1. Introduction
It is well known that dissolved and colloidal substances (DCS) present in papermaking system have great detrimental effects on paper machine runnability, paper properties, papermaking chemicals efficiency, etc. Numerous researches have been carried out to tackle the DCS-related problems. However, knowledge on how dissolved substances (DS) and colloidal substances (CS) contribute separately, is rare. DS and CS are very different chemically and physically1,2), therefore, their performances in papermaking wet-end process will be very different. It is known that, for a typical DCS water such as pulp filtrate or white water, cationic demand is mainly contributed by DS, while turbidity is mainly contributed by CS.3-5) Fixing agents (FA), as a class of water-soluble polyelectrolytes with high cationic charge density but low molecular weight, are generally effective in alleviating DCS depositing problems. Often these chemicals are found successful in decreasing the cationic demand and turbidity of a DCS water, however, this will mislead papermakers to believe that fixing agents are easy to remove DCS components. As a matter of fact, fixing agents are generally effective in removing CS, but not as effective in removing DS.3-5) A notable example is the low efficiency in reducing chemical oxygen demand (COD) of pulp filtrate when a pulp is treated with a fixing agent. It is reported that the removal ratio of COD by various fixing agents is generally only around 15-20%.5-8)
It is believed that DS, in contact with fixing agent, forms polyelectrolyte complexes (PECs) by charge neutralization mechanism.9-11) Although some of the PECs formed may remain soluble in water11,12), most of the PECs may separate out, in which some remain suspended in water as new CS particles, some may precipitate and deposit onto pulp fibers, fillers, or other CS particles. Obviously, the removal efficiency of FA on DS is closely related to characteristics of the PECs formed.
This study intended to unearth some deeper knowledge on the formation of DS-FA PECs and their characteristics. Our idea is using different fixing agents to treat different modeling water. The fixing agents are three polyamines with different levels of molecular weight and slightly different levels of charge density. The water includes a DCS water separated directly from a deinked old newspaper pulp (ONP) by centrifugation followed by filter paper filtration, another DCS water separated from the same deinked ONP but with most of the large CS particle being removed, a DS water modeled by oxidized starch (OS) solution, and a DS water modeled by hemicelluloses (HC) solution. Technologies such as focused beam reflectance measurement (FBRM), laser particle size analysis (LPSA) based on dynamic light scattering (DLS) principle, turbidity measurement, and charge demand measurement, are used to investigate the possible PEC formation.
2. Experimental
2.1 Materials
The polyamine fixing agents used in this study were synthesized in our laboratory. Table 1 shows two of their most important properties, intrinsic viscosity (IV) and charge density. In this study, IV indirectly represents molecular weight.
Table 1.
Properties of polyamine fixing agents
| Polyamines | Intrinsic viscosity, mL/g | Charge density, meq/g |
|---|---|---|
| PA-A | 4.52 | 5.93±0.05 |
| PA-B | 11.5 | 6.67±0.07 |
| PA-C | 49.0 | 6.21±0.02 |
The deinked ONP was taken from the machine chest of a newsprint line in Shangdong Province of China and kept frozen in a refrigerator. Before using, a proper portion of the pulp was de-frozen and the pulp consistency was adjusted to 3.6% by tap water.
Corn starch was provided by Papermate Science and Technology Co., Ltd. APMP spent liquor (APMP pulp were made from the mixture of 50% aspen, 25% acacia, and 25% eucalyptus) was taken from a pulp line in Shangdong Province of China, with 15-16% solid content, in which hemicelluloses took 7.5%.
Polydiallydimethylammonium chloride (PDADMAC) and potassium salt of polyvinyl sulfate (PVSK) standard solutions for colloidal titration were purchased from Wako Pure Chemical Industries, Ltd., and other chemicals are all chemical pure reagents.
2.2 Methods
2.2.1 Preparation of DCS water 1
The method used by Rundlof et al.13) was cited to prepare the named DCS water 1 in this study: First the 3.6% DIP was filtered with a 200 mesh wire, and the filtrate was centrifuged at 2,000 rpm for 15 min, then the supernatant was filtered again with 25-30 μm pore size filter paper, and the final filtrate was taken as DCS water, which does not contain fiber fines and fillers.
