1. Introduction
2. Materials and Methods
2.1 Raw materials
2.2 Organosolv pulping
2.3 Measurement
3. Results and Discussion
3.1 Effect of reaction temperature and time on pulping and fiber characteristics
3.2 Effect of glycol ether and catalyst concentration on pulping and fiber characteristics
4. Conclusion
1. Introduction
Lignocellulosic fibers, derived from woody and non-woody materials, are sustainable and renewable bioresources [1,2,3,4]. Bamboo is one of the non-woody biomass and valuable raw materials for pulp production, due to its advantages such as fast growth and high yield, as well as its suitability for textile fiber due to its unique fiber shape, unlike woody fibers [5,6,7,8]. Moreover, bamboo pulp has an excellent potential for applications such as the paper industry, dissolving pulp, and new cellulose or ligno-cellulosic materials, including nanofibrillated cellulose, nanoparticles, and composite reinforcement [9,10,11,12]. There are two primary types of pulp used in the industry: papermaking pulp and dissolving pulp. Within the woody materials, the papermaking pulp is no longer considered necessary. However, sustainable raw materials, such as non-woody biomass, could be an alternative way to utilize in the papermaking area in the near future [13,14]. The other one, dissolving pulp, does not require further treatment to dissolve in solvents such as cellulose derivatives, including cellulose acetate. Although most dissolving pulp is derived from cotton, the woody and non-woody biomass-based dissolving pulp has also captured people’s attention as a high-value product [15,16].
To pulping biomass resources, three main elements determining final pulp quality: pulping methods, type of plant, and reaction severity. The pulping method, which includes chemical, mechanical, and semi-chemical pulping, is one of the primary methods for attacking hemicellulose and lignin to obtain lignocellulosic fibers [17,18]. By the type of plant, the final quality of pulp has different properties after pulping. The condition of the pulping process is controlled by the type of plant, such as hardwood and softwood. The fiber length of hardwood kraft pulp (HwBKP) is shorter than that of softwood kraft pulp (SwBKP) because the cell type of the plant is different [19,20,21,22]. In bamboo pulping, the chemical pulping process is similar to the wood pulping process, which includes sulfate, sulfite, organosolv, and soda pulping. The fiber length exceeds 1.5 mm, which is longer than that of HwBKP [23]. Therefore, it is essential to optimize the pulping conditions for bamboo pulping. The optimized reaction severity is still necessary even though the pulping method and the type of plant are set for pulping. Organosolv pulping is typically based on ethanol and a catalyst, often supplemented with acetic acid, hydrochloric acid, sulfuric acid, formic acid, magnesium chloride, and sodium hydroxide [24,25]. These conditions are applied to substrates, including woody and non-woody materials, to control the solvent content, catalyst content, temperature, and time.
Recently, glycol ether-based organosolv pulping has gained attention due to the high boiling point and strong solvating ability of glycol ethers, which enable effective delignification and fiber separation at comparatively low temperatures [26]. Moreover, pulps produced using glycol ether–water systems have shown favorable dissolution behavior in alkaline aqueous solutions, suggesting their potential for both papermaking and dissolving pulp applications [27,28].
Despite these advantages, studies on bamboo pulping using glycol ether-based organosolv systems have been limited, particularly with respect to separating the effects of reaction condition and pulping liquor composition. Reaction temperature and time directly control thermal severity, influencing fiberization, lignin removal, and cellulose degradation. In contrast, the concentration of glycol ether and acid catalyst governs the chemical environment of the pulping liquor, affecting solvent penetration, delignification efficiency, and cellulose accessibility. Clarifying the individual and combined effects of these parameters is crucial for tailoring the properties of bamboo pulp to meet specific application requirements.
Therefore, the objective of this study is to evaluate the effects of key pulping parameters on bamboo organosolv pulping using a glycol ether–water system with sulfuric acid as a catalyst. Specifically, the influence of reaction temperature and reaction time on pulp yield, fiber morphology, papermaking-related properties, and alkaline dissolution behavior was first investigated. Subsequently, the effects of glycol ether concentration and acid catalyst dosage on pulp characteristics were examined under controlled reaction conditions. This study aims to provide foundational resources for the production of bamboo pulps suitable for papermaking and dissolution-oriented applications using an organosolv pulping process.
