1. Introduction
With the increasing consumption of non-renewable energy and increasingly prominent environmental pollution problems, exploring the energy from the renewable agriculture and forestry biomass has gained significant interest lately.1) Corn stalk, as a widely distributed agricultural resource, is largely discarded in the fields and is not adequately and effectively utilized. Corn stalk has high contents of cellulose (35%-40%) and hemicellulose (20%-35%), and can produce energy, polymeric materials, chemical products with high value added, developing new fields of industrial chemistry and biomaterials. The hemicelluloses extracted from corn stalk would have high potential for the utilization of the corn stalk and full use of agricultural waste.
Hemicelluloses are found in the primary and secondary cell walls in many plants. Hemicelluloses contain branched chains polysaccharides, and composed of a variety of sugar and uronic acid. The hemicellulose in plant tissue is very rich in content, annual gramineous plants such as corn stalk and straw contain 20%-35% hemicelluloses. The principle saccharide components in hemicelluloses are D-xylose, D-mannose, D-glucose, D-galactose, L-arabinose, D-glucuronic acid, 4-O-methyl-D-glucuronic acid, D-galacturonic acid, and to a lesser extent, L-rhamnose, L-fucose, and various O-methylated sugars. The hemicelluloses of the gramineae family have a backbone of β-1,4-linked xylopyranosyl units, and have furan arabia sugar units attached to some C-2 position of the main xylan chain and single arabinofuranosyl units attached to some C-3 position of the main xylan chain. These large amounts of hemicelluloses are worth exploiting to serve as renewable high-density fuel,2) paper coatings and adhesives,3) xylooligosaccharides, 4) latex,5) hemicellulose-reinforced nanocellulose hydrogels.6) The current research was mainly concentrated on the application of hemicellulose products. However, the structure of hemicellulose extracted by various extraction methods has not been deeply studied yet.
In recent years, the conversion of hemicelluloses extracted from lignocellulosic biomass to various chemicals fuels and fibers materials has attracted more and more attentions. Many methods have been used to extract hemicellulose such as hot water extraction and acid extraction. Vallejos et al.7) reported that sugarcane bagasse was hydrothermally treated under isothermal conditions at 160, 170, and 180°C, using a liquid to solid ratio of 14:1. Sánchez et al.8) reported the extraction of hemicelluloses from corn stalks using different reagents (H2SO4, HNO3, HCl, CH3COOH, CF3COOH, Ca(OH)2, NaOH) with autoclave hydrolysis processes (121°C, 1:20 solid/liquid ratio, 60 min, pH=4 or 8). In these extraction methods, hemicellulose was extracted only once, and only one type of hemicellulose was extracted. Thus, it limits the use of a variety of hemicelluloses and the full utilization of lignocellulosic resources. Among these extraction methods, alkaline extraction methods are rarely studied, especially stepwise alkaline extraction. A variety of hemicellulose can be isolated by stepwise extraction, which can meet the diversified application of hemicellulose, and can be used effectively in lignocellulosic biomass. Hemicelluloses extracted from gradient increasing concentration of alkali solution represent different branching degrees, thermal stability and sugar fractions. To some extent, hemicelluloses extracted by the higher alkaline extraction contain lower branching degree and higher thermal stability, these properties are beneficial to the modification of straight chain hemicelluloses and high temperature reactions. Hemicelluloses are not homogeneous polysaccharides, which are connected to the cellulose and lignin by hydrogen bond and covalent bond in the cell wall. In addition, hemicellulose can also be linked to acetyl groups and hydroxycinnamic acid via ester bonds in the cell wall of gramineous plants.9) The presence of these bonds limit the dissolution of hemicellulose, so it is necessary to develop a method for better dissolution and the availability of highly purified hemicellulose. Alkaline solution can destroy the plant cell wall, it can also break the hydrogen bonds, the covalent bonds and ester bonds. With the increase in alkaline solution concentration can dissolve hemicellulose in close contact with the cell wall. In addition, it has been reported that the addition of ethanol in alkaline solution can break the resistance of the cell wall of the plant.10)
In this work, hemicelluloses were step by step extracted from corn stalk with water, KOH aqueous solution (0.6%, 1.0%, 1.5%, and 2.0%) and the mixture of 2.5% KOH and ethanol. The composition and structure of hemicellulose in corn stalk were studied. In addition, effect of alkaline solution concentration on the extraction efficiency of the hemicelluloses was investigated. The yields, chemical composition, functional group, thermal stability and structural features of hemicelluloses were analyzed.
