1. Introduction
Since phosphorus does not exist as a gas in the natural world, it is easily precipitated, its solubility is low except for calcium salt in phosphate, it is a substance that accumulates in soil and is difficult to circulate because it is too small in diffuse into and migrat soil. It is always separated from water in the form of a precipitate because of difficulty in converting to the gaseous state. For this reason, it is absorbed by microorganisms as a nutrient and is chemically or physically bound to the flocculant during the chemical process of flocculation. However, there is a limit to the amount of phosphorus that can be absorbed by microorganisms during biological treatment, therefore, it is common to see a concentration of approximately 1 mg/L as a phosphorus concentration that can be achieved in biological processes.1) Therefore, a combination of biological treatment and chemical treatment is required for high-efficiency inhalation such as that required for total dose.2)
The chemical treatment is simple addition facility to the existing sewage treatment facility. The chemical treatment has the advantage of being able to remove phosphorus with high efficiency, and it actively cope with sudden influx of water quality. Aluminum and iron salts, which are inexpensive and effective for improving water quality, are widely used as coagulants used in water treatment. Increasing use of flocculants due to increased contamination of the water source causes secondary pollution problems such as formation of coagulated sediments.3-5) Aluminum absorbed in the human body through the consumption of contaminated water causes neuropathologic disorders such as Alzheimer’s disease, presenile dementia, and speech disorders.6-9) Activated carbon is the most widely used absorbent to reduce water contamination.
Activated carbon is manufactured through an activation process using charcoal heated steam, etc. It has a large specific surface area and is excellent for adsorption,10-11) however, it is difficult to use it in large quantities because of its high price. Therefore, in this study, semi-carbonization treatment of forest wastes using high heat treatment and surface charge modification using cationic polymer electrolyte (generally used as a retention enhancer in disciples industry) were employed. The characteristics of the adsorbent using adsorption principle were investigated and the possibility of its use as an adsorbent was verified.
2. Materials and methods
2.1 Materials
2.1.1 Wastewater
Wastewater samples were collected from an inlet in a N wastewater treatment plant in City B. The analysis of the supernatant water quality of the samples was conducted by using a polymer mixture containing torrefied wood for 30 minutes.
2.1.2 Torrefied wood powder (TWP)
Torrefied wood powder (TWP) used as the disclosure material was prepared from wood chips with a moisture content of less than 12%, and the torrefaction process was carried out using a self-developed superheated steam process at a temperature of 250℃ for 15 minutes. After finishing and grinding, TWF of 75-106 μm was used as the raw material.
2.2 Experiment methods
2.2.1 Preparation of a sample
The TWP used in this study had a moisture content of 6%. The TWP was surface-modified with C-PAM, and a cationic monomer aqueous solution used was stirred at a concentration of 0.2%. TWP was added to the monomer aqueous solution so that the mixing ratio of C-PAM and TWP was 1 wt%, 5 wt%, and 10 wt%, respectively.
2.2.2 Sample analysis
The beaker was filled with 0.5 L of the sample, and the coagulant was added. The mixture was stirred in a jar tester at 200 rpm for 1 minute, and then precipitated for 30 minutes; only the, supernatant was collected. COD, TP, TN, and turbidity of the specimen were analyzed according to the standard method.12) The experiment was repeated three times.
3. Results and Discussion
3.1 Analysis of target wastewater characteristics
Table 1 shows the water quality of the sewage used in the experiment. The pH of water samples remained steady at a value of 7.6 during the experimental period. The recorede contentrations for NH3 were 14.9 mg/L, 2.2 mg/L for PO4-P, 3.5 mg/L for TP and 29.8 mg/L for TN.
3.2 Comparative study of reduction efficiency of turbidity using TWP
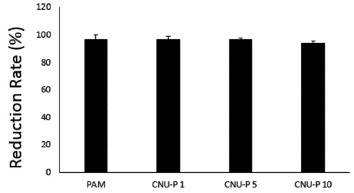
The coagulation experiment was carried out using the influent of the stream as the raw water sample. As a result of the preliminary experiment, the flocculation effect was remarkable even when only a small amount of TWP flocculant was used, and an amount of TWP flocculant added was 0.5 mg/L. Fig. 1. shows the turbidity reduction rate. When c-PAM containing 1% TWP was added, the turbidity reduction rate was 96.63%, and the reduction efficiencies were 96.60% and 93.94% for 5% TWP and 10% TWP, respectively. The turbidity is lowered in treated water containing 10% TWP due to the characteristics of TWP post the torrefaction process.
3.3 Comparative study on the reduction efficiency of COD using TWP
The coagulant is used to remove colloidal particles ranging from 1 μm to 1 nm in size, which hardly sediment even after a long period of time. The coagulant is injected into the floc to form colloidal particles with large flocs and then separated by precipitation or filtration.
