1. Introduction
The Earth’s natural environment constantly changes; in recent years, the change has been both rapid and artificial, which is cause for concern. To stabilize the global ecosystem, fossil fuels should be replaced with environmentally friendly resources. In particular, the pulp and paper industry is focusing on developing new materials from woody biomasses. Wood is an abundant biomass resource, but its complex chemical structure makes its direct use as a raw material problematic.1)
Thus, wood must be converted before it can be used as a resource. The thermochemical method of conversion, which includes gasification, pyrolysis, supercritical fluid extraction, and liquefaction, has received the most attention among the various transformation methods. During wood liquefaction, the wall components of cells are dissolved with a solvent in a depolymerization process. The liquefaction of woody biomasses has been attempted using phenol and glycol at high temperatures.2)
Generally, there are two main methods of wood liquefaction: one uses high temperature and pressure without the use of a catalyst, and the other uses a lower temperature with an acid catalyst.3) Additionally, the wood liquefaction products are renewable alternative resources for foam or adhesive components.4,5) Currently, research is underway to develop various new materials from woody biomasses, such as nanofiber and carbon fiber. Furthermore, microwaves can be used to reduce the wood liquefaction time. Aguilar-Reynosa et al. reported that the reaction of wood powder with microwaves resulted in liquefied wood containing cellulose crystals.6) Wood liquefaction using microwaves is a rapid and effective method because heat energy is generated from within the wood by the molecular action of water and a solvent. Moreover, microwaves can be applied efficiently and uniformly while the reaction time is shorter than that of other methods. Therefore, a short reaction time and the use of less solvent is considered advantageous.7-9)
In spite of many studies on efficient wood liquefaction, the reaction mechanism of wood liquefaction remains completely unknown. However, it is suggested that the cellulose and lignin are degraded by the catalyst, while the low molecular weight components and hemicellulose are directly soluble.10) Additionally, it is known that lignin plays an important role in the re-condensation process that forms insoluble precipitates.11) Mašek et al.12) reported that the use of microwaves can rapidly heat the interior of the reaction mixture, resulting in a rapid reaction. Kržan and Kunaver13) also found that the liquefaction of wood is possible using microwaves as a heat source. Moreover, it was found that wood can be completely liquefied within 20 minutes when phosphoric acid is used as a catalyst, instead of simple glycol and organic acid anhydrides, and that liquefaction provides a higher yield with higher microwave intensity, longer projection time, and higher phosphoric acid concentration.
Wood liquefaction studies have revealed that the extent of the process is determined by some of the unfiltered residues that remain when the reaction products are filtered through a glass filter. Most of these remaining residues consist of very fine wood fragments. Thus, by controlling the reaction conditions for wood liquefaction, fine wood particles can be obtained, while lignin is removed from the wood.9,14) Furthermore, the fine (microscale) wood fragments obtained from the liquefied wood powder are very soluble in various cellulose solvents, even though they may contain quantities of lignin.15) In a previous study, the authors conducted microwave- assisted organosolvation of wood in glycol ether using microwave irradiation under various conditions and compared the characteristics of the micronized wood residue after the reaction.9) In comparison with thermal pyrolysis, the advantage of the microwave-assisted organosolv process is that the reaction time can be reduced from several hours to several minutes, because the microwave penetrates the core of the sample and simultaneously generates heat internally.16,17)
The realization of a cellulose material containing lignin that can be easily dissolved in an alkaline solvent in a comparatively simple and economical manner could contribute to the breakthrough development of a new cellulose-based material industry. The possibility of recycling the organic solvent used in the organosolv process for the production of cellulose-based materials will be an important factor in improving the economic efficiency of the process.
The purpose of this study was to investigate the changes of the chemical composition of lingocellulosic micro-fines (LCMFs) with successive microwave-assisted organosolv process of pinewood by reusing solvents.
2. Materials and Methods
2.1 Materials
This investigation used Korean pinewood powder, and its characteristics are presented in Table 1. Glycol ether (pure grade, Sigma Aldrich Co.) was used as a solvent for the microwave-assisted organosolv process. The method used a mixture of glycol ether and sulfuric acid (95%, extra-pure grade, Daejung Chemicals & Metals Co., Korea) in a ratio of 96:4 (v/v) of glycol ether to acid.