2. Materials and Methods
2.1 Materials
Corn stalk was obtained from a farm in Shandong province. It was air-dried and then cut into pieces in size of 1-3 cm. The pieces were grounded into powder of 40-60 mesh sieve. Corn stalk was first extracted with the mixture of 2:1 (v/v) benzeneethanol in a soxhlet apparatus for 6 h, and the dewaxed powder were dried in an oven for 16 h at 60°C. The chemical composition (based on mass) of corn stalk were 42.3% cellulose, 21.8% hemicellulose, 18.0% acid-insoluble lignin and 5.9% ash according to TAPPI standard methods.
Ethanol (99.7 vol%), ethanol (95 vol%) and glacial acetic acid (99.5 wt%) were purchased from Tianjin Fu Yu Fine Chemical Co., Ltd. Potassium hydroxide (96 wt%) was purchased from Tianjin Da Mao Chemical Co. Benzene (99.5 wt%) and hydrochloric acid (36-38 wt%) were purchased from Lai Yang Chemical Co., Ltd. L-Arabia sugar, D-glucose, D-xylose, D-galactose, D-mannose, L-rhamnose, D-glucuronic acid and D-galacturonic acid were purchased from Sigma company.
2.2 Hemicellulose extraction
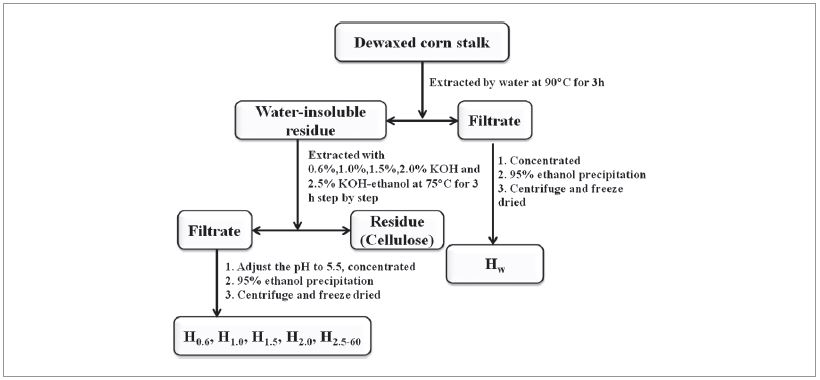
The flow chart of stepwise alkali extraction of hemicelluloses in corn stalk is shown in Fig. 1. The corn stalk was dewaxed by extraction with benzene and ethanol. The dewaxed corn stalk was step by step continuously extracted with water, alkaline solutions at different concentrations and alkali ethanol solution. First, the dewaxed corn stalk (20 g) were extracted with water at 90°C for 3 h under a solid to liquid ratio of 1:20 (g/mL). The reaction system was cooled to room temperature and filtrated. Then the filtrate was concentrated to about 50 mL and was poured into 3 times volumes of 95% ethanol with vigorously stirring. Then the mixture was placed for 1 h, the hemicellulose precipitate was generated, and the hemicellulose was obtained by centrifugation and freeze drying. Water insoluble solid residue was dried in an oven at 60°C for 16 h. Then, the water insoluble solid residue was stepwise extracted with gradually increased KOH concentration at 75°C for 3 h under a ratio of 1:20 (g/mL), in which KOH solutions concentrations were of 0.6%, 1.0%, 1.5% and 2.0%. In final extraction step the mixture of KOH-ethanol was selected to improve hemicellulose yield, in which 2.5% KOH was mixed with 99.7 vol% ethanol at volume ratio of 2:3. In the stepwise extraction process the water insoluble solid residue was extracted by KOH concentrations from 0.6% to 2.5%, and then the mixture was filtered to obtain the different types residue and filtrate. The obtained residues were dried in oven at 60°C for 16 h and then used to the next step extraction. There are five stepwise extraction in the continuously extraction process, and five types of filtrates and residues were obtained. The five types of alkaliextractable filtrates were acidified to pH 5.5 with glacial acetic acid before they were concentrated to about 50 mL. Subsequently, the concentrated solution was poured into 3 volumes of 95% ethanol with vigorously stirring. After the mixture was placed for 1 h, the hemicellulose precipitate was generated, and the hemicellulose was obtained by centrifugation and freeze drying. Hemicellulose extracted from water, 0.6%, 1.0%, 1.5%, and 2.0% KOH aqueous solutions and 2.5% KOH containing 60% ethanol were labeled as Hw, H0.6, H1.0, H1.5, H2.0, and H2.5-60 according to the order of stepwise extractions, respectively. The yields of hemicellulose extracted by six solvents concentrations were calculated according to the ratio of initial amount of the dried dewaxed corn stalk and the total content of hemicellulose in the corn stalk.