2.2.2 Preparation of DCS water 2
First, 1,000 g of DIP was put in a Bucher funnel without using any filter paper. By gentle dewatering, a fiber mat was formed on the funnel, and the first batch of filtrate (primary filtrate) was obtained. Then this fiber mat was used as the filter medium, and the primary filtrate was filtered once again to get the secondary filtrate. The filtration was repeated another two times with the fiber mat formed during the filtration procedure so that a transparent filtrate was obtained. After that, the last filtrate was filtered by a 0.22 μm pore size membrane and the obtained filtrate was named as DCS water 2. This water contained much less “large” colloidal particles than DCS water 1, but their dissolved components were actually the same.
2.2.3 Preparation of oxidized starch modeled DS water
The pH was adjusted to 8-10 with sodium hydroxide solution, then sodium hypochlorite solution was added at the dosage of 5% (calculated as content of available chlorine based on dry weight of starch). The oxidation reaction was conducted at between 35 and 40°C while the pH was kept at between 8 and 10 during the reaction process. After oxidation for 2.5 hours, the suspension pH was adjusted between 6.0 and 6.5, and sodium sulphite solution was added to remove the residual chlorine. After washing and filtering, oxidized starch powder was obtained by vacuum drying under 50°C, then it was dissolved in deionized water and diluted to 500 mg/L, and the solution was used as the first modeled DS water.
2.2.4 Preparation of hemicelluloses modeled DS water
The APMP spent liquor was acidified to pH 3 using 98% sulfuric acid and kept stirring at 45°C for 30 min, then the treated sample was centrifuged, and the supernatant was neutralized to pH 7 with a 6 M NaOH solution. Afterward, ethanol (three times volume of the supernatant) was added and the mixture was stood for 24 hours to give the hemicelluloses precipitates, which were collected by filtration, vacuum-dried at 50°C into hemicelluloses powder, and the purity of the powder were checked by FT-IR and 1H-NMR.14) A proper amount of the powder was dissolved in deionized water to prepare a 500 mg/L solution which was used as the second modeled DS water.
2.2.5 Application of polyamine fixing agents in DCS water and DS water
FBRM analysis was conducted on-site during the polyamines treatments on all four kinds of modeling water. The main procedures were as follows: 50 mL sample water was added into the test beaker, the FBRM probe was immersed in the sample. After setting the scanning speed at 2 m/s, scanning interval at 2 seconds, and agitation speed at 250 rpm, the FBRM was started to run. Once the baseline collected by FBRM was stable, a predetermined amount of polyamine solution (1 g/L) was added, the change of size and counts of the particles in the tested sample water were recorded by FBRM, and the test was stopped after collecting stable data for 2 minutes. The FBRM used in this work was an S400 from Mettler-Toledo, USA, installed with iC-FBRMTM 4.2 software for data collection. After FBRM test, the polyamines-treated samples were subjected to particle size analysis with a 90 Plus particle size analyzer from Brookhaven Instruments Corporation, turbidity measurement by a 2100 N turbidity meter from HANNA Instruments, and charge demand measurement by a charge analysis system (CAS) from AFG Analytic GMBH, Germany.
3. Results and Discussion
3.1 Effect of polyamines in treating two kinds of DCS water
First, the particle size and distribution of the DCS water 1 and 2 were measured by the particle size analyzer. The result showed that the size of the particles in DCS water 1 concentrated in the range of 200-1,500 nm and averaged at 472.1± 37.3 nm, by contrast, that in DCS water 2 concentrated in the range of 50-300 nm and averaged at 124.8±1.9 nm. This indicates that the procedures described in the Experimental, i.e., multiple times of filtration by pulp mat followed by 0.22 μm membrane filtering, was efficient in removing the “large” colloidal particles that existed in DCS water 1.
