1. Introduction
Cellulose fibers, known as pulp, have been dominantly used for producing disposable hygiene products in the health care industry. The products are classified into the two groups: 1) hygiene papers and 2) disposable absorbents. Bathroom tissue, facial- tissue, napkins and paper towel belong to the first group whereas baby diapers, sanitary napkins and incontinence pads belong to the second group. Hygiene products require good softness, strength, and absorbency, though each product may place more emphasis on different attributes. Each user may also have different expectations and demands even regarding the same product. For example, for bathroom, one user might prefer more softness as a priority over absorbency while the opposite may be true for another user.1)
The objective of this paper is to discuss the fundamental absorbency mechanisms for a hygiene paper. The hope is that it can be used as a guideline in designing a hygiene paper which displays excellent absorbency performance. Additionally, this paper suggests ways of increasing the in-use absorbency capacity to the potential full capacity. A hygiene paper is characterized by having a low density (approximately 0.1 g/cm3) and a low basis weight (BW) (approximately 20-40 g/m2). Due to such low density and basis weight, a hygiene paper has structure and properties which are quite different from conventional paper products such as printing paper, newspapers, and boxes which have much higher density and basis weight than those of hygiene paper. A hygiene paper forms a network structure and behaves very much like nonwovens in many ways.2) Regarding the absorbency of hygiene paper, a lower density is preferred since density has an inverse relationship to absorbency capacity. The inverse of density is referred to as “bulk”. If a hygiene paper has a density of 0.1 g/ cm3, its bulk becomes 10 cm3/g, suggesting that the paper should have a theoretical absorbency capacity of 10 g-water/g.3)
In the real world, however, a hygiene paper is disposed well before it reaches this theoretical capacity because the time during which it is used is extremely short, typically only lasting a few seconds. In short, the potential absorbency capacity is seldom realized due to its limited time of absorption. Thus, the in-use capacity at the time of disposal is less than the potential full absorbency capacity.
2. Fundamental absorbency mechanisms
For a hygiene paper to have good absorbency, it requires a large absorbency capacity with a fast speed of absorption. Absorbing water by hygiene paper consists of two steps: The first step is to pick up water from the surface. The second step is to transport the water from the surface into the internal pores (or capillaries).
2.1 Fiber wettability
Fiber wettability is one of the fundamental factors in influencing absorbency of a hygiene paper. It is also important to determine the printability and coating on the substrate. In determining the fiber wettability, the contact angle measurement techniques have been commonly applied.4-6) The principle of contact angle measurement is based on Young-Dupre’ equation.6-8) The Young-Dupre’ equation is shown in Eq. (1).
where, σs = the surface free energy of the solid in equilibrium with liquid vapor;
σLS = the interfacial free energy between the liquid and the solid at the interface;
σLS = the interfacial free energy between the liquid and the solid at the interface;
σLV = the surface tension of liquid; and
θ = the contact angle of the liquid on the solid surface.
The following assumptions have been applied in deriving Eq. 1.
1) The system is in an equilibrium state;
2) The solid surfaces are smooth, immobile, and rigid (i.e., no structural change occurs); and
3) The shape of the liquid drop is spherical and the liquid drop size is small enough such that gravitational forces can be ignored.
Depending on the contact angle of water on the solid surface, its wettability may be classified into four cases.
Case 1: θ = 0°, cos(θ) = 1: completely wettable (wet out),
Case 2: 0° < θ < 90°, 0 < cos(θ) < 1: wettable,
Case 3: 90° < θ < 180°, -1 < cos(θ) < 0: non-wettable,
Case 4: θ = 180°, then cos(θ) = -1: completely non-wettable.
For a hygiene paper, the lower contact angle should be more preferable for fast wicking, thus θ = 0° is most preferable.
2.2 Contact angle measurement techniques
Three techniques have been commonly used for contact angle measurements.
