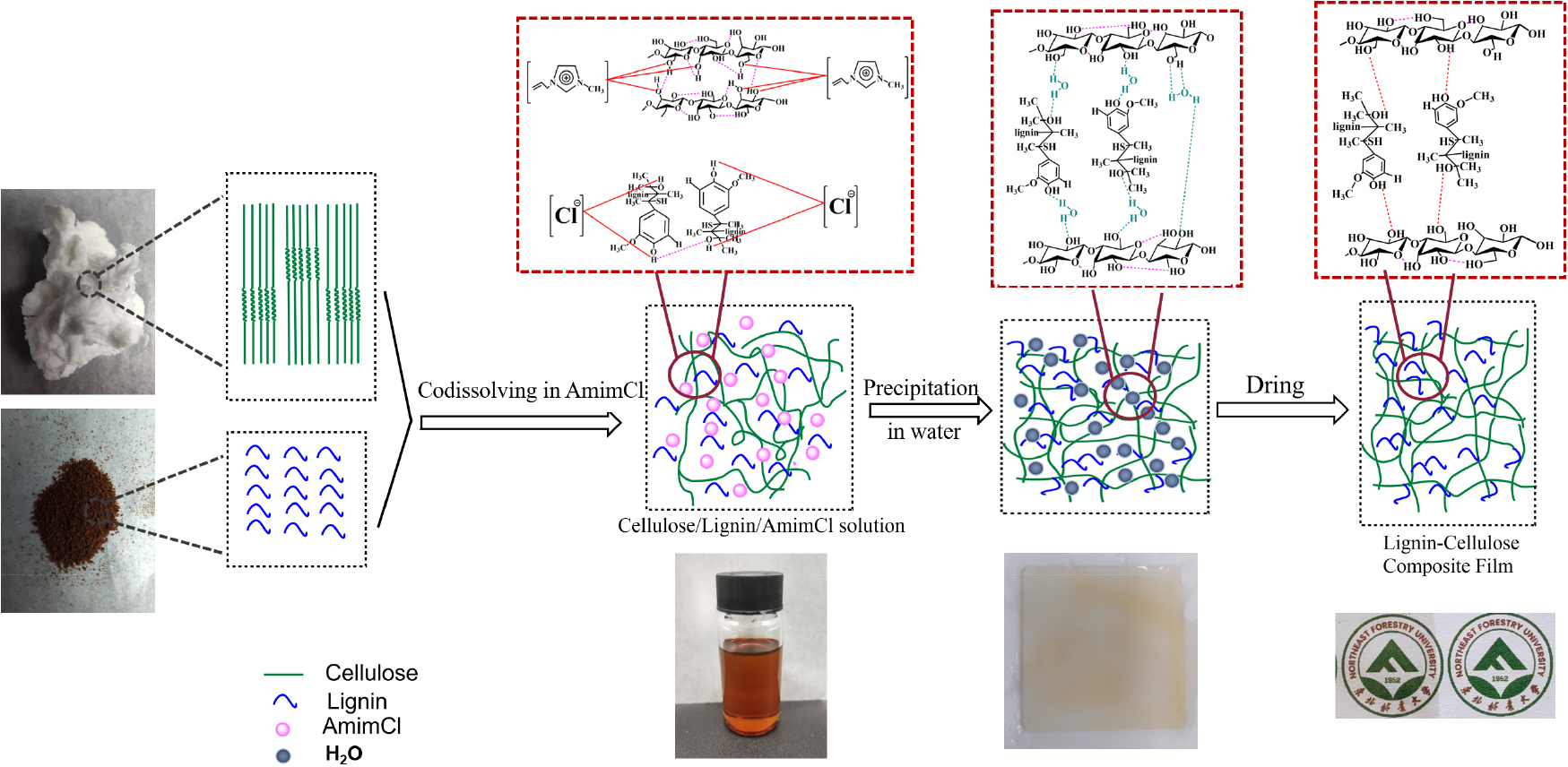
Fig. 1 shows the preparation process of cellulose-lignin composite film, the thickness of the composite film was obtained to be 20 µm by the codissolution and regeneration method. During the codissolution process, eucalyptus dissolving pulp is dissolved by the cations of AmimCl, forming new hydrogen bonds between cellulose and AmimCl by disrupting inter- and Intramolecular cellulose hydrogen bonds [24], and forming new hydrogen bonding connections [25]. Kraft lignin is dissolved by the anions of AmimCl via breaking the hydrogen bonds between lignin molecules [26]. During codissolution, cellulose and lignin are evenly dispersed in the AmimCl and form a homogeneous solution. During the precipitation process, the AmimCl is displaced by water, and water molecules form “water bridges” with cellulose and lignin.