2.3 Sugar analysis
The chemical compositions of the hemicellulose were analyzed by high performance anion exchange chromatography (Thermo Fisher, ICS-5000, America). Each of the samples (5 mg) was hydrolyzed with 0.125 mL of 72% sulphuric acid and 1.375 mL of ultra pure water in the 15 mL pressure vessel at 105°C for 2.5 h. Then the hydrolysate was filtrated with a pore size of 0.22 μL membrane, diluted 500 times and injected into a HPAEC system to determine monosaccharide and uronic acid of hydrolysate. The mobile phase firstly eluted with 10 mmol NaOH for 22 min, followed by 200 mmol NaOH and 300 mmol NaAc gradient elution for 5 min. The neutral sugar and uronic acid could be separated. In the end, 10 mmol NaOH was used to balance the chromatographic column for 10 min, the column temperature was maintained at 30°C and the flow rate was 0.4 mL/min.
2.4 Functional group analysis
Infrared spectra measurements were performed on FTIR spectrometer (Shimadzu, Japan). Dried hemicellulose samples were grounded and pelletized with KBr powder and the spectra were recorded in the range of 500-4,000 cm-1.
2.5 Thermal analysis
Thermal stability of the samples were investigated by using TG and differential thermogravimetric (DTG) analysis on a simultaneous thermal analyzer (TG Q500, USA). 10 mg of hemicelluloses were heated in a sample disk from room temperature to 600°C with a heating rate of 10°C/min under nitrogen atmosphere. The flow rate was 30 mL/min.
2.6 NMR spectroscopy
Nuclear magnetic resonance of hemicellulose sample was determined by nuclear magnetic resonance spectrometer (AVANCE II 400). 20 mg of hemicellulose sample was dissolved in 1 mL of D2O for 1H-NMR analysis, and 80 mg of hemicellulose sample was dissolved in 1 mL of D2O for 13C-NMR analysis.
3. Results and Discussion
3.1 Hemicellulose yield
The yields of hemicelluloses from corn stalk by hydrothermal and stepwise alkali extraction were given in Table 1. As can be seen from Table 1, the hemicellulose yields of Hw, H0.6, H1.0, H1.5, H2.0, H2.5-60 based on the dried dewaxed corn stalk weight were 0.8%, 3.0%, 4.9%, 7.3%, 3.3%, and 2.6%, respectively. The hemicellulose yields of Hw, H0.6, H1.0, H1.5, H2.0, H2.5-60 based on the total hemicellulose content in corn stalk were 2.6%, 10.2%, 16.5%, 24.8%, 11.2%, and 8.7%, respectively. Obviously, the total yield of the six types of hemicelluloses accounted for 74% of the total hemicellulose in corn stalk. The -OH in alkaline solution can make cellulose swelling, damage hydrogen bonds between cellulose and hemicellulose. In addition, a certain alkali concentration can damage the chemical connection between the hemicellulose and lignin in the cell wall, which leads to a better dissolution of hemicellulose. In five types of alkali-extractable hemicelluloses, the hemicellulos yield increased from 10.2% to 24.8% with the increment of KOH concentrations from 0.6% to 1.5%, and then the hemicellulos yield decreased to 8.7% with increment of alkali concentration. The reason might be that most of hemicelluloses were separated at the low concentration alkali solution, and the high concentration of the alkali solution could only separate a small fraction of the hemicellulose. Among the six types of hemicelluloses, the yield (24.8%) of hemicellulose extracted by 1.5% KOH was the highest.