2.2.1 Optical method
The most common method of measuring contact angles is optically with a goniometer.9-11) This angle is measured by the use of movable cross hairs in an eye piece of a microscope. By the use of a syringe, liquid is added or withdrawn from the solid to obtain an advancing or receding contact angle.
2.2.2 The Wilhelmy plate technique
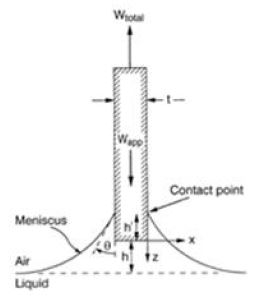
In this method, the plate is submerged perpendicular to the interface, and the force exerted on it is measured using micro-force balance. Then, the contact angle is calculated from the Wilhelmy equation (Eq. 2).
where, F= the force on the plate,
This method has been intensively used by John Berg and Kevin Hodgson at the University of Washington to determine the contact angles of individual cellulose fibers.7,12,13)Fig. 1 shows the Wilhelmy plate technique.
It is noted that the Wilhelmy plate technique can also measure both the advancing and receding contact angle on individual fibers by allowing the liquid to advance up and down on the fiber surface. It is observed that they do not provide the same value, with the advancing contact angles usually being greater than the receding angles. The difference between the two angles is referred to as the contact angle hysteresis.13,14)
2.2.3 High speed videotaping/image analysis technique
This is a very powerful technique to determine the dynamic contact angles of liquids on the substrates. This technique is especially useful for developing a high speed printing and coating system, as well as to characterize incipient wicking or spreading of a liquid onto the surface.
2.3 Absorbency mechanism
Absorbency of a hygiene paper may be characterized by a 2-dimensional capillary flow through the porous media since its thickness is much shorter compared with the length and width.
2.3.1 Laplace equation: Capillary flow
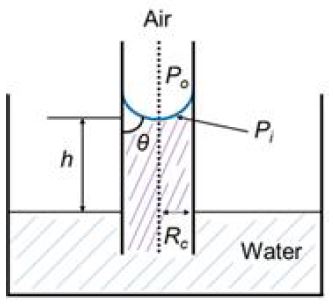
The driving force that moves the liquid from the surface into the inside in the paper is the capillary pressure which is the differential pressure across the curved meniscus of the liquid air interface in a capillary. The magnitude of the capillary pressure for an idealized cylindrical capillary tube is given by the Laplace equation (Eq. 3).6)
where, Pc = ΔP = the capillary pressure,
Fig. 2 shows the idealized simple cylindrical capillary.
Po = the pressure at the outside meniscus (the atmosphere pressure),
Pi = the pressure at the inside meniscus,
Pc = Po – Pi = the capillary pressure = 2σ cos(θ) /Rc,
σ = the surface tension of water,
In the figure, Pc = Po – Pi = ρ·g·h
where, ρ = density of a liquid,
Likewise, the capillary pressure between the two points, 1 and 2, inside the capillary may be given by
where, PC1 = P0 – P1; PC2 = P0 – P2.
Therefore,
Thus, simply ΔP12 indicates the pressure difference between position 2 and position 1. From Eq. 4, when ΔP12 > 0, then water moves from position 2 to position 1. On the other hand, ΔP12 < 0, water would move from the position 1 to the position 2.