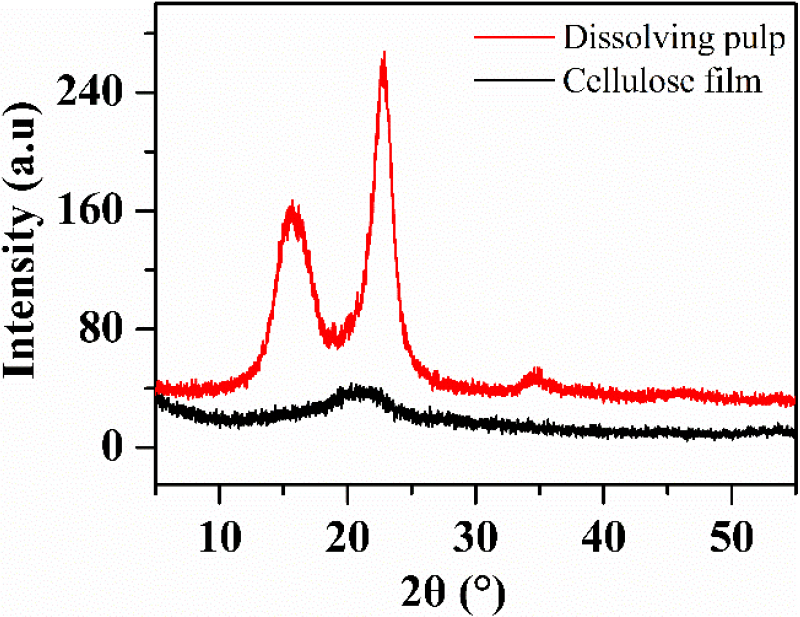
During codissolution-precipitation, the crystalline structure of cellulose changes from cellulose I to cellulose II. As shown in Fig. 2, cellulose exhibited four diffraction peaks at 2θ = 14.8°, 16.8°, 22.6°, and 34.8°, corresponding to the (1-10), (110), (002), and (040) crystal planes of cellulose I. The prepared regenerated cellulose film exhibited two diffraction peaks at 2θ = 20° and 2θ = 21.5°, corresponding to the crystalline peaks of cellulose II. This observation suggests that the cellulose crystal structure underwent a transformation during the dissolution and regeneration process, changing from cellulose I to cellulose II, while the lignin structure remains unchanged [27]. After drying, the water molecules are removed, and the hydroxyl groups between cellulose and lignin are connected by hydrogen bonds [28]. The picture covered can be clearly observed through the obtained composite film.