Then the charge demands of DCS water 1 and 2 were determined to be 0.660 meq/L and 0.606 meq/L, respectively. The dosage of polyamines was “arbitrarily” determined to be 60 mg/L based on stoichiometric calculation, which would neutralize 53.9%, 60.7% and 56.4% of the anionicity of DCS water 1, and 58.7%, 66.1% and 61.5% of the anions in DCS water 2 by PA-A, B and C, respectively.
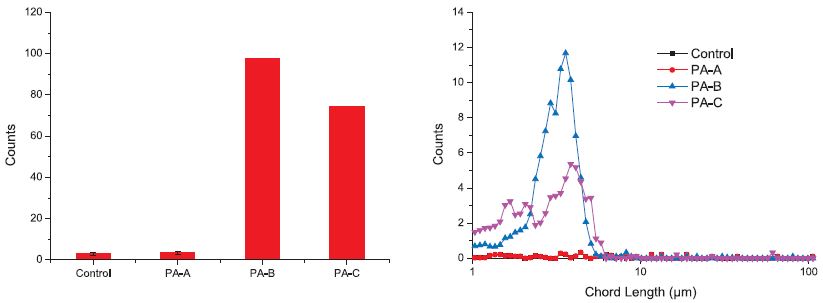
During the treatments, it was found that the originally clear DCS water 1 turned turbid immediately after the addition of polyamines, indicating a substantial amount of new particles were formed. FBRM was then used to measure the numbers (counts), sizes (chord length) and size distribution (chord length distribution) of the new particles in DCS water 1, and the results are shown in Fig. 1. It can be seen that large amounts of new particles emerged during the polyamine treatments. Compared to those of the polyamine-treated samples, the particle counts in the control sample was actually neglectable. Fig. 1 also shows the visible difference among three polyamines in producing new particles, which is notable that the polyamine with higher molecular weight formed more particles with larger sizes, especially those with chord length higher than 40 μm. That polyamine with higher molecular weight induces more CS agglomeration has also been discovered in our other publications.15,16)
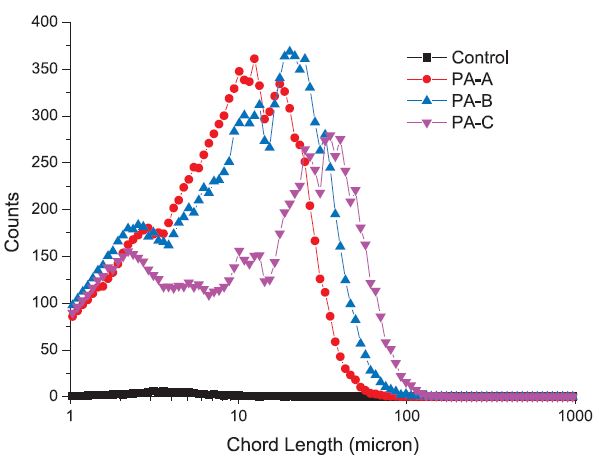
Fig. 1.
Size distribution of particles in DCS water 1 under the treatment of different polyamine fixing agents.
The very lower counts of particles in the control sample shown in Fig. 1 indicates that in DCS water 1, there are little amount of colloidal particles which can be measured by FBRM (hereafter annotated as CS>FBRM Limit), but the rise of CS>FBRM Limit under the treatment of polyamines implies that there may be a chance that the dissolved substances (DS) or the colloidal particles which are too small to be measured by FBRM (hereafter annotated as CS<FBRM Limit) give the large amount of new CS>FBRM Limit under the treatment of polyamine. However, whether the CS>FBRM Limit are mainly originated from DS-FA complexes, or CS<FBRM Limit agglomerates, or “capsules” formed by automatic deposition of DS-FA agent complexes around CS<FBRM Limit particles (hereafter annotated as “capsules”), remains unclear.