3.2 Carbohydrate composition analysis
The neutral monosaccharide composition and content of uronic acids of the six types of hemicelluloses were given in Table 2. The water-soluble hemicellulosic fraction (Hw) contained xylose (36.8%) together with arabinose (18.7%) and glucose (16.0%) as well as galactose (22.4%). Mannose was not detected in five types of alkali-soluble hemicelluloses, content of uronic acid (1.8%-2.7%) of the five types of alkali-soluble hemicelluloses were relatively low compared with uronic acid (3.6%) of the water-soluble hemicellulose. And this implied that the water-soluble extraction was easier to separate multi-branched hemicellulose.11) In addition, the content of glucose in water-soluble hemicellulose was much higher than that of alkali-soluble hemicellulose, which indicated that water-soluble hemicellulose may contain a certain amount of non-cellulose dextran.12,13) Compared to the water-soluble hemicellulose, xylose (78.4%-90.0%) was main carbohydrate component of alkali-soluble hemicellulose, which indicated that xylan was the backbone of hemicellulose. The second was the arabinose (5.3%-14.8%), suggesting that arabinose was the main branched chain of hemicellulose.14) In addition, the alkali-soluble hemicellulose also contained a small amount of glucose and galactose, accounting for 1.8%-4.0% and 1.1%-4.9%, respectively. It can be seen from the Table 2 that as the concentration of KOH increased from 0.6% to 1.5%, the xylose content increased from 78.4% to 90.0%, while the contents of arabinose decreased from 14.8% to 5.3%. When the alkali concentration increased from 1.5% to 2.5%, the xylose content decreased from 90.0% to 87.6%, while the contents of arabinose increased from 5.3% to 7.75%. This might be due to that increasing the alkali concentration can dissolve the straight chain type of hemicellulose, decreasing alkali concentration can dissolve the branched chain type of hemicellulose.
Table 2.
The neutral sugar and uronic acid content of hemicelluloses
Generally, the ratio of arabinose to xylose (Ara/Xyl) represents the degree of substitution of hemicellulose backbone.15) From the Table 2, it can be seen that with the increment of alkali concentration from 0.6% to 1.5%, the ratio of arabinose to xylose decreased from 0.2 to 0.1. This implied that the increment in the alkali concentration could decrease the branching degree of the obtained hemicellulose. The ratio of arabinose to xylose increased slightly with further increment of the alkali concentration, which was in accordance with the results discussed above. The corn stalk alkali-soluble hemicellulose was xylan containing a small amount of arabinose glycosyl, the relatively high alkali concentration can dissolve straight chain type of hemicellulose, while relatively low alkali concentration can dissolve branched chain type of hemicellulose.
3.3 Functional group analysis
The FT-IR spectra of the water-extraction hemicellulose and five types of alkali-soluble hemicellulose were shown in Fig. 2. The results indicated that stepwise alkaline extraction did not result in any significant change in functional group of hemicellulose. As shown in Fig. 2, Hw, H0.6 and H1.0 showed very similar spectra. The band at 3,430 cm-1 corresponded to the -OH stretching vibration of the hemicelluloses, the band at 2,923 cm-1 was attributed to the C-H stretching vibration, and the band at 1,415 cm-1 was attributed to the -COO stretching vibration. Furthermore, a small peak at 1,254 cm-1 was due to the C-H bending vibration, whereas a prominent absorption at 1,038 cm-1 was assigned to the C-O-C stretching of glycosidic bond. From the spectrum diagram, three kinds of alkali-soluble hemicelluloses (H1.5, H2.0 and H2.5-60) showed rather similar spectra, indicating that these hemicelluloses had similar structure. The absorption peak at 1,565 cm-1 was the vibration of the aromatic compound, indicating that the three alkali-soluble hemicellulose contains an aromatic compound.16) The absorption of 1,640 cm-1 and 1,415 cm-1 were the anti symmetric stretching vibration and symmetric stretching vibration of C=O in carboxyl group. The absorption peaks at 1,415 cm-1 was symmetrical stretching vibration of C=O in the carboxyl group. In addition, the absorption peak at 1,254 cm-1 was derived from the bending vibration of C-H. The bond at 1,038 cm-1 was the stretching vibrations of the C-O-C of the sugar group.17)
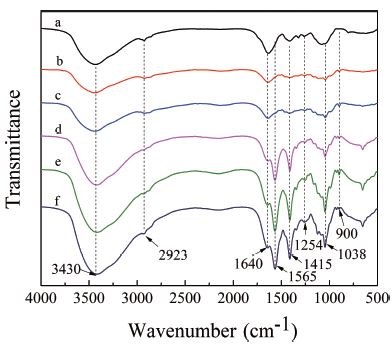
Fig. 2.