From Eq. 3,
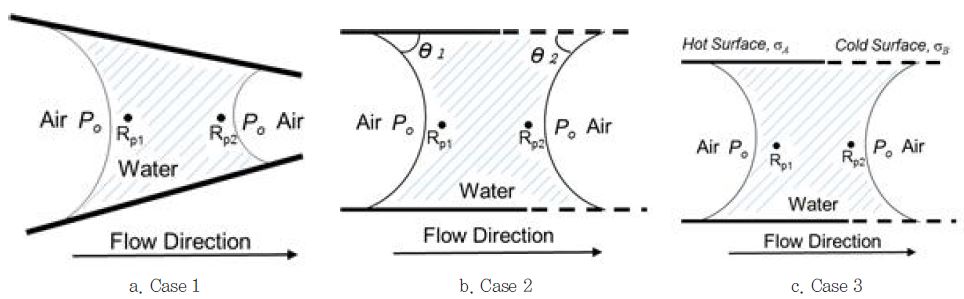
In Eq. 5, the subscripts, 1 and 2, represent the values at positions 1 and 2, respectively. For a liquid to move from one position to another, a force (or energy) difference is necessary. Such a difference may be referred to as the gradient. Ko has illustrated the three cases of generating the pressure gradients and has named them capillary flow/pumping.15)
Case 1: Different pore width, RP1 > RP2
In Eq. 3, when the same liquid is used and the contact angle is the same, then Eq. 3 simplifies to:
where, σA = σB = σ and θA = θB
If RP1 > RP2, ΔP12 < 0, therefore the flow will move from position 1 to position 2. Thus, the liquid will flow from the larger pores to the smaller pores, as shown in Fig. 3-a.
Case 2: Different contact angles, θ1 > θ2
If all other variables are the same (i.e., RP1 = RP2 = RP and σA = σB= σ) then Eq. 3 reduces to:
If cos(θ2) > cos(θ1), then ΔP12 > 0, so the water will move from position 2 to position 1. Since ΔP12 > 0 if θ1 > θ2 when 0° < θ < 90°, the water will move from the positon of the larger contact angle to that of the lower contact angle as shown in Fig. 3-b. Meanwhile the flow direction will be reversed when 90° < θ < 180° for a non-wetting liquid.
Case 3: Different surface tensions σA < σB < 90°
If it is assumed that Rp1 = Rp2 = Rp, and θ1 = θ2= θ.
Then Eq. 3 reduces to:
When σ1 < σ2, then ΔP12 > 0. So, the flow direction will be from the position of the lower surface tension region (1) to the higher surface tension region (2) as shown in Fig. 3-c.
2.3.2 Lucas-Washburn Theory
In characterizing the flow through the porous media, the Lucas-Washburn theory has been commonly used.16-18) For the horizontal wicking in a single capillary, the Lucas-Washburn equation may be expressed as follows.
or
where, l = the penetration length,
Rp = the pore radius,
σLV = the surface tension of a liquid,
θ = the contact angle,
μ = the viscosity of the liquid,
and t = absorbency time.
In deriving Eq. 9, the following assumptions have been made: A) Simple, cylindrical capillary, B) No capillary structure changes during water absorption, C) No contact angle changes during water absorption.
Eq. 9b is simplified into:
Then, the flow amount, Q, up to time, t, can be determined by multiplying l, the penetrating length with the cross-sectional area of the capillary, i.e., πRp2.
Thus, from Eq. 9b,
or
If we let K= πRp2*k, then Eq. 13 simplifies to:
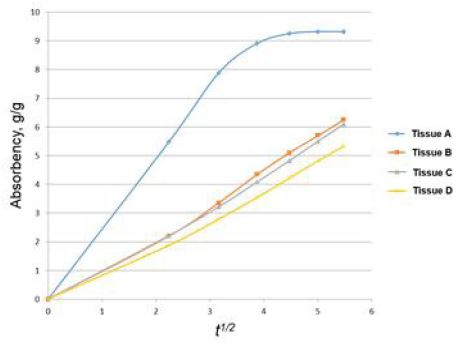
Eq. 14 shows that Q is proportional to t1/2. So, the validity of applying the Lucas-Washburn theory for a hygiene paper can be checked by examining the linearity between the amount of liquid absorbed (Q) and square root of time, t1/2. Plotting Q against t1/2 is referred to as the Lucas-Washburn (L-W) plot. Then, the slope, K, in the plot is called the Lucas-Washburn (LW) slope.