3.2 Strength properties of the composite film
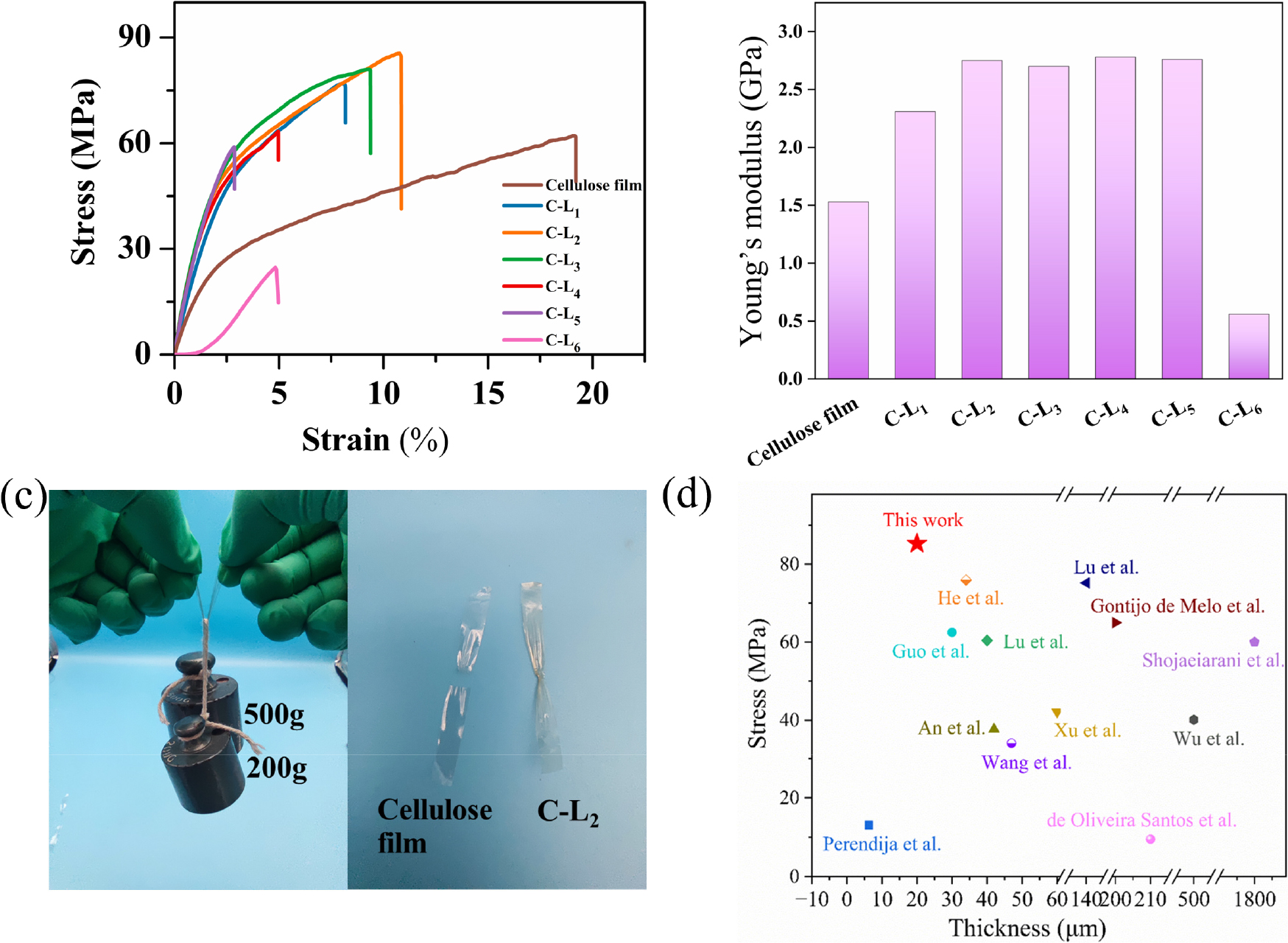
Fig. 3a and b show the composite film’s mechanical properties. Compared to pure cellulose film, the elongation at break of the composite film was reduced by the addition of lignin, this is because the slip of cellulose fragments is limited by the rigidity of lignin [29]. The stress and Young’s modulus in the composite films changed with lignin dosage. The stress and Young’s modulus of C-L2 were 85.33 MPa and 29.30 MPa (Fig. 3a and b), the stress increase by 37.45% compared with pure cellulose film, which indicated that the incorporation of an appropriate amount of lignin changed the hydrogen bonding interaction. The hydrogen bonds existing between lignin and cellulose improved the adhesion between them, thereby increasing the stress of the fibers [30]. The high mechanical properties of C-L2 allow it can hold up to 700 grams of weight, but pure cellulose film breaks directly and cannot hold 700 g (Fig. 3c). However, lignin aggregation will occur if too much lignin is added [31], the formation of hydrogen bonds between cellulose and lignin will be affected [32,33], and the stain and stress of the composite films decreased [34].