2. Materials and Methods
2.1 Raw materials
Bamboo (Phyllostachys bambusoide and Phyllostachys nigra var. henonis) was provided by Forest Biomaterials Research Center, National Institute of Forest Science (Jinju, Republic of Korea), and used for the raw materials in this study. The bamboo was cut into lengths of 50–60 mm and chopped to a width of about 10–15 mm. The air-dried bamboo chips were used in the pulping process, and their chemical properties were analyzed based on Technical Association of the Pulp and Paper Industry (TAPPI) standard methods, as shown in Table 1. Moisture content was calculated each time before preparing the pulping liquor. All the reagents and solvents, glycol ether, and sulfuric acid for the organosolv pulping process were purchased in extra-pure grade from Daejung Chemicals Co., Ltd., Republic of Korea.
Table 1.
Chemical composition of bamboo chips
2.2 Organosolv pulping
An organosolv-based pulping process was applied to bamboo using glycol ether as a high-boiling-point solvent, with sulfuric acid employed as the acid catalyst. Two sets of pulping experiments were conducted to separately evaluate the effects of (i) reaction temperature and time and (ii) pulping liquor composition, respectively. For the evaluation of reaction temperature and time, the glycol ether concentration and catalyst concentration were fixed at 95% (v/v) and 1.0 M, respectively, to isolate the influence of reaction temperature and time on bamboo pulping behavior. Air-dried bamboo chips (300 g, oven-dry basis) were immersed in the pulping liquor at a liquor-to-solid ratio of 3:1 (w/w) in a 2 L sealed glass reactor. To enhance solvent penetration into the bamboo chips, the mixture was subjected to vacuum impregnation for 1 h prior to pulping. The soaked samples were then reacted in an autoclave at two temperatures (100°C and 120°C) and two reaction times (60 min and 180 min), resulting in four pulp samples, designated according to their reaction temperature and time (Table 2). In a separate experiment, the effects of glycol ether and catalyst concentration were investigated at a fixed reaction temperature of 110°C and a fixed reaction time of 120 min. The pulping conditions were varied by adjusting the glycol ether content and sulfuric acid concentration, while all other operating parameters were maintained constant. Each pulp sample was designated according to the corresponding glycol ether content and catalyst concentration, as listed in Table 3.
Table 2.
Pulping condition based on reaction temperature and time
| Sample type | Temperature (°C) | Time (min) | Note |
| T100-60m | 100 | 60 |
GE 95% Catalyst 1.0 M |
| T100-180m | 180 | ||
| T120-60m | 120 | 60 | |
| T120-180m | 180 |
Table 3.
Pulping conditions based on glycol ether and catalyst concentration
| Sample type | Glycol ether (%) | Catalyst (M) |
| G0-0.6M | 0 | 0.6 |
| G60-0M | 62.5 | 0.0 |
| G60-0.6M | 0.6 | |
| G60-1.4M | 1.4 | |
| G100-0.6M | 100 | 0.6 |
After completion of each pulping reaction, the solid fraction was separated and thoroughly washed with distilled water. The pulped material was subsequently disintegrated and screened using a Somerville screen equipped with a 0.2 mm slit (Daeil Machinery Co., Ltd., Republic of Korea) to remove uncooked residues. The screened pulps were repeatedly washed with distilled water until a neutral pH was reached to eliminate residual chemicals and detached lignin particles. The resulting pulps were stored in a wet state at 4°C for subsequent characterization.
2.3 Measurement
After each pulping condition, the screened pulp yield (TAPPI/ANSI T 275), fiber length, fiber width, and fines content were measured using an L&W Fiber Tester Plus (ABB, Sweden). The Klason lignin content was determined according to TAPPI T 222. To evaluate the papermaking properties of bamboo handsheets with a basis weight of 65 ± 3 g/m², the zero-span tensile strength was measured using a zero-span tensile tester (Zero-span™ TS 100, Pulmac Inc., USA) according to TAPPI T 231 to assess the intrinsic fiber strength. Additionally, inter-fiber bonding was evaluated using the tensile strength index. For the characterization of dissolving pulp properties, dissolution in a NaOH solution was performed to determine the complex dissolving properties of cellulose, hemicellulose, and lignin. A 7% NaOH aqueous solution was prepared to dissolve the components of the organosolv pulp. 0.6 g of dried bamboo pulp was soaked in 10 mL of a 7% NaOH solution. The mixture was frozen at -15°C for 12 h and thawed at room temperature. The dissolved fraction was separated by centrifugation (1580, Labogene, Korea) at 12,300 G force for 20 min and washed with 40 mL of 1% NaOH aqueous solution repeatedly until the washed solution was clear. Finally, the undissolved fraction was washed with water and weighed to calculate the dissolution yield (Eq. (1)).