2.4 Absorbency measurements: Orifice(point source) method
There are many methods available for determining absorbency properties of tissue and towel. The principle is based on the capillary forces according to the Laplace equation. At present, however, very few methods have been standardized.19) Burgeni and Kapur has designed the capillary sorption apparatus to apply the Laplace equation.14) This method is intended to determine the absorbency of thick absorbent pads such as baby diapers where the z-directional (i.e., thickness direction) is much more important than the plane (i.e., X- or Y-direction). Water is supplied through the porous plate where all sample surface is contact with the water source. So, the flow in the plane direction would be meaningless. In contrast with the thick absorbent pads, for hygiene paper the X- and Y-directional flow is much more important than the Z-directional flow which may be neglected.
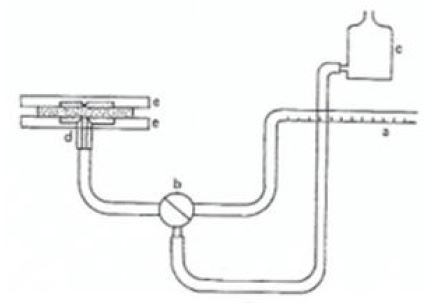
Reid has designed an apparatus where water is supplied through a capillary of a small orifice (or a point-source) to determine absorbency of paper. 19,20) The water is allowed to spread radially. Thus, this can be regarded as a 2-dimensional radial wicking, neglecting the z-direction (i.e., thickness direction). Fig. 4 shows an apparatus developed by S.G. Reid.20)
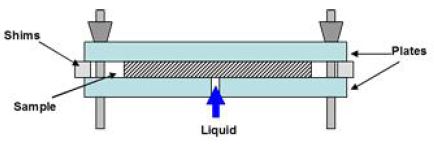
The Reid method has been subsequently automated and the instrument is commercially available, called the “Gravity Absorbency Tester (GAT)”, or “Absorbency Testing System (ATS)”.21,22) The testing procedure is described by TAPPI.23) The method is sometimes called “Automatic Gravimetric Absorbent Tester, or as AGAT”.3,13) The conventional absorbency testers have used the configuration of the flat-flat plates to hold the sample as shown in Fig. 5.13) It has been observed, however, that this configuration should cause the channeling of water between the plate and the sample. Such channeling should bring an erroneous result, producing a higher absorbency than the actual.
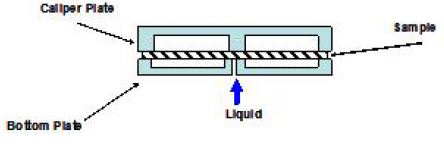
To eliminate the channeling completely, a configuration of the recessed-recessed plates shown in Fig. 6 has been designed.3,13) In this configuration, the sample contacts only with the orifice. Fig. 6 shows the configuration of the recessed-recessed plates. After trying both configurations, Draper Fig. 4. Orifice method by S.G. Reid.20) has concluded that the recessed-recessed configuration should be employed for a hygiene paper. His conclusion is further supported by Hermans, et al.3)
2.5 Validation of the Lucas-Washburn theory applying for hygiene paper
Few data have been available from the literature to validate the Lucas-Washburn theory for absorbency of hygiene paper. Hermans, et al. have determined the absorbency properties of several commercial bathroom tissue products.3) For the products, tissue A and B from K Co. Ltd., tissue C from P Co. Ltd. and tissue D from G Co. Ltd. were used. All these are a 2-ply product made by laminating two of each ply. For the absorbency measurement, the AGAT (Automatic Gravimetric Absorbency Tester) has been used with both configurations as the source of the water supply.