2.2 Microwave-assisted organosolv process
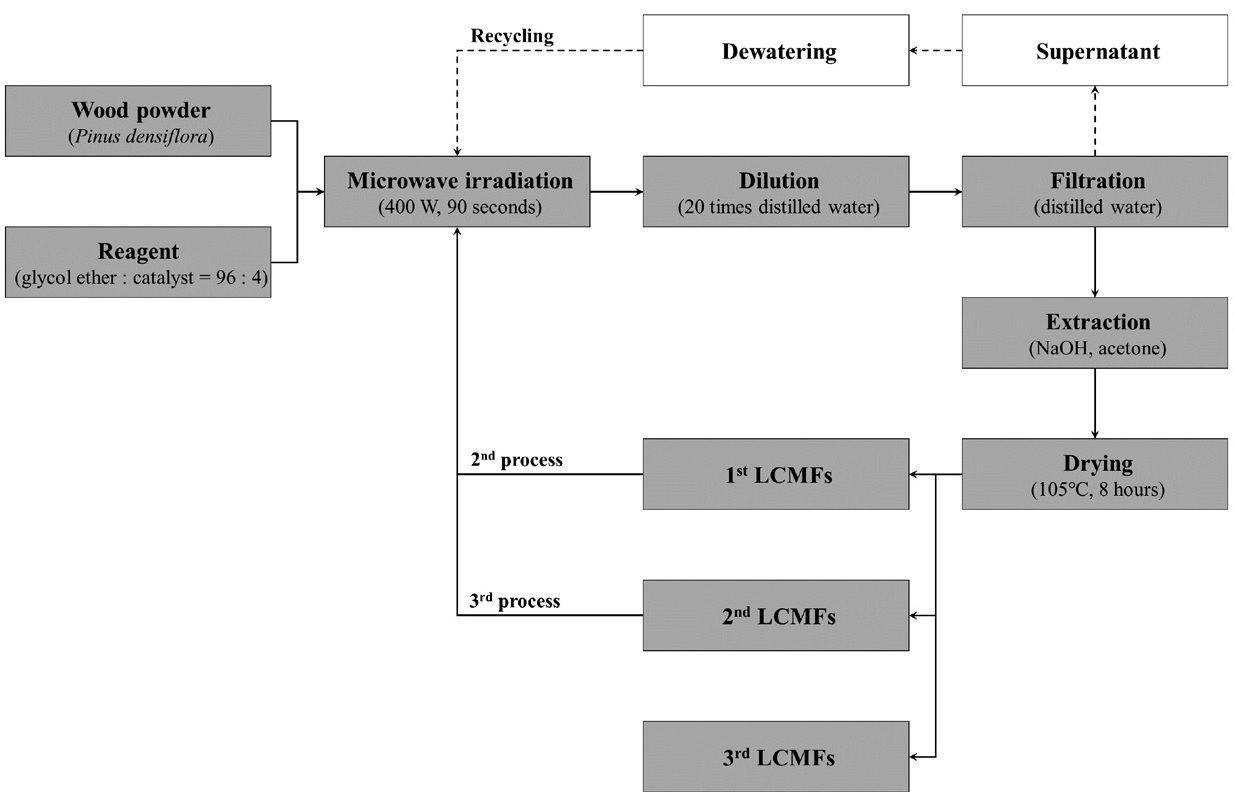
A scheme of the microwave-assisted organosolv process is shown in Fig. 1. The conditions of the organosolv process used in this study were based on data reviewed in a previous paper.18) A total of 45 g of pinewood powder and 90 g of solvent were mixed inside a 300 mL Erlenmeyer flask. The samples were reacted within a microwave oven (400 W, SE-MW 50018, Sun Enterprise, Korea) for 90 seconds. Every 10 seconds, the flask was taken out and stirred well with a Vortex mixer (G-560, Scientific Industries Inc., USA). After 90 seconds, the reaction was terminated and the reaction mixture was poured into 1 L of distilled water, stirred well and allowed to stand. After the wood product was sufficiently precipitated, the precipitate was collected by paper filtration. The supernatant was dewatered with a rotary evaporator and the dewatered solvent was used for the successive microwave organosolv processes. The residue was sequentially extracted with 1 L of 0.5 N aqueous NaOH solution and 300 mL of acetone. After extraction, the residue was dried in a drying oven at 105±2℃ for more than 8 hours to obtain the first LCMFs. The second and third LCMFs were obtained through successive process.