where, : Insoluble weight in dissolution solvent (g)
: Dried pulp weight in dissolution solvent (g)
3. Results and Discussion
3.1 Effect of reaction temperature and time on pulping and fiber characteristics
Table 4 summarizes the effects of reaction temperature and time on the pulping yield (total and screened yield) of bamboo pulp prepared by the glycol ether-based organosolv process. At 100°C, increasing the reaction time from 60 to 180 min decreased the total yield from 60.85 to 42.58%, whereas the screened yield increased from 25.70 to 33.30%. These results suggest that prolonged reaction times at a constant temperature promote more effective fiber separation into screened fibers without excessive cellulose degradation [29]. In contrast, increasing the reaction temperature to 120°C resulted in a further decrease in total yield, particularly at longer reaction times. At 120°C and 180 min, the total yield decreased to 19.41%, with a corresponding screened yield of only 9.79%. The significant loss in total and screened yields indicates excessive pulping conditions, likely resulting in extensive dissolution of cell wall components and degradation of carbohydrate.
Table 4.
Effect of reaction temperature and time on pulping yield
| Sample type | Total yields (%) | Screened yields (%) | Shive (%) |
| T100-60m | 60.85 | 25.70 | 35.15 |
| T100-180m | 42.58 | 33.30 | 9.28 |
| T120-60m | 35.01 | 24.18 | 10.83 |
| T120-180m | 19.41 | 9.79 | 9.62 |
These results indicate that reaction temperature and time significantly influence pulping yield, and that higher temperature (120°C) and longer pulping time (180 min) promote the solubilization of cellulosic structural components, thereby reducing fiber yield [30,31].
Table 5 presents the changes in fiber length, fiber width, and fines content of screened pulp as a function of reaction temperature and time. Extending the reaction time from 60 to 180 min at 100°C resulted in no significant changes in fiber length, fiber width, or fines content. At 120°C and 60 min, the fiber length (1.437 mm) and width (21.4 µm) were comparable to those obtained at 100°C, indicating that a suitable increase in temperature alone does not immediately lead to fiber degradation when the reaction time is limited.
Table 5.
Fiber characteristics of bamboo pulp as a function of reaction temperature and time
| Sample type | Fiber length (mm) | Fiber width (µm) |
Fines (%) (< 0.2 mm) |
| T100-60m | 1.439 | 21.9 | 4.3 |
| T100-180m | 1.484 | 21.5 | 4.4 |
| T120-60m | 1.437 | 21.4 | 5.0 |
| T120-180m | 1.099 | 24.6 | 18.9 |
In contrast, pulping conditions at 120°C for 180 min resulted in pronounced changes in fiber morphology. The average fiber length decreased substantially to 1.099 mm, accompanied by a sharp increase in fines content to 18.9%, indicating fiber deformation such as cutting under harsh conditions of high temperature and long reaction time. This behavior is consistent with the substantial pulping yield loss observed at T120-180m. Unexpectedly, the average fiber width measured at T120-180m was the largest, at 24.6 µm. Fibers are more susceptible to harsh pulping conditions, tending to be converted to fine fibers below the measurement threshold (<0.2 mm). Consequently, the remaining fiber population is skewed toward relatively coarse fibers, which is believed to be the cause of the apparent increase in the measured average fiber width [32].