In this study, high-strength composite film was prepared by codissolution and precipitation of eucalyptus dissolving pulp and kraft lignin using AmimCl and water. In contrast to most cellulose-based composite films reported in the literature, this study did not require pre-treatment of the raw material or the addition of other substances in dissolution (Table S1). The method was simple, green, and the obtained composite films have higher mechanical strength than most of the cellulose-based composite films reported in the literature (Fig. 3d).
3.3 Surface morphology analysis
Fig. 4a shows the SEM images of eucalyptus dissolving pulp, pure cellulose film, and composite films. For eucalyptus dissolving pulp, the fiber structure was complete, and a small number of fines existed on the flat surface of the fibers (Fig. 4a). The surface of the pure cellulose film without adding lignin was smooth. With the increase of lignin dosage, the surface of the composite films became rougher, and the more lignin incorporation, the more bumps on the surface of the composite films and the more irregular the surface (Fig. 4c). Too much lignin incorporation will increase lignin aggregation, which makes lignin agglomerate into large particles on the surface of the composite films and affect the bonding of cellulose and lignin. In addition, the dissolution behavior of cellulose and lignin in ionic liquids was observed. As shown in Fig. 5a, cellulose and lignin remained undissolved and retained their original morphology after addition to the ionic liquid. After 3 hours (Fig. 5b), both cellulose and lignin dissolved completely, forming a uniform solution. The incorporation of the appropriate amount of lignin makes it evenly dispersed in the solution and closely bonded with cellulose, which is conducive to improving the mechanical properties of the composite films.
The pictures in Fig. 4b show the cross-section of the film, the cellulose film has obvious gaps in the cross-section and therefore has a lower tensile strength. C-L2 cross-section has a dense laminar structure, and the addition of lignin makes the structure of the cellulose membrane denser. Cellulose and lignin are connected through hydrogen bonding to form a network structure of lignin connected to the cellulose, which results in a denser inter-gap structure and an increase in the tensile strength of the film.
3.4 FTIR and UV shielding and thermal stability properties of the composite film
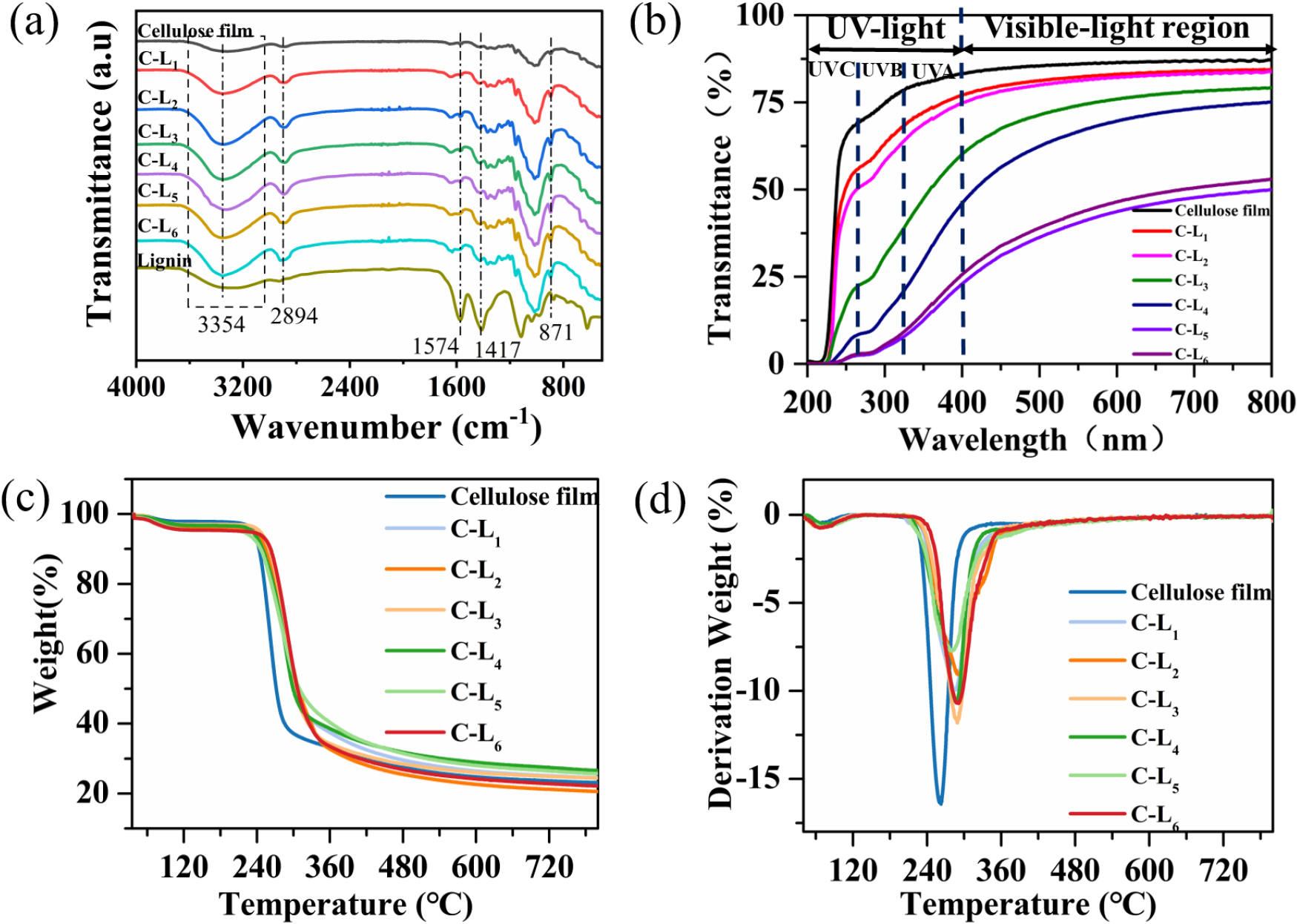
