1. Introduction
Cellulose is the world’s most abundant green non-exhausted energy source, mainly in the form of plant fibers, in wood, grass and so on. Cellulose fibrils having diameter in the range of 1-100 nm is referred to as nanocellulose. When changing from micron to nanoscale, cellulose has many new properties, such as small size effect, macroscopic quantum tunneling effect and surface effect, low coefficient of thermal expansion (10-7 K-1),1) high Young’s modulus (138 GPa),2) high availability as a renewable material, high mechanical strength, large specific surface area and high aspect ratios, barrier properties, dimensional stability, biodegradability, and biocompatibility.3)
Cellulose nanocrystal (CNC) is one type of nanocelluloses. It has a needle-like whisker structure with a diameter of 5 to 70 nm and a length of 100 to 250 nm of a small length to diameter ratio. It has a high crystallinity, usually 60% to 90% with strong mechanical properties. The two common methods for preparing cellulose nanocrystals are inorganic acid hydrolysis and enzymatic hydrolysis. Acid hydrolysis can destroy the glycosidic bond of cellulose, thereby breaking the cellulose molecular chain, and after the amorphous zone is destroyed, the CNC with high crystallinity can be obtained.4) CNCs are obtained from a variety of renewable bioresources such as wood, cotton, bacterial cellulose, agricultural crops, and agricultural cellulosic wastes.5) Wood or cotton have been considered as the most important source of cellulosic fibers, thus, the cost of CNC is relatively high. In addition, concerns on the environment and diminishing of forest, the interest in the exploitation of non-wooden cellulosic materials is increasing.6) The ligno-cellulosic resources derived from agricultural wastes or by-products are of particular interest of preparing CNCs due to their abundance, low cost, hollow cellular structure, renewability, and biodegradability.7)
Wheat straw and corn stalk, as widely distributed agricultural resources, are largely discarded in the fields and are not adequately and effectively utilized with a series of environmental problems. Corn stalk, as a by-product of corn, is composed of approximately 32.73% cellulose,8) while wheat straw is composed of cellulose (30-40%), hemicellulose (20-35%) and lignin (15-25%).9) In China, the annual production of corn stalk has reached 400 million tons. Therefore, the utilization of corn stalk material has become a significant problem. In addition, billions of tons of wheat straws have been produced in the world, but only a small portion of them is used for animal feed, production of biogas, biofuel, extraction of hemicellulose and lignin, and isolation of cellulose, whereas most of them are burned, especially in the developing countries.3,7) The problem with burning corn stalk and wheat straw is that it poses health and environmental hazards. Therefore, it is very important to develop suitable scientific and economic method of recycling of corn stalk and wheat straw cellulose. For the value-added utilization, efforts have been made to develop environmentally-friendly composite materials using celluloses obtained from agricultural by-products. It was reported that CNCs have been isolated from wheat straw and soy hulls, banana rachis, sugar beet and potato tuber.10-14) To make CNCs from corn stalk and wheat straw more attractive for commercial end-use applications, it is significant and necessary to quantify their properties through experimental and theory analysis. However, little research has been reported about the isolation of CNCs from corn stalk and wheat straw.
The objectives of the present work were to produce CNCs from wheat straw and corn stalk by acid hydrolysis and to characterize their chemical, morphological, and thermal stability properties. Atomic force microscopy (AFM), Fourier transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), a laser diffraction analyzer and X-ray diffraction (XRD) were applied to investigate the morphology and properties of isolated CNCs.
2. Materials and Methods
2.1 Materials
Wheat straw and corn stalk were used as the raw materials for preparation of CNC. Wheat straw and corn stalk were collected, pulverized and sieved with 40-60 mesh from local farms in Jinan in 2017. All samples were air-dried and stored at room temperature. Sodium hydroxide (NaOH), hydrogen peroxide (H2O2), anthraquinone (C14H8O2, AQ), chlorine dioxide (ClO2) and sulfuric acid (H2SO4) used in this experiment were purchased from Sinopharm Chemical Reagent Co., Ltd. Fiber characteristics of wheat straw and corn stalk pulp were analyzed by fiber quality analyzer (FQA) according to ISO 16065:2007 method, the results was listed in Table 1. According to National Renewable Energy Lab (NREL) methods, the chemical composition (based on mass) of the bleached wheat straw pulp and bleached corn stalk pulp were analyzed and listed in Table 1.