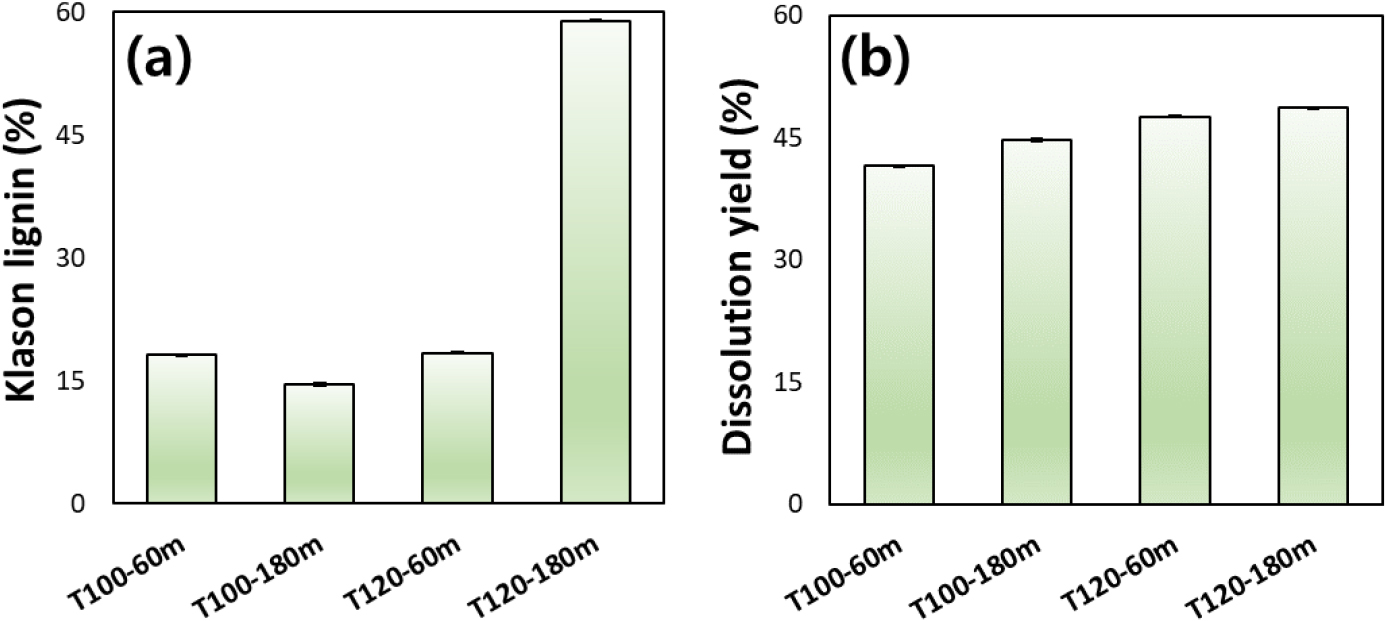
Fig. 1a shows the Klason lignin content of bamboo pulps prepared under different reaction temperature and time conditions. At 100°C, extending the reaction time from 60 to 180 min resulted in a slight decrease in Klason lignin content, indicating that delignification progresses with prolonged reaction times under mild thermal conditions. A similar lignin level was observed at 120°C for 60 min, suggesting that a slight increase in temperature alone does not substantially enhance lignin removal when the reaction time is limited. In contrast, pulping at 120°C for 180 min led to a pronounced increase in the measured Klason lignin content. This apparent increase is likely associated with the extensive degradation and dissolution of carbohydrate components under excessive thermal severity, which increases the relative proportion of acid-insoluble lignin in the residual solid fraction. Such behavior has been reported in several studies, where loss of polysaccharides leads to an artificial increase in the Klason lignin fraction due to the formation of lignin-like (pseudo-lignin) materials [33,34].
The dissolution yield increased gradually with increasing reaction intensity, from T100-60m to T120-180m (Fig. 1b). The absence of a clear increase in dissolution yield under the T120-180m condition, despite severe fiber damage, can be attributed to the use of screened pulp for the dissolution test. The screened yield was below 10% at T120-180m, suggesting that highly degraded or weak fiber fractions were likely solubilized or removed during the pulping and washing steps.