Fig. 6a, the bands appearing at 1,574 cm-1 and 1,417 cm-1 correspond to the aromatic ring skeleton of lignin and the C–H skeleton vibrations on the aromatic ring [35,36,37], the C–H stretching in aromatic and aliphatic groups at 2,894 cm-1[38,39], the out-of-plane C–H of the p-hydroxyphenyl unit at 871 cm-1[40] demonstrated the presence of lignin in the composite films. The broad band centered at 3,354 cm-1 was the O–H stretching vibrational peak, the change in the peak indicated the change in hydrogen bonding, the increase in the peak proved the increase in hydrogen bonding within the film, suggesting that lignin and cellulose were connected through hydrogen bonding [28]. During dissolution, inter- and intramolecular hydrogen bonds between cellulose and lignin are dissolved, and during rapid solidification, hydrogen bond connections are formed between cellulose and lignin.
Fig. 6b shows the ultraviolet transmittance of the composite films. From Fig. 6b, the highest transmittance of pure cellulose film in the visible and UV regions indicated that the cellulose film had good transparency and did not have UV shielding performance. The transmittance of the cellulose-lignin composite films in UV light regions decreased with the increase of lignin dosage due to the influence of lignin chromophores, which proved that the composite film had UV shielding performance. The incorporation of lignin shielded a small amount of UVA (400–320 nm) and UVB (320–275 nm), and most of UVC (275–200 nm) [41]. The UV shielding performances are attributed to the phenyl propane structure and phenolic hydroxyl groups in the lignin molecules, which have UV shielding capability [42,43].
The thermal performance of the composite films was analyzed using TGA. Fig. 6c shows the degradation trend presented by the composite films. The degradation of the composite films containing lignin below 100°C is the volatilization of free water in the film and the removal of unstable substances in the film. The slow degradation from 100 to 230°C due to the thermal stability of phenolic hydroxyl and carbonyl groups in lignin [44]. The rapid degradation occurs at 230–290°C due to the broken C–C bonds in lignin and the glucosidic bonds in cellulose [45]. The minimal degradation below 290°C is due to the presence of various aromatic groups of lignin and various ethers and C–C bonds [33]. Fig. 6d shows that the range of 240–300°C has a large peak, corresponding to the temperature at the maximum weight-less rate (Tmax). The Tmax of the composite films increases, among them, the Tmax of C-L2 is the highest, indicating C-L2 has the best thermal stability. Adding the right amount of lignin can improve the thermal stability of the composite films, mainly because of the stable structure of the aromatic units in lignin and the existence of hydrogen bonds between cellulose and lignin [46].