Attempting to decouple the above mentioned three effects, DCS water 2 with smaller CS particles than DCS water 1 was used in the second step. Similar to DCS water 1, it was found that during the experiments, the originally clear DCS water 2 became turbid when the polyamines were added. However, when the FBRM was used to measure the treated samples, the results showed that the newly-formed particles were not detectable (the results for the treated sample were the same as that of the control which was only no more than 1/10 of the control in Fig. 1). This is to say, the particles formed from the DCS water 2, no matter in what mechanism, belong to CS<FBRM Limit, therefore, it can be deduced that those new particles detectable by FBRM in Fig. 1, are mainly formed by agglomerating CS<FBRM Limit into CS>FBRM Limit, or by deposition of newly-formed PEC onto the CS<FBRM Limit to such a degree that the resultant particles turn into CS>FBRM Limit, in another word, PEC formation alone, and agglomeration of CS particles as small as those in the untreated DCS water 2, can not result in CS>FBRM Limit particles.
That DS can be attached to CS by polyamines, resulting in an increase of DS removal ratio, whereas CS are more difficult to remove due to the stabilization effect of DS, has been revealed in another publication of ours.17)
Turbidity and particle size for DCS water 2 were continued to be measured and the data are shown in Table 2. Unexpectedly, PA-A, which has the lowest molecular weight and charge density, induced the lowest turbidity but largest particles, while PA-B with the highest charge density and PA-C with the highest molecular weight, had the similar effect.
That PA-A formed the largest particle in DCS water 2 is actually very unexpectedly, since it contradicts with what was seen in Fig. 1. However, one should pay attention to the fact that DCS water 1 contained higher amount of CS and the CS particles were larger than those in DCS water 2. In a sense it can be said that the DCS water 2 was much more like a “real” DS water. That PA-A formed less but larger CS<FBRM Limit also indicates that, if PEC formation is the main mechanism contributed to the increase of turbidity and particles size of DCS water 2 under the treatment of polyamines, then PA-A tends to form loose but dispersible PEC with DS18), while PA-B and PA-C tend to form denser PECs with DS, which are likely in “ladder” structure.19,20)
3.2 Effect of polyamines in treating two kinds of modeled DS water
In this section, first the turbidity and laser particle analysis were carried out for the two modeled DS water. The results show that there was no contribution of any particle, therefore the two models are ideal for simulating DS systems. Then, by colloidal titration, the cationic demands of the OS modeled DS water was found to be 0.0699 meq/L and for the HC modeled DS water it was 0.361 meq/L.
For the OS water system, the dosages of the polyamines were fixed at 0.8% and 1.6%, calculated as dry polyamine to dry OS (hereafter the unit is expressed as % d/d). The theoretical neutralization degree based on stoichiometric calculation, and the practically measured neutralization degree, are illustrated in Fig. 2 (a). Interestingly it is found that, for PA-A whose molecular weight was the lowest, the measured neutralization degree was lower than the theoretical one, for PA-B, the measured degree almost equaled the theoretical one, and for PA-C, the measured one was higher than theoretical. This indicates that fixing agents interact with the component in DS in a stoichiometric way only when their molecular weight is well-matched. If the molecular weight of fixing agent is too low, the neutralization effect will be less efficient than the stoichiometric neutralization degree, and if the molecular weight is high enough, the neutralization effect will be higher than the stoichiometric neutralization degree. In another word, it was found by comparing three polyamines that the PA-C works in a way more similar to a flocculant.
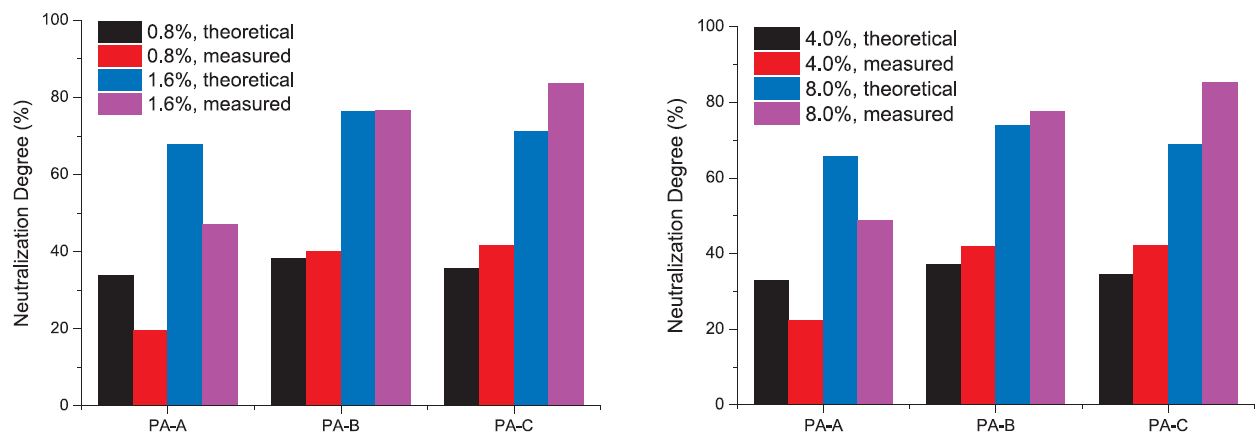
Fig. 2.