2.3 Measurements
2.3.1 Total crystallinity index
Attenuated total reflectance infrared (ATR-IR) spectroscopy (Alpha-P model, Bruker Optics, Germany) was used to identify the chemical properties of the LCMFs. The spectra were recorded between 4,000-400 cm-1 and one spectrum was measured as an average value of 16 repeated measurements per sample. The crystallinity index of LCMFs was determined using the method described by Ṧiroký et al.,19) which divides 1,327 cm-1 (C-H bending in cellulose) by 2,900 cm-1 (methylene groups in cellulose), as follows:
2.3.2 Yield and lignin contents of LCMFs
The yield of LCMFs obtained by each successive process was based on the dry weight. The residual lignin content in each LCMFs, including in the extracts, was determined using the 72% sulfuric acid method (Klason lignin, KL) based on the TAPPI Standard (T 222).
2.3.3 Morphology
An optical microscope (BX50, Olympus Optical Co., Ltd., Japan) at ×60 magnification was used to observe the morphology of the LCMFs. The LCMF particle size was calculated from the surface area of the particles, which were measured with an image analyzer (i-Solution, IMT Technologies, Canada).
2.3.4 Neutral sugar analysis
The alditol-acetate method20,21) was utilized to determine the neutral sugar composition of the LCMFs obtained by successive microwave-assisted organosolv processes. Alditol-acetate was dissolved in 2 mL of acetone and the neutral sugar composition was analyzed by gas chromatography (GC, HP-6890, Agilent, USA). Table 2 shows GC conditions for neutral sugar analysis.
Table 2.
GC condition for alkaline nitrobenzene oxidation and neutral sugar analysis
2.3.5 Alkaline nitrobenzene oxidation
A sample equivalent to 10 mg of lignin was weighed and mixed with 0.24 mL of nitrobenzene and 4 mL of 2 N aqueous potassium hydroxide solution. Then, the mixture was sealed in a 10 mL stainless bomb and reacted for 2 hours in a shaking oil bath at 160℃. The reaction mixtures were described by Billa et al.22) and Huang et al.23) The resulting alkaline nitrobenzene oxide of LCMFs was analyzed by GC from the trimethylsilylate of the aromatic aldehydes.