3.5 Swelling properties and hydrophobic of the composite film
As Fig. 7a shows, the contact angle of C-L2 (65.9°) was higher than that of cellulose film (41.2°), indicating that is suggested that the addition of lignin to the hydrophobic properties of the cellulose film, which was due to the hydrophobic functional group of lignin improving the water resistance of the cellulose film.
The swelling properties of the films were measured by water absorption experiments. Fig. 7b shows the films after 0 and 1 day soaking in deionized water. There was no significant change in the film after soaking, and the lignin in the composite film did not dissolve or precipitate. The water absorption is shown in Table 1. Water absorption primarily depends on the material’s overall porous structure and internal capillary forces, hydrophobic surfaces can only slow down the initial rate at which water comes into contact with the surface, but cannot slow down the water absorption process driven by capillary effects within the internal pores after prolonged contact [47]. A finer pore structure can increase the permeability of the solvent, and it can be judged that the lignin-containing composite film has a finer pore structure. The hydrophilic groups of lignin were predominantly encapsulated within the composite film, enhancing the binding of hydrophilic groups with water molecules and microporous permeation within the composite film, thereby increasing its swelling properties. The cross-section of the film at different soaking times was further observed by optical microscope (Fig. 7c). The thickness of the composite film did not change significantly after 1 day of soaking because the lignin was attached to the fibers through hydrogen bonding, which reduced the exposure of the hydrogen bonds of the film, enhanced the water-repellent properties of the film surface and improved its internal expansion characteristics. The cross sections at 1 and 120 days of immersion clearly showed that the pure cellulose film swelled inconsistently under prolonged immersion, and that the lignin-containing composite film had higher water resistance than the pure cellulose film.
Table 1.
Contact angle and water absorption of the films
| Contact angle (°) | Water absorption (%) | |
| Cellulose film | 41.2 | 86.30 ± 0.2 |
| C-L2 | 64.9 | 111.84 ± 0.4 |
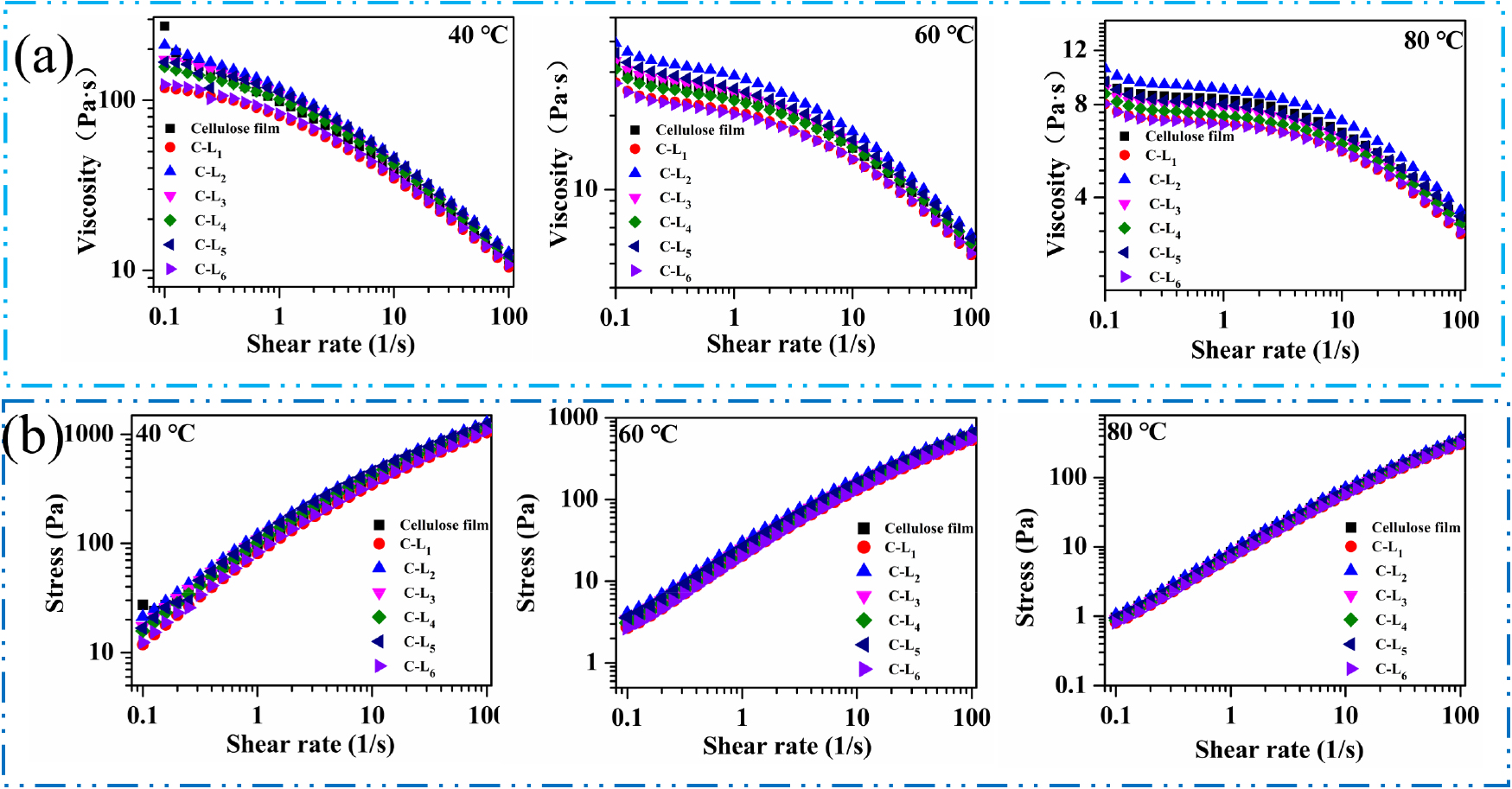
3.6 Rheological performance analysis