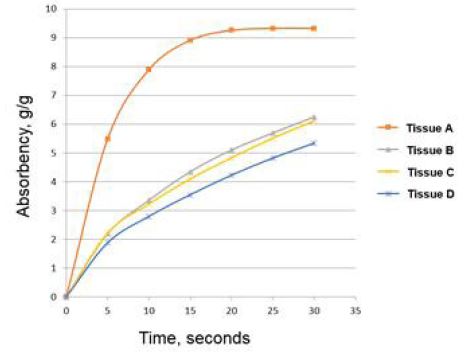
Fig. 7 is a typical example of a plot of absorbency vs. time.3) Then, the absorption rate is calculated from the slope from the curve.21-23) It appears that applying the Lucas-Washburn theory for commercial hygiene paper has not been validated at present. 3) In this paper, to validate the Lucas-Washburn theory for hygiene paper, the absorbency amount vs. √t from Table 1 is plotted. The result is shown in Fig. 8.
Table 1
The regression results
| Sample | L-W Slope | R2 |
|---|---|---|
| Tissue A | 1.950 | 0.95 |
| Tissue B | 1.163 | 0.99 |
| Tissue C | 1.117 | 0.99 |
| Tissue D | 0.99 | 0.98 |
A regression analysis has been performed between absorbency and square root of time, √t for these products. In the case of tissue, A, the regression analysis is performed up to t=25 seconds since it has already reached the maximum. The regression results are shown in Table 1.
The results show that all products have a R2 of greater than 0.95. This validates the Lucas-Washburn theory in determining absorbency rates of the commercial tissue products. According to Table 1, tissue A has the fastest absorbency rate, and tissue D has the lowest. The rate of tissue B and that of tissue C may not be statistically significant.
3. Discussion
It seems surprising that all the products follow the Lucas-Washburn theory though they are produced by the different manufacturers. In this section, it is worthwhile to discuss the factors that influence on the absorbency of hygiene paper products which follow the Lucas-Washburn theory.
3.1 Factors in influencing on absorbency
According to the Lucas-Washburn equation (Eq. 9), the Lucas-Washburn Slope, K, is the function of pore size (Rp), contact angle (θ), viscosity (μ) and surface tension (σ) of a fluid which is water in the present case. It is important to realize that these four variables should not function independently, but depend on each other.
3.1.1 Effect of the contact angle on K
The wettability of cellulose fibers is critically important in determining the absorbency rate of hygiene paper. Hodgson and Berg have measured the contact angles of individual wood pulp fibers using the Wilhelmy plate method.7) From the contact angles (θ), the values of cos(θ) and √θ are calculated. Their results are shown in Table 2.
Table 2
Contact angles of wood pulp fibers7)
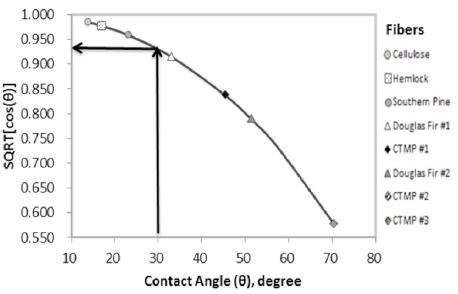
The table shows that wood pulp fibers have a wide range of contact angles from 14.0° to 70°. The same CTMPs show different contact angles depending on the producers. Of note, it is √cos(θ), not θ or cos(θ), which determine the L-W slope, according to the Lucas-Washburn theory. The relationship between θ and √cos(θ) is quite non-linear as shown in Fig. 9 for the wood pulp fibers from Table 2.
Fig. 9 shows that √cos(θ) declines continuously as θ increases. When θ = 20°, √cos(θ) = 0.97, so, the absorbency rate would be reduced by 3%. And when θ = 30°, then √cos(θ) = 0.93. Then, the absorbency rate may be reduced by 7%. Thus, it seems unproductive to attempt to improve the fiber wettability to increase the absorbency rate when θ is lower than approximately 20° - 30°.