2.2 CNC production
The isolation of cellulose from the two raw materials was performed by NaOH-AQ pulping (alkali dosage of 12% on pulp weight (oven dried, O.D.), AQ dosage of 0.05% on pulp weight (oven dried, O.D.), liquid ratio of 1:6, and then they were bleached by chlorine dioxide and hydrogen peroxide according to the method described by Bright et al.15) The pulps were absolutely dried by oven drying that was torn into pieces bean size before hydrolysis. CNCs were prepared by 64% concentration of sulfuric acid at acid-to-pulp ratios of 17.5, with a mechanical stirrer t at 300 rpm for 45 minutes at the temperature of 45℃. The reaction was stopped by diluting 10-fold in the suspensions. The hydrolyzed material was washed by centrifugation at 10,000 rpm at 15℃ for 15 minutes to remove acidic solution. Then the resulting suspensions were purified in a dialysis membrane having a molecular weight range of 1,200-14,000 Da by replacing the deionized water every 4 hours, until the pH of the deionized water remained constant.16) To achieve the resulting suspensions dispersion, the suspensions were sonicated for 5 min at 100% power in ice bath to avoid overheating for further used.17) Part of the suspensions was filtered on a polytetrafluoroethylene film (filter hole diameter: 0.22 μm) through vacuum, and the film was air-dried and stored at room temperature. Another part of the suspension was stored at 4-5℃ for analysis of particle size and AFM. And the yield of CNCs (in percent) relative to the initial weight of the initial cellulose sample were determined.
2.3 Particle size distribution
The particle size distribution of the CNC was determined by using a laser diffraction analyzer (Nano-ZS90, Malvern, UK). The particles were analyzed in the size range between 0.6 nm and 6 μm by this technique. A suspension of 0.01% CNC consistency was prepared, and dispersed the suspension with an ultrasound for 5 min at 100% power. Three measurements of 120s each were taken and the averaging was done and the results were an average of five replicated measurements.18)
2.4 Fourier transform infrared spectroscopy (FTIR)
The FTIR spectra for the CNC film were analyzed by a Fourier transform infrared instrument (ALPHA), with the range~4,000-500 cm-1 and a resolution~2 cm-1. The FTIR spectra were obtained from analysis of the correlation function using the OPUS/Mentor software.
2.5 Degree of crystallinity
Specimens of CNC film was X-rayed using an equipment (XRD D8-ADVANCE, Bruker, Germany) with a copper target X-ray tube, generated at a voltage of 40 kV and a filament emission of 20 mA. The data were collected in reflection mode with diffraction angle 2θ varying from 10° to 50° with a rate of 6°/min. Degree of crystallinity (%) of each sample was calculated from its X-ray diffraction pattern by Segal’s methods.18)
The crystallinity index, CrI was determined using Segal’s formula as given below.19)
Where I002 is the overall intensity of the peak at 2-theat about 22.5°, Iam is the intensity of the baseline at 2-theat about 15.7°.