3. Results and Discussion
3.1 Yield and residual lignin contents of LCMFs
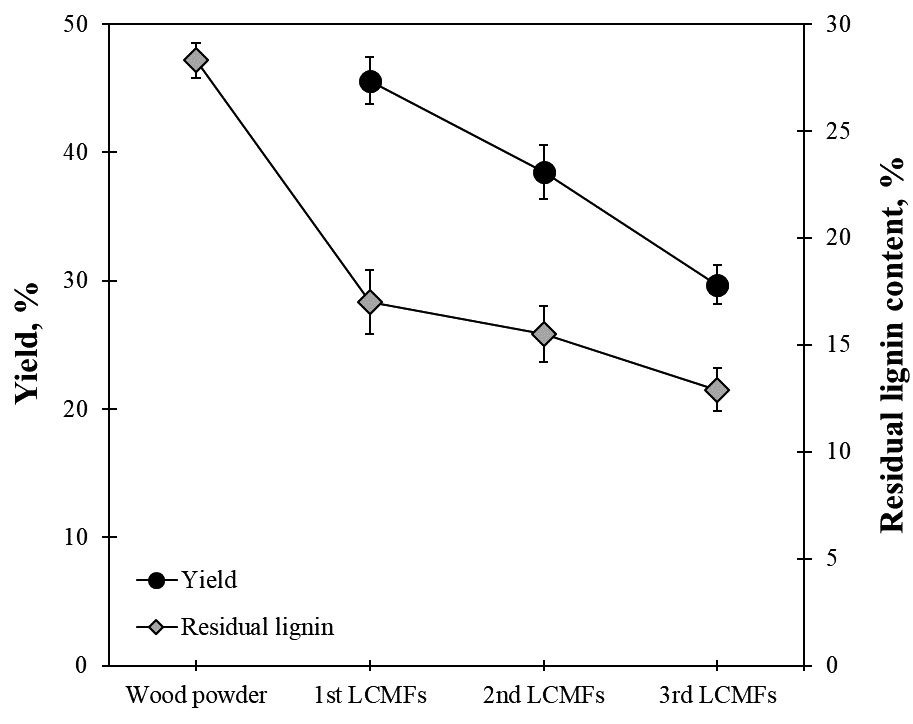
The yield and residual lignin content of LCMFs obtained through successive microwave-assisted organosolv processes are shown in Fig. 2. The yield from the first microwave-assisted organosolv process decreased sharply while the residual lignin content of the first LCMFs reduced by approximately 40% compared to the wood powder. This is because most of the components in the wood were decomposed and eluted by the first microwave-assisted organosolv process. In the second and third microwave-assisted organosolv processes, the residues became more resistant to the reaction and thus, the microwave organosolv effect was insignificant, resulting in a relatively high yield of approximately 80%. Furthermore, the remaining lignin content of successive organosolved LCMFs was also reduced by approximately 10-16%, which was less than that of the first LCMFs.
3.2 Morphological properties of LCMFs
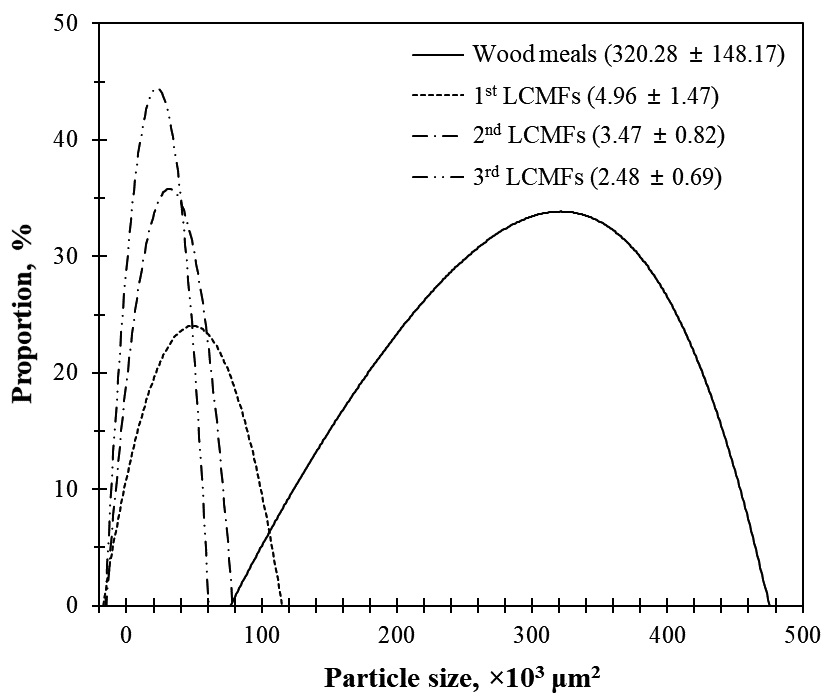
Fig. 3 presents the optical microscope image of LCMFs obtained by successive microwave-assisted organosolv processes. The particle size of LCMFs was significantly reduced compared with that of the wood powder because the sulfuric acid catalyst used in the microwave-assisted organosolv process penetrated into the wood and decomposed it. As shown in Fig. 4, it can be seen that the particle size decreased drastically during the first microwave- assisted organosolv process by producing wood fragments of 49.61×103 µm2. Furthermore, after the second and third microwave-assisted organosolv processes, the LCMFs of 49.61×103 µm2 was changed to smaller particle sizes (34.72 and 24.79×103 µm2, respectively). This agrees with the outcomes of a previous study on chemical dispersion using microwave.9) However, the change in particle size of LCMFs was not significant during successive microwave-assisted organosolv processes. This indicates that even a single microwave-assisted organosolv process is sufficient to obtain micronized residues of a sufficiently small size.