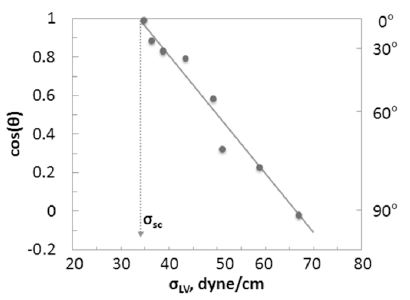
3.1.2 The Zisman’s critical surface tension for wetting
At this point, it may be worthwhile to introduce the concept of the critical surface tension of a solid by Zisman.24) According to the Young-Dupre’ equation, a higher surface tension of a liquid should create a larger contact angle. By plotting cos(θ) vs. σ using a series of liquids on the same solid surface, a linear-relationship between the two may be obtained. Then, by extrapolating the line to cos(θ) = 1 (namely, θ = 0), the corresponding surface tension value of the liquid can be determined. Zisman has defined this surface tension value as the critical surface tension of the solid for wetting. Fig. 10 shows Zisman plot as an illustration.
The critical surface tension (σc) of the solid is the minimal liquid surface tension (σLV) of a liquid when the contact angle of the solid surface is zero. If σLV> σc, the liquid will not spread completely. It is important to realize that when σLV < σc, any attempt to increase the fiber wettability by reducing the contact angle should be in vain.
3.1.3 Effect of a surfactant on wicking rates
A surfactant reduces the surface tension of water. According to the Young-Dupre equation (Eq. 1), adding a surfactant will result in increasing the value of cos(θ) to enhance the wettability of water onto the solid surface.25,26) It is important, however, to realize that it is the product of σLV and cos(θ) that determines the wicking rate according to the Lucas-Washburn equation, Eq. 9. Since adding a surfactant usually results in both reducing σLV and increasing cos(θ), the net effect will depend on the ratio of the product after adding the surfactant to the product before adding the surfactant, i.e.,
In the equation, the subscript, a and b represent, respectively, before adding the surfactant and after adding the surfactant. If R > 1; then the wicking rate will increase. (Positive effect), R = 1; then the wicking rate will remain constant (i.e., no change, No effect), and R < 1; then the wicking rate will decrease (Negative effect). It should be noted that if the surface tension of liquid falls below the critical surface tension for wetting, adding a surfactant only reduces the value of the surface tension of the liquid while the contact angle remains zero. In this case, adding surfactant only results in reducing a wicking rate.
3.1.4 Surface roughness factor
In deriving the Young-Dupre equation, a smooth solid surface has been assumed. However, the contact angle may depend on the surface roughness (or profile). Wenzel has defined the relationship between surface roughness and wettability as:27)
where, R = roughness ratio,
θm = the measured contact angle on the rough surface,
θs = the (young) contact angle on the smooth surface.
R is often referred to as rugacity and is defined as the ratio between the actual and projected surface area. For a smooth surface, R = 1. For Eq. 16 to be valid that the size of water droplet should be larger than the roughness scale by two to three orders of magnitude. In Eq. 16, since R > 1 for a rough surface, θm decreases, or cos(θm) increases when 0° < θs < 90° (i.e., cos(θs) > 0), and vice versa when 90° < θs <180° (i.e., cos(θs) < 0). This suggests that the surface roughness should help improve the wettability when the liquid is partially wet. On the other hand, it should increase the non-wettability when the liquid is hydrophobic. The Wenzel’s theory seems to be supported by Dettre and Johnson who observed that if the surface is hydrophobic (i.e., θ > 90°), it will become more hydrophobic when the surface roughness increases.28) This observation seems rather unexpected from the conventional practice in the printing and coating industries, where a substrate is designed to minimize the surface roughness to improve the wettability and printability.29)
3.1.5 Contact angle hysteresis
So far, we have discussed based on the advancing contact angles. However, the receding contact angles can be quite different from the advancing contact angles. The advancing angle is the angle which forms when a liquid drop is moving forward along the solid surface, and the receding angle is the angle which forms when a liquid drop is moving backward (i.e., retreating) from the solid surface. The difference between the two is called “Contact Angle Hysteresis”.13,14) It has been commonly observed that the receding contact angles are always smaller than the advancing contact angles. For hygiene paper, the advancing contact angle is far more important to be considered since it is seldom reused when it is wet.