Fig. 8a shows the composite film solution viscosity characteristics at different temperatures. The viscosity of all solutions for composite films at different temperatures shows that the viscosity and shear rate changes present the same trend, with the viscosity decreasing with increasing temperature. At the same temperature, the viscosity decreases with an increase in shear rate. Reduced viscosity proves that the interactions between solution components are weakened as shear force increases, and the rearrangements between the components occur [48]. Among them, the viscosity of the solution from C-L2 showed the highest in all three temperatures, indicating the hybrid system of C-L2 was more uniform, and the performance of the composite film was optimal. The stresses of all samples at different temperatures showed an increase in yield stress with the increase in shear rate, in which C-L2 had the largest yield stress (Fig. 8b). The higher the yield stress, the closer the combination between the components, and the better the performance of the composite film.
3.7 Biodegradability
The degradation process of the composite film was determined by observing the changes in the morphology of the composite film over time (Fig. 9). The microorganisms in the soil can degrade the cellulose-based film. The mechanical strength of the pure cellulose film was lower than that of C-L2, and the pure cellulose film was easier to degrade than C-L2. The pure cellulose film started to degrade with tiny holes and ruptures on 5 days, and a large number of holes and cracking on 9 days, and completely degraded on 11 days. C-L2 was unchanged on 5 days, and it showed degradation on 9 days and degraded by about 2/3 after 11 days. The hydrophobic functional groups of lignin can prolong the survival time of the film in a humid environment. The results showed that the degradation time of the cellulose composite film increased after the addition of lignin, but it still had good degradation properties.
4. Conclusion
In this paper, the cellulose-lignin composite film was successfully prepared by the codissolution-precipitation method. Lignin and cellulose were connected by hydrogen bonding to construct an interacting network structure, which could transfer the force to the whole connection structure and improve the mechanical properties of the composite film. When the lignin to cellulose mass ratio was 1:20 in the composite film, the stress could be as high as 85.33 MPa. The composite film showed an increase in Young’s modulus due to the addition of lignin, so that the film can withstand greater stress. Furthermore, the composite film shows good UV-shielding, water-resistance, and biodegradation properties. The composite film is biodegradable and does not cause pollution to the environment.