FT-IR spectra of hemicelluloses: a. hemicellulose extracted by water; b. hemicellulose extracted by 0.6% KOH; c. hemicellulose extracted by 1.0% KOH; d. hemicellulose extracted by 1.5% KOH; e. hemicellulose extracted by 2.0% KOH; f. hemicellulose extracted by 2.5% KOH-ethanol.
The above analysis showed that the main chain of the six types of hemicelluloses were xylan, and the arabinosyl group was attached to the xylan as a side chain. In addition, the absorption peak at 900 cm-1 was caused by the frequency vibration of the C-1 group, indicating that the 1-4-β-glycosidic bond was the main mode of attachment of the xylan. Compared with Hw, H0.6 and H1.0, peak at 1,565 cm-1 was observed in the H1.5, H2.0 and H2.5-60 spectra, the peak was the vibration of the aromatic compound, indicating that H1.5, H2.0 and H2.5-60 contained an aromatic compound.
3.4 Thermogravimetric analysis
Thermogravimetric curves of Hw, H0.6, H1.0, H1.5, H2.0 and H2.0-60 were shown in Fig. 3. The thermal decomposition of hemicelluloses can be divided into three phases. At the first phase, the weight loss of hemicellulose observed below 100°C was ascribed to moisture evaporation. The second phase was rapid decomposition phase, the temperature range was 200°C-400°C, six kinds of hemicelluloses weight decreased rapidly at this phase, the weight loss was 40%-50%, suggesting that hemicellulose has been degraded at this stage, the C-C and C-O bonds connected with the main branch of hemicelluloses were broken. At the same time, some decarboxylation and decarbonylation reactions occurred in hemicelluloses. These chemical changes led to the degradation of hemicellulose into CO, CO2 and light hydrocarbons such as CH4 and C2H4.18) In addition, all of the hemicelluloses began to decompose at about 200°C. The temperature range of 400°C-600°C was the third stage, which was mainly for the carbonization stage. The weight loss gradually in the third stage, which was generally considered to be the further crack of C-C and C-H bonds.
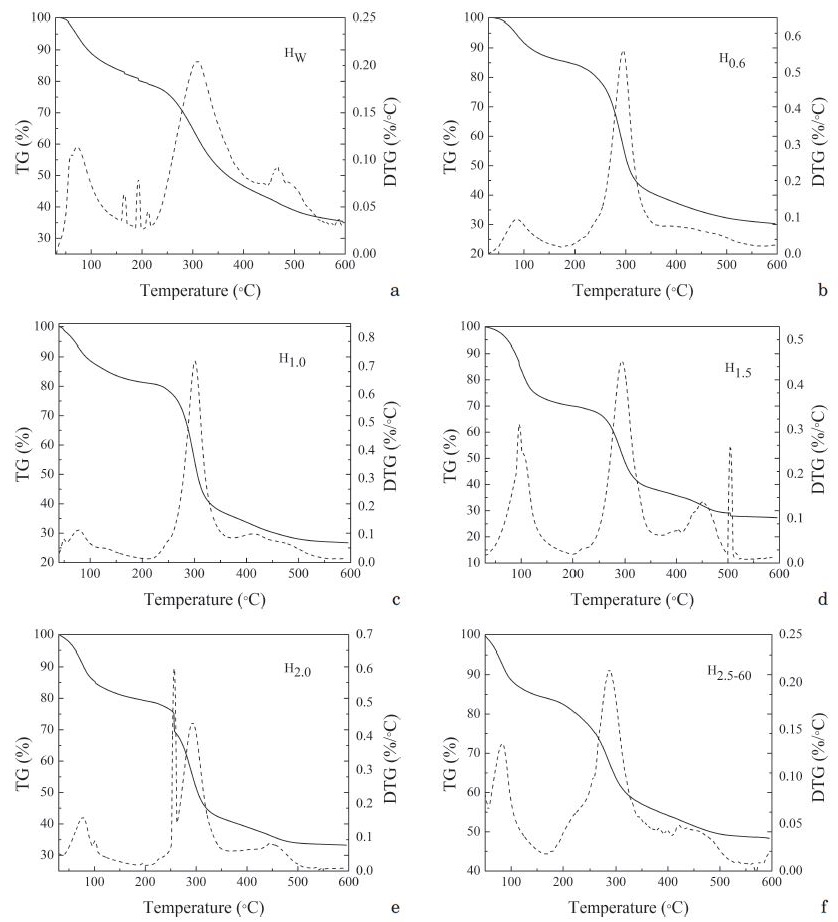
Fig. 3.