3.1.6 Surface heterogeneity
The Young-Dupre equation is derived by assuming the solid surface which is homogeneous. However, if the solid surface is made of two different chemical compositions, the contact angle will depend on the location. For example, when a product is made of mixed pulp having both lower and higher contact angles, the wicking direction will be from the position of the larger angle to that of the lower as shown in Case 2, in Fig. 3-b. In practice, layering of each pulp is more commonly used than mixing the pulp homogenously in a slurry for making a hygiene paper. Surface heterogeneity may be also caused by non-uniform distribution of additives such as pigment, fillers, inks, etc. onto the surface.
Cassie and Baxter have investigated on the effect of a chemically heterogeneous surface on the contact angle.30) For surfaces that contain two regions that are different in their wettability, their equation for the apparent contact angle is:
where, f1 and f2 are area fractions of the different regions.
3.1.7 Absorbency capacity, bulk, and in-use holding capacity
So far, we have discussed the factors that influence on the absorbency rate based on the Lucas- Washburn equation. Absorbency capacity may be classified into two groups: 1) Saturation Absorbency Capacity, and 2) Effective In-use Absorbency Capacity. In general, these two groups go hand in hand, i.e., the latter may increase as the former increases. However, it may not always be the case. A theoretical, maximum holding capacity, g-water/ g-product may be estimated from the bulk of the product. The bulk is defined as the pore volume per mass, cm3/g, which is the inverse of the density.3)
Hermans et al have determined the water holding capacity (WHC) of various tissue samples using the AGAT (automatic gravity absorbency tester) using the flat-flat configuration of the point source shown in Fig. 5. Here, the WHC is determined when the sample does not absorb anymore, i.e., to reach the equilibrium. Table 3 shows the results of some commercial tissue products.3)
Table 3
Properties of some commercial tissue products3)
| Sample | Bulk (cm3/g) | WHC (g/g) | WHC/Bulk (%) |
|---|---|---|---|
| Tissue A | 11.04 | 7.30 | 66.2 |
| Tissue C-1 | 10.56 | 7.44 | 70.5 |
| Tissue B-1 | 8.47 | 6.05 | 71.5 |
There seems a poor correlation between the bulk and the water holding capacity. It is particularly noticeable that the WHC is in a range of only 60- 70% of the bulk. This suggests that the available pore volume in the product may not have been fully utilized to absorb the water. Another important point is to notice the difference between the water-holding capacity, g-water/g-sheet and the in-use holding capacity per sheet, g-water/sheet. The latter is determined from:
The hygiene paper producers have attempted to increase the bulk by increasing the apparent thickness. Creping is one of the key process is widely used to increase softness and absorbency by increasing bulk.31-33) Through-Air Drying (TAD) is also employed to this end. In addition, laminating, embossing, and patterning have been employed.3,33)
4. Conclusion
The classical Lucas-Washburn theory can be used to characterize the absorbency of a hygiene paper which is made of cellulose fibers having a low density of about 0.1 g/cm3 and a low basis weight of about 30-40 g/m2.
Several factors participate in determining the absorbency performance of the product. They are: fiber wettability, pore size distribution, fluid properties (i.e., viscosity, and surface tension value). These factors may, however, interact with each other. So, maximizing each factor one by one may be detrimental to overall efficiency. For example, adding a surfactant to reduce the contact angle may result in the decrease in the wicking rate according to the Lucas-Washburn theory.
Bulk may be used to estimate the potential absorbency capacity of a product. In practice, however, for a hygiene product, this potential absorbency capacity has been never utilized. It is disposed of well before it reaches the maximum capacity. Many absorbency test methods exist, but few are standardized. It is highly desirable to standardize the test methods since the results should depend on the test methods employed. For developing standard absorbency test methods, it should be based on subjective tests by users for in-use situation.