Comparison on theoretical and measured neutralization degree of anionicity for OS (a, left) and HC (b, right) solution treated with different polyamine fixing agents.
In Fig. 2 (b) for HC modeled water system where the polyamines were dosed at 4% and 8%, very similar phenomenon was seen. This reinforces our viewpoint that PA-C works more like a flocculant than PA-B and PA-A do.
It was discovered that, when the polyamines were added to the OS solutions, no turbidity change was seen, nor did FBRM detect anything. Our attention then turned to laser particle analysis, and the results are shown in Fig. 3 (a) for 0.8% d/d PA addition and Fig. 3 (b) for 1.6% d/d PA addition. It can be seen that particles with a mean diameter of 200 nm came out under the treatment of 0.8% d/d PA-C, but no particles were seen under the same dosage of PA-A and PA-B. When the dosage was increased to 1.6% d/d, particles with a mean diameter of 200 nm also came out for PA-B, but still none for PA-A. The results indicate that formation of DS-polyamine PEC is related to the dosage and molecular weight of the polyamines. Higher molecular weighted polyamine forms PEC easier than lower molecular weighted ones.
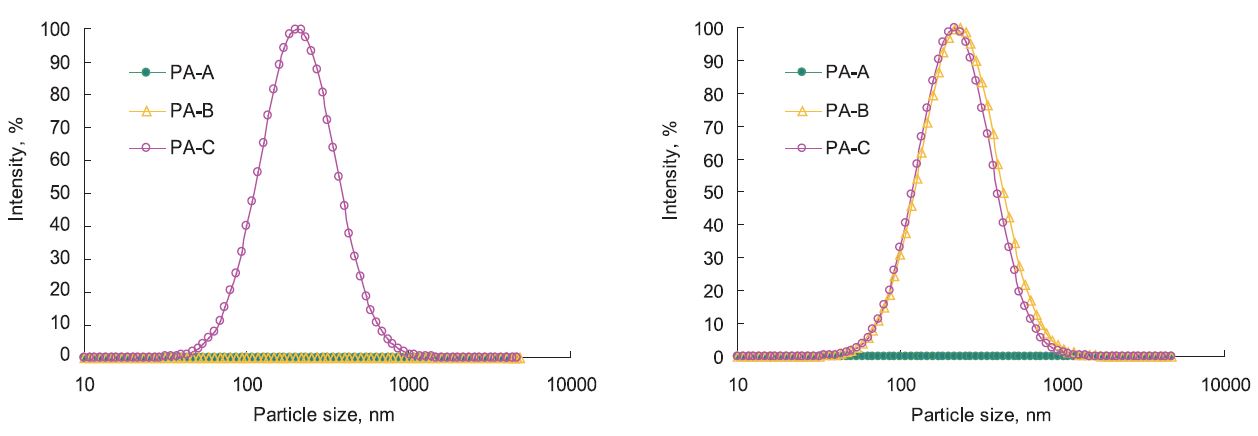
Fig. 3.
Size distribution of PEC formed in OS solution with the treatment of (a, left) 0.8% and (b, right) 1.6% polyamine treatment.