2.6 Thermogravimetric analysis (TGA)
The thermal stability of the CNC film samples was characterized by using Thermo Gravimetric Analyzer (TGA Q50, TA Instruments, America). Approximately 6 mg of each sample was heated from 50℃ to 690℃ at a heating rate of 10℃/min under a nitrogen atmosphere with a gas flow of balance gas~40.0 mL/min and sample gas~60.0 mL/min in order to prevent any premature thermal oxidative degradation. The thermal stability was obtained from analysis of the weight-loss rate as a function of time.20)
2.7 Atomic force microscopy (AFM)
The size distribution and three-dimensional appearance of the CNCs were determined by AFM. Original CNC suspensions were diluted to about 1×10-3% to 1×10-5% concentration. A ~1 cm2 piece of freshly cleaved mica was placed in 0.1% w/v solution of poly-L-lysine for 3 min, and then the mica was dried. The resulting CNC suspensions was evenly kept on the mica by easy coater (EASYCOATER-6, Schwan Technology, China), and then the mica was dried. The mica was analyzed by attaching to an AFM specimen disk. Images were obtained using an AFM (Multimode8, Bruker, Germany), samples were scanned with scan sizes ranging from 5 to 10 μm. The AFM software was used to the section analysis particle size.17)
3. Results and Discussion
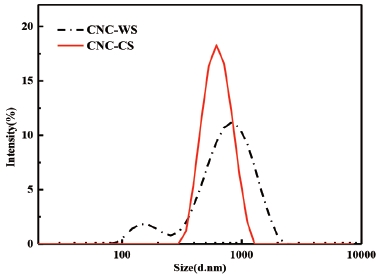
3.1 Yield and particle size distribution
The yields of CNC-WS and CNC-CS were 31.2% and 32.4%, respectively, which were similar to the conventional CNCs preparation by mineral acids (about 30%).20) And The particle size distributions of CNCs from two different materials were shown in Fig. 1. It should be noted that the Malvern Nano-ZS90 assumes spherical particles when calculating particle size. This implied that the particle size based on the size distributions should be considered to be relative.20) The mean size of CNCs from wheat straw and corn stalk were 840 nm and 630 nm, respectively. Fig. 1 showed that the particle size of the CNC-CS was more uniformity than that of the CNC-WS. In addition, a unimodal peak position of CNC-WS shifted to lowest values with a mode at ca. 149 nm which might be the diameter of CNC-WS. This implied that the CNC-WS (L/D=20.1) had a higher aspect ratio than CNC-CS (L/D=12.2) calculated from the software (Nano-Scope Analysis) analysis in AFM analysis. From these results, it was evident that CNC-CS presented a greater uniformity in size distribution than CNC-WS. One possible reason might be the relatively high lignin content of CNC-WS which hindered the penetration of sulfuric acid for hydrolysis.
3.2 FTIR analysis of CNCs
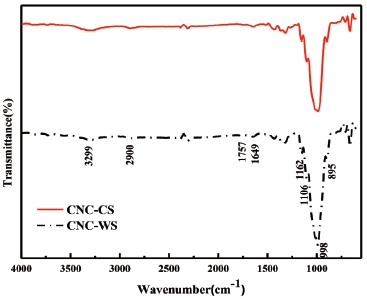
Fig. 2 illustrated that FTIR spectra of two samples were almost identical similar bands. The bands in the 3,299 cm-1 region were assigned to O-H stretching vibrations and cellulose nanocrystals was identified as due to hydrogen bonding.21) The bands at 2,900 cm-1 region were assigned to C-H stretching vibrations,22) The bands at 1,649 cm-1 region were assigned to the O-H bending of the adsorbed water, the band at 1,757 cm-1 region were assigned to C=O stretching vibrations. The deformation, wagging and twisting modes of anhydro-glucopyranose vibration was shown from 600 to 1,700 cm-1.21) The spectra also confirm the structure of cellulose I as cellulose Iβ.23) It was reported that the acid hydrolysis treatment had no effect on the chemical structure of CNCs isolated from corn stalk and wheat straw pulp, and the results showed in this work was in good agreement with previous works.24,25)
3.3 Thermogravimetric analysis (TGA)
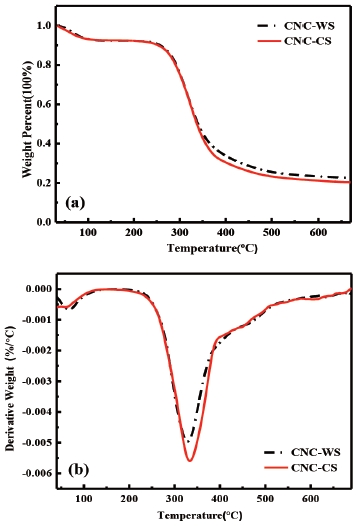
Thermal stability plays an important role in restricting the properties and application scope of polymer and CNC composites.24) Thermogravimetric (TG) and its derivatives (DTG) curves of CNCs from wheat straw and corn stalk were shown in Fig. 3(a) and Fig. 3(b). The initial small amount of weight loss observed in both wheat straw and corn stalk related to the evaporation of water loosely bound to the surfaces of cellulose at the temperature below 100℃.21,26-30) This finding was in accordance with the characteristic peak of bending vibration of intermolecular hydrogen bonding of water interaction at 1,649 cm-1 in the FTIR spectra.21) In addition, Fig. 3(a) and Fig. 3(b) showed that CNC began to decompose at the same temperature around 231℃. The thermal degradation temperature of CNC-WS and CNC-CS was lower than their pulp cellulose fibers while the bleached wheat straw pulp and bleached corn stalk pulp was 274℃ and 278℃, respectively. And two maximum decomposition temperatures could be observed in the DTG curves, CNC-WS decomposition temperature occurred around 327℃, while a higher decomposition temperature could be observed of CNC-CS which was around 333℃. Moreover, the amount of char residue at 660℃ in CNC-WS sample (22%) was larger than that of CNC-CS sample (20%). Since the functional groups were almost same in two groups, the main reason of this difference might be smaller size of CNC-CS, reflected in the particle size analysis.