TG and DTG curves of hemicelluloses: a. hemicellulose extracted by water; b. hemicellulose extracted by 0.6% KOH; c. hemicellulose extracted by 1.0% KOH; d. hemicellulose extracted by 1.5% KOH; e. hemicellulose extracted by 2.0% KOH; f. hemicellulose extracted by 2.5% KOH-ethanol.
It can be seen from Fig. 3 that the initial decomposition temperature of Hw, H0.6, H1.0, H1.5, H2.0 and H2.5-60 were 240°C, 245°C, 248°C, 252°C, 240°C, and 200°C, respectively. In six types of hemicelluloses, the initial decomposition temperature of the obtained hemicellulose increased from 240°C to 252°C with the increment of alkali concentrations and reached the maximum in 1.5% KOH, and then decreased from 252°C to 200°C with further increment of alkali concentration. It was shown that the thermal stability of the hemicellulos obtained from 1.5% KOH extraction process was optimal compared to the other types of hemicelluloses. The weight loss of Hw, H0.6, H1.0, H1.5, H2.0 and H2.5-60 were 35.4%, 30.3%, 26.7%, 27.3%, 33.2%, and 48.3% at 600°C, respectively. The weight loss of hemicellulose might be oxidation of the organics in hemicelluloses. At 600°C, solid residues were some char residue with graphite structure and some C-C bonds may not be completely broken. This resulted more solid residues at 600°C.19) The samples obtained by the 1.5% KOH extraction showed the stronger thermal stability than the other five types of hemicelluloses, the reason was the initial decomposition temperature of H1.5 was highest in the six types of hemicelluloses, and H1.5 contained more straight chains and fewer branched chains. According to the above analysis, it indicated that the hemicellulose extracted with 1.5% KOH has higher thermal stability than the other five types of hemicelluloses.
3.5 NMR spectroscopy analysis
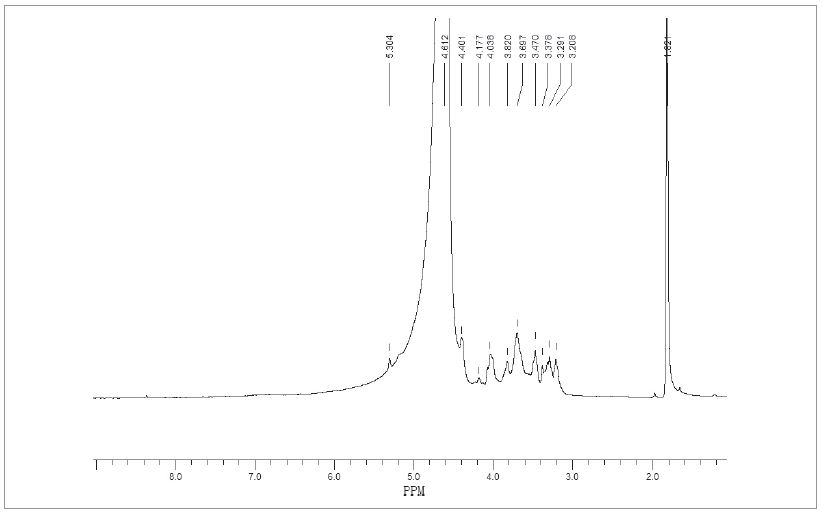
According to the analysis above, including the yield analysis, sugar components analysis functional groups analysis and thermal stability analysis, corn stalk alkaline extraction with 1.5% KOH was optimal condition. In order to further understand the structure of H1.5, nuclear magnetic resonance was used to analyze the skeleton of hemicellulose polymer and the side chain structure attached to the main chain. The signal peaks were analyzed according to the relevant literatures.20,21) The 1H-NMR spectrum of optimum hemicellulose extracted with 1.5% KOH was shown in Fig. 4. The chemical shift of anomeric hydrogen in the 1HNMR were generally in the range of 4.3 to 5.9 ppm. When the anomeric hydrogen was presented at 4.9 ppm to 5.9 ppm indicating that the α was connection mode and appeared at 4.3 ppm to 4.9 ppm indicating the β was connection mode. The signal of the anomeric hydrogen appeared at 4.4 ppm, indicating that the D-xylosyl group formed the backbone with the β-glycosidic bond. 3.2 ppm to 4.4 ppm was the signal peak of the β-D-xylopyranose unit of hemicellulose. The H-1, H-5eq, H-4, H-3, H-5ax and H-2 signals in the β-D-xylopyranose units appeared at 4.4 ppm, 3.8 ppm, 3.69 ppm, 3.47 ppm, 3.29 ppm, and 3.2 ppm. In addition, 1.8 ppm signal peak might be from the hydrogen atoms of methyl and methylene, 3.37 ppm was -OCH3 signal peak, 5.13 ppm signal peak was from the anomeric hydrogen of the L-furan arabinosyl. In addition, the strong signal peaks at 4.6 ppm was due to the residual solvent HDO.