When the polyamines were dosed to the HC modeled DS water at 4% and 8% dosages, it was apparently seen that the clear solution changed into turbid suspension for all the treatments except for PA-A at 4% d/d. Different from that no turbidity was detected in the OS modeled DS water, turbidity increase of HC modeled DS water under the treatments of polyamines was seen (shown in Table 3). Obviously, HC is easier to for complex with the polyamines than OS. It is also seen that turbidity values at 8% d/d dosage were higher than those at 4% d/d dosage, indicating more PECs are formed with increased dosing of polyamines.
Table 3.
Turbidity of HC modeled DS water samples under the treatment of different polyamines
| Polyamine dosage | PA-A | PA-B | PA-C |
|---|---|---|---|
| 4% d/d | 0 | 0.82 | 1.27 |
| 8% d/d | 1.56 | 15.9 | 7.82 |
The laser scattering particle analysis results for the HC modeled DS water treated at 4% d/d polyamine are shown in Fig. 4 (a). Compared to what was seen in Fig. 3 for the OS modeled DS water, where PA-A at both dosages and PA-B at the lower dosage failed to form PEC, here, PEC formation was successful for all the treatments. Interesting again, the size of the PEC formed by PA-A with HC was larger than those by PA-B and PA-C, reaching as high as a 400 nm mean diameter, and the size distribution was also wider. On contrast, PA-B and PA-C formed PEC particles with smaller average diameter around 100- 200 nm. The phenomenon agreed with what was seen in the interaction of polyamines with DCS water, where PA-A also formed the largest particles compared to PA-B and PA-C. Therefore, it is more convincing to say that, PA-A, with the lowest molecular weight and charge density among the three polyamines, has the tendency to form loose and dispersed PEC. This is actually identical to say, polyamine with higher molecular weight will form denser PEC with DS components.
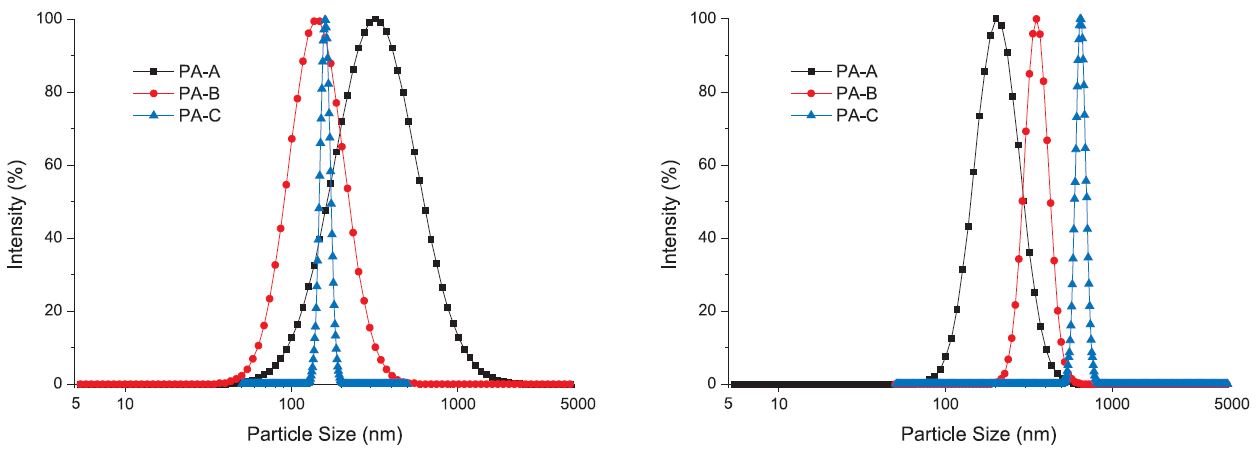
Fig. 4.
Size distribution of PEC formed in HC modeled DS water under the treatment of polyamines at (a, left) 4% d/d and (b, right) 8% d/d.
The laser scattering particle analysis results for the HC modeled DS water treated at 8% d/d polyamine are shown in Fig. 4 (b). It is seen that at this higher polyamine dosage, the story is different, as the size of PEC formed by HC and polyamines is consistent with the molecular weight of polyamines. This indicates that increasing the dosage of polyamine may induce more agglomeration of PEC to form larger particles.