3.3 Total crystallinity index of wood powder and LCMFs
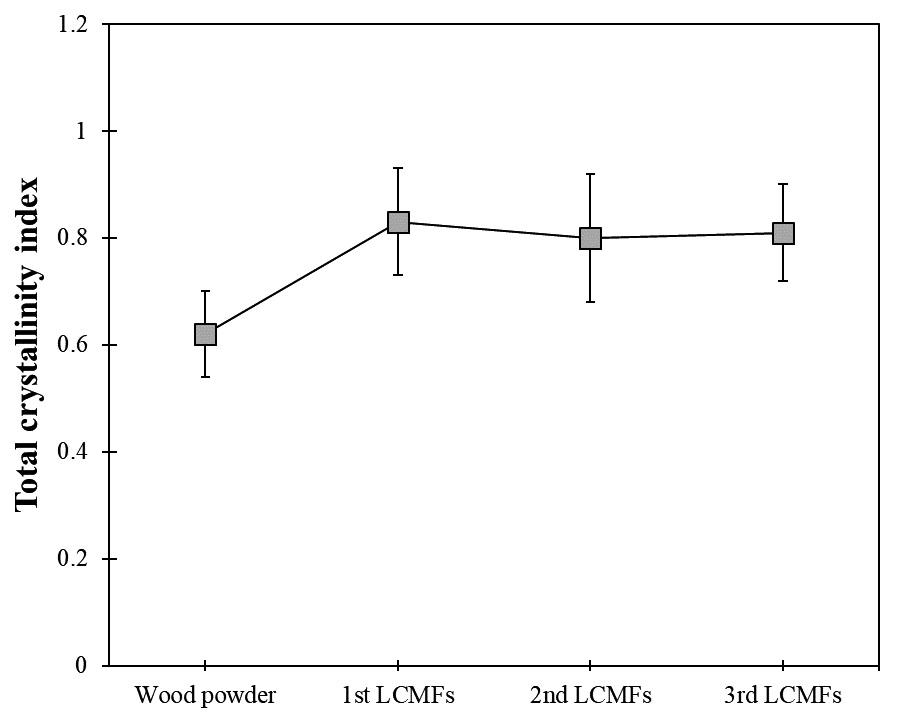
Fig. 5 shows the crystallization index values calculated from the IR spectra.19) The crystallinity index of the LCMFs was slightly higher than that of the wood powder, which is attributed to the destruction of the LCMFs’s amorphous regions during the microwave-assisted organosolv process. However, the crystallinity indices between the various LCMFs were similar, as most of the amorphous regions in the wood powder were destroyed by the first microwave-assisted organosolv process. Additionally, there is a possibility that part of the crystal structure was changed during the organosolv process. It is presumed that the LCMFs obtained via the first microwave-assisted organosolv process were highly resistant to the successive organosolv processes.
3.4 Neutral sugar compositions of LCMFs
Table 3 shows the relative neutral sugar compositions of the LCMFs obtained with successive microwave-assisted organosolv processes. It should be noted that the relative neutral sugar composition of pinewood powder was similar to that of other softwoods. Furthermore, hemicellulose was easily degraded by the microwave organosolv process and thus the total amount of monosaccharides constituting hemicellulose was greatly reduced in the first LCMFs. Kim et al.24) suggested that, in comparison with wood powder, the low neutral sugar content of the first LCMFs was attributed to the oxidative decomposition of hemicellulose by the sulfuric acid catalyst during the microwave-assisted organosolv process. Moreover, the monosaccharide content of the second and third organosolved LCMFs was lower than those of the first LCMFs because most hemicellulose elution occurred during the first organosolv process, while elution was reduced in subsequent processes. Furthermore, the glucose content in LCMFs increased with the number of microwave-assisted organosolv processes, which means that the decomposition of hemicellulose occurred predominantly while degradation of cellulose was not greatly affected by the microwave organosolv process.
Table 3.