3.4 XRD analysis
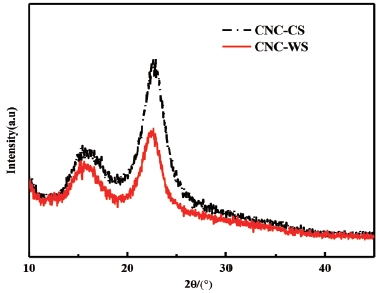
The XRD pattern of CNC-WS and CNC-CS were shown in Fig. 4, three main diffraction peaks of the crystallographic planes of monoclinic cellulose Iβ lattice at 2θ=14.8°, 16.3°, and 22.7° appeared, and this indicated that the present extracted cellulose was cellulose type I32) which was in accordance with FTIR analysis. Therefore, the degree of crystallinity of cellulose I was calculated in this work. From Table 2, the degree of crystallinity of CNC-WS and CNC-CS were 52.1% and 65.2%, respectively, which increased 57.4% and 131.2% compared with the original pulps (Table 1). These results clearly demonstrated that the crystallinity of the material progressively increased. The higher crystallinity value of CNC-CS compared to CNC-WS can be well understood by the reduction and removal of amorphous non-cellulosic compounds, such as lignin and hemicellulose induced by the bleaching, alkali and hydrolysis treatments conducted in the purification process. Thus, it seemed that the crystallinity value was depending on different types of plants and the duration of fibers purification and hydrolysis process. Higher crystallinity in the chemical treated fibers is interrelated with higher tensile strength of the fibers.33,34)
3.5 AFM Analysis
To get an accurate knowledge about the dimension scale of the CNC samples, AFM analysis was carried out. Fig. 5 showed AFM micrographs of CNC-WS (a-1, a-2) and CNC-CS (b-1, b-2). Fig. 5 (a-2) and Fig. 5(b-2) were from the elliptical position in Fig. 5(a-1) and Fig. 5(b-1), respectively. It could be clearly seen from the Fig. 5 that the CNC-WS and CNC-CS were both rod-shaped.31) According to software (NanoScope Analysis) analysis, CNC-WS had an average diameter of 41.74±15.3 nm and length of 836.62±87.2 nm (L/D=20.1), while CNC-CS had an average diameter of 51.15±9.4 nm and length of 624.13±54.3 nm (L/D=12.2), and this made CNCs facilitate the stress transfer from the matrix to the fibers for any significant reinforcement to occur.4) And these results were basically consistent with the particle size distributions.
4. Conclusions
CNCs were successfully extracted and isolated from two types of agricultural waste (wheat straw and corn stalk) using the sulfuric acid hydrolysis treatment. The yield of CNC-WS and CNC-CS were 31.2% and 32.4%, respectively. FTIR indicated acid hydrolysis treatment had no effect on the chemical structure of CNCs isolated from corn stalk and wheat straw pulp. The crystallinity of CNC-CS (65%) was higher than that of CNC-WS (52%). Particle size distribution and AFM images showed that CNC-CS had a smaller size with more uniformity than CNC-WS. The decomposition of CNC-CS started around 231℃, which showed a similar thermal stability with CNC-WS. Our experiments had revealed that corn stalks and wheat straws can be used as starting materials for the preparation of cellulose nanocrystals.