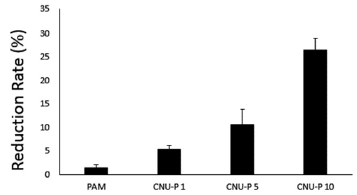
Fig. 2. shows the COD reduction rate according to the amount of TWP added to the coagulant. COD showed a reduction rate of 26.58% and 1.33% in 10% TWP and conventional PAM, respectively, demonstrating a high reduction efficiency when TWP was added.
3.4 Comparative study on the reduction efficiency of TN using TWP
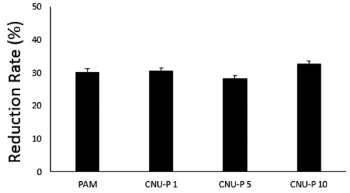
Fig. 3. shows the TN reduction rate according to TWP. TN showed a reduction rate of 32.53% with 10% TWP at an initial concentration of 29.8 mg/L. However, there was no change in the reduction rate when compared to the TN reduction rate of 29.90% for C-PAM. The results of these preliminary analyses show that industrial wastewater has a high level of coagulant use and a low throughput compared to conventional wastewater, which, in turn, is closely related to the concentration of organic matter and minerals. Denitrification is a reaction in which nitrate nitrogen (NO3-), which has undergone nitrification process, is used as a hydrogen acceptor in an anaerobic or anoxic state and is released into the atmosphere as nitrogen gas. At this time, since the nitrification process is aerobic, oxygen is used as an energy source, but carbon is required as an energy source because denitrification is performed in an anoxic state. Therefore, when denitrifying biological treatment wastewater or biological wastewater under biological treatment through sewage treatment plant, it is necessary to supply an energy source such as methanol because it lacks organic carbon. In the denitrification reaction, various organic carbon containing substances such as methanol, ethanol, acetate, sugar, and glucose are used as energy sources.13)
3.5 Comparative study of reduction efficiency of TP using TWP
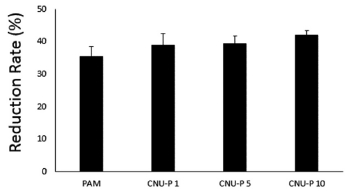
Fig. 4. shows the TP rate according to the TWP content. TP showed a reduction rate of 41.96% at 10% TWP.
Sewage treatment using the conventional flocculant was conducted by using the coagulant of iron salt or aluminum salt alone and the flocculant aid or the metal salt alone. When the iron salt is used as the flocculant, the reduction effect on organic matter in wastewater is relatively high however, there is a problem with regard to wastewater being colored by the residual iron salt after treatment.14) Such a problem results in an additional reprocessing cost when the wastewater is to be recycled after treatment. In addition, when an aluminum salt is used as a coagulant, the amount of chemical used to adjust the pH to neutral can be reduced in the post-treatment process, however, the reduction efficiency of the organic material is low.15) Such an inorganic coagulant has a low coagulation efficiency when a large amount of wastewater is present, thus, requiring a large amount of coagulant to be added, and has problems pertaining to reprocessing and high corrosiveness.16) However, the TWP-containing coagulants used in this study are inexpensive and environmentally friendly. In addition, it is possible to coagulate small colloidal particles in the form of physico-chemical coagulation, which can reduce the processing cost without polluting sewage.
4. Conclusions
The coagulation of water is implemented with the aim to agglomerate fine particles and colloids into larger particles for reducing turbidity, natural organic matter as well as other soluble organic and inorganic pollutants in the wastewater.17) This process comprises two distinct stages: rapid mixing of dispersed coagulant into water/wastewater to be treated via violent agitation and flocculation for agglomeration of small particles into well-defined flocs via gentle agitation.18) Finally, the flocs are allowed to settle and then removed as sludge while the treated water/wastewater (supernatant) is transferred into subsequent treatment process or for discharge into a watercourse. Owing to its easy operation, relatively simple design, and low energy consumption, coagulation has been successfully employed in different types of industries.19,20) Moreover, coagulation can be used as a pretreatment, posttreatment, or even as the main treatment of wastewater because of the versatility of the treatment process. This study aimed to investigate the coagulation characteristics and applicability of contaminant treatment by injecting coagulant-containing wood powder through a torrefaction process.21,22) We observed that the higher the TWP content, the better the reduction efficiency, because the influent and flocculant combined well and the floc was not broken down easily. Thus, in this study, TWP-based natural polymer flocculants did not leave any harmful substances such as aluminum in the treated water compared to conventional coagulants such as PAC and Alum. Moreover, these TWP-based natural polymer flocculants, and are highly biodegradable and effective for coagulation of various colloidal floats, sand should be considered as suitable flocculants.