Relative neutral sugar compositions in LCMFs by successive microwave-assisted organosolv process
3.5 Alkaline nitrobenzene oxide of LCMFs
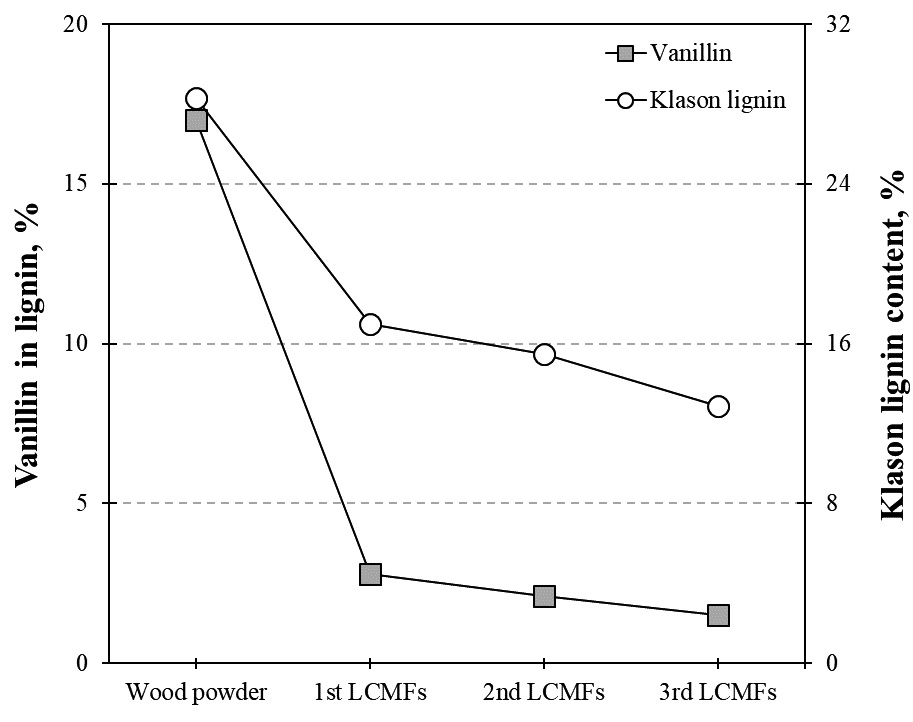
The results of alkaline nitrobenzene oxidation of wood powder and LCMFs are shown in Fig. 6. The KL content of pinewood was 28.3% while the vanillin content of lignin was 17.0%. The first organosolved LCMFs had a KL content of 17.0% while the vanillin content of lignin is approximately 3%. This means that the lignin and vanillin content is significantly reduced by the first microwave-assisted organosolv process, and this results were consistent with that of Kim et al.24) Therefore, it is suggested that the lignin was eluted and decomposed by the sulfuric acid catalyst used for the microwave-assisted organosolv process. At this stage, both the low molecular weight and the condensation of lignin contributed to the decrease of the β-O-4 bond.25,26) However, the KL and vanillin contents of the second and third LCMFs only decreased slightly with successive microwave-assisted organosolv processes, which indicates that the first organosolved LCMFs cannot be expected to undergo dramatic decomposition by successive organosolv processes, as the results of the IR spectra analysis described above (Fig. 5).
4. Conclusions
This study compared the chemical characteristics of LCMFs that were subjected to successive microwave- assisted organosolv processes using a glycol ether. Significant decreases were observed in both the yield and KL content of the LCMFs after the first microwave-assisted organosolv process. However, the KL content of the LCMFs that was subjected to successive organosolv processes decreased only slightly compared with that of the organosolved LCMFs from the first process. A dramatic decrease in the particle size of the LCMFs occurred after the first microwave-assisted organosolv process, but there was no considerable change with successive processes. The chemical composition of the LCMFs was characterized by the elution of most of the hemicellulose and lignin contents during the first microwave-assisted organosolv process. Moreover, it was found that the first reaction condensed a large amount of lignin. Additionally, the content of monosaccharides constituting hemicellulose decreased considerably during the microwave-assisted organosolv process, whereas the crystallinity index of the first LCMFs increased. However, the crystallinity index, vanillin content, and monosaccharide content of the LCMFs remained comparable with each other in the subsequent repetitions of the microwave-assisted organosolv process.