Comparison of dosage effect on the size distribution of HC-polyamine PECs is shown in Fig. 5, which tells an even more interesting story that for PA-A with too low molecular weight, increasing its dosage will produce more dense and therefore smaller PECs; for PA-B, dosage increase results in larger but denser PECs; for PA-C, the PECs formed at the lower dosage are dense enough, so higher dosage mainly increases the size of PECs.
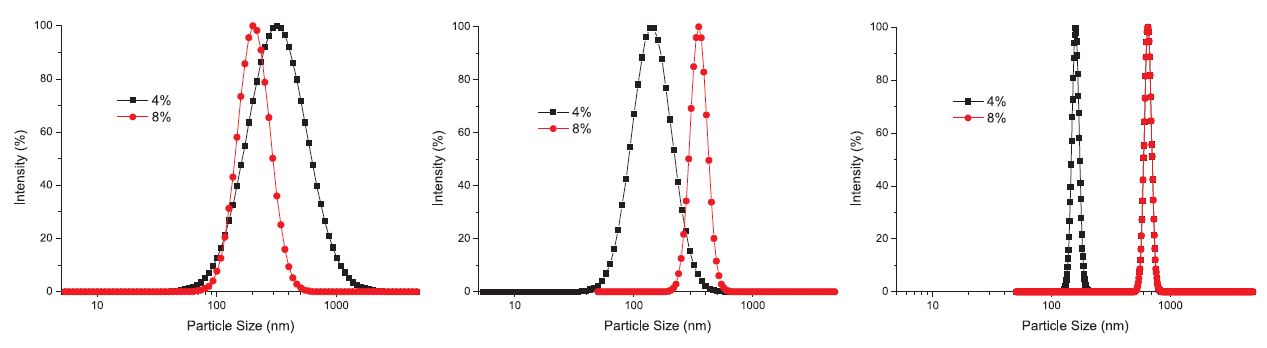
Fig. 5.
Comparison of size and distribution for HC-polyamine PECs for different polyamine at different dosages (a (left), b (middle), c (right) for polyamine A, B, C, respectively).
Moreover, that the size distribution of the PECs was always in the order of PA-C < PA-B < PA-A also supports the viewpoint that fixing agent with higher molecular weight tends to produce denser PECs.
The difference between the OS and HC modeled DS water in forming PEC was also revealed by FBRM. Although the PEC formed in the OS system was totally undetectable by FBRM, the PECs formed by PA-B and PA-C with HC were detected by FBRM at 8% dosage of polyamines (at 4% dosage, PEC formed in the HC system was also undetectable). As illustrated in Fig. 6, the FBRM detectable PECs formed by PA-A is neglectable, but PA-B was found giving rise to more FBRM detectable PECs than PA-C, while PA-C formed more PECs with larger sizes.
4. Conclusions
1. Formation of DS-fixing agent PEC highly depends on the DS components, fixing agent molecular weight and its dosage, as very different phenomena can be seen in this study for different DS components, for polyamines with different molecular weight, and for the same polyamine used at a different dosage.
2. For fixing agent with enough low molecular weight and at enough low dosage, it can form loose and dispersible DS-fixing agent PECs, and increasing its dosage will produce denser and smaller PECs; for fixing agent with enough high molecular weight, it can directly form dense DS-fixing agent PECs, and increasing its dosage mainly increases the size of PECs; for fixing agent with medium molecular weight, its behaves between the two cases described above.
3. Although hemicelluloses modeled DS water showed that a small part of PECs formed are detectable by FBRM, the performance of DCS water 2 used in this study showed that DS-fixing agent PECs alone are generally too small to be detected by FBRM.
4. Based on the performance of DCS water 1 and DCS used in this study, it can be concluded that small CS particles undetectable by FBRM can be the “seeds” for producing larger particles detectable by FBRM, either by agglomerating the small CS particles or by depositing DS-fixing agent PECs onto the small CS particles. For the formation of FBRM-detectable particles, the agglomeration of medium sized CS particles (between 125-470 nm in this study), or deposition of DS-fixing agent PECs onto such particles, seems critical.