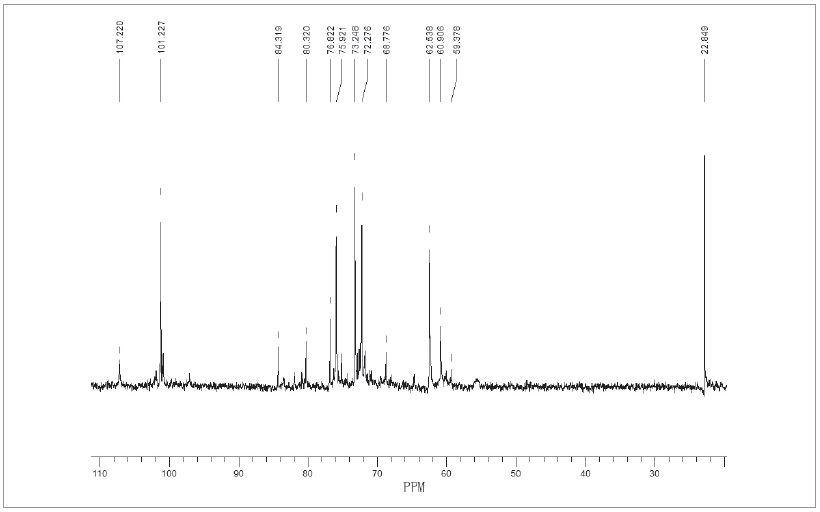
The 13C-NMR spectrum of optimum hemicellulose extracted with 1.5% KOH was shown in Fig. 5. The five strong signal peaks 101.2 ppm, 75.92 ppm, 73.2 ppm, 72.2 ppm, and 62.5 ppm were characteristic peaks of β-D-pyran xylose, corresponding to C-1, C-4, C- 3, C-2 and C-5 positions. Weak signal peaks 107.2 ppm, 84.3 ppm, 82.7 ppm, 80.3 ppm, and 60.9 ppm were C-1, C-4, C-3, C-2 and C-5 positions in the α-L-furan-type arabinosyl group, respectively. In addition, a signal peak at 59.38 ppm was derived from 4-O-methyl-glucuronic acid. From the above analysis of 1H-NMR and 13C-NMR spectra, it was found that the hemicellulose extracted with 1.5% KOH contained a certain amount of L-arabinosyl and 4-O-methylglucuronide side chains.
4. Conclusions
Hemicelluloses yields of 2.6%, 10.2%, 16.5%, 24.8%, 11.2%, and 8.7% from corn stalk were obtained in stepwise extraction process using water, KOH aqueous solution (0.6%, 1.0%, 1.5%, and 2.0%) and the mixture of KOH-ethanol as extraction solvents. Xylose in the obtained hemicelluloses were predominant component sugar, accounted for 36.8%, 78.4%, 80.9%, 90.0%, 88.2%, and 87.6% of the total sugars of Hw, H0.6, H1.0, H1.5, H2.0, and H2.5-60, respectively. Xylose content of hemicellulose obtained from water extraction process was lower and the multi-branched hemicellulose was easier to separate in water extraction process. Increase of the alkali concentration could decrease the branching degree of the obtained hemicellulose. The relatively high alkali concentration dissolve straight chain type of hemicellulose, while relatively low alkali concentration dissolved branched chain type of hemicelluloses. The content of arabinose, glucose, and galactose presented in small amounts, and mannose were identified as minimal quantities. The thermal stability of the hemicellulos obtained from 1.5% KOH extraction process was optimal compared to the other types of hemicelluloses. The optimal alkali concentration of extraction process was found to be at 1.5% KOH with the higher hemicellulose yield, xylose content and thermal stability of the hemicelluloses, at which the alkaline extraction process did not result in obvious changes in the macromolecular structure of hemicelluloses.