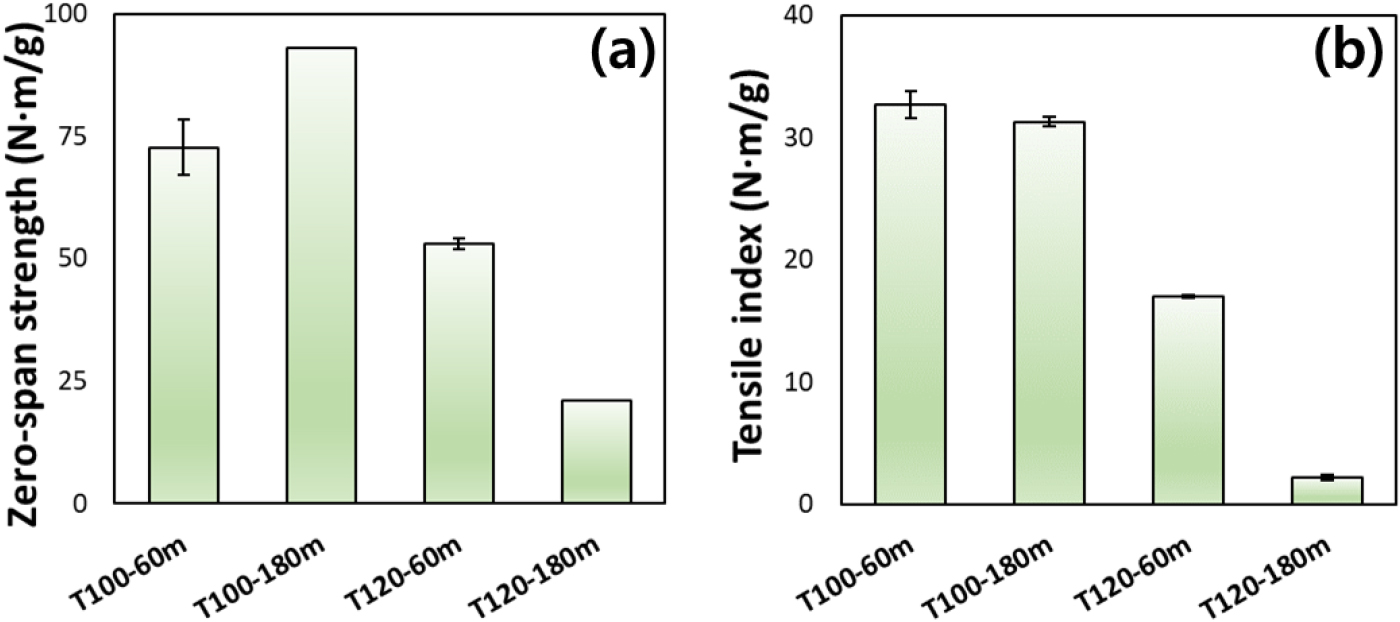
Fig. 2 shows the strength properties of bamboo pulps prepared under different reaction temperatures and times. Both strength parameters exhibited strong dependence on pulping condition, reflecting changes in intrinsic fiber strength and inter-fiber bonding ability [35,36]. The zero-span tensile strengths of T100-60m and T100-180m were 72.7 N·m/g and 93.1 N·m/g, respectively, indicating that the strength increased by approximately 28% as the reaction time increased. This indicates that the inherent strength of the fibers was enhanced. This enhancement can be attributed to more effective removal of non-cellulosic components and improved fiber individualization under mild pulping conditions, without causing significant fiber damage [37]. When the reaction temperature was increased to 120°C, a marked deterioration in mechanical properties was observed, particularly at a reaction time of 180 min. At 120°C and 60 min, both zero-span strength and tensile index decreased compared to those obtained at 100°C, indicating the onset of fiber weakening under higher thermal severity. The marked loss of tensile strength under the T120-180m condition reflects extensive fiber fragmentation and depolymerization, as previously evidenced by Table 5. Mild pulping conditions enhance intrinsic fiber strength while maintaining sufficient bonding ability, whereas excessive harsh condition leads to irreversible fiber damage and dramatic deterioration of mechanical properties [38]. These findings are consistent with the pulping yield, fiber morphology, and dissolution behavior discussed earlier and highlight the importance of controlling reaction conditions to balance pulping efficiency and fiber performance in glycol ether-based organosolv pulping of bamboo.
3.2 Effect of glycol ether and catalyst concentration on pulping and fiber characteristics
Table 6 presents the effects of glycol ether content and catalyst concentration on the pulping yield of bamboo pulp obtained under fixed reaction temperature (110°C) and time (120 min). Under glycol ether-free conditions (G0-0.6M), a high total yield (61.70%) was obtained; however, the screened yield remained low (20.33%), accompanied by a high shive content (41.37%). This result suggests that in the absence of glycol ether, lignin removal is insufficient, and fiber separation is limited, resulting in uncooked fiber bundles. When glycol ether was introduced at 60% with a low catalyst concentration (G60-0M), the total yield increased markedly to 93.14%, while the screened yield decreased to 8.15% and shive content increased sharply to 84.98%. Glycol ether alone, without sufficient acid catalysis, is ineffective in promoting fiber separation and instead results in the retention of largely unpulped material. In contrast, at 60% glycol ether with 0.6 M catalyst (G60-0.6M), the screened yield increased substantially to 40.44%, while shive content decreased to 2.72%. A further increase in catalyst concentration to 1.4 M at the same glycol ether content (G60-1.4M) resulted in a similar total yield (41.47%) and a slightly reduced screened yield (35.83%), accompanied by a modest increase in shive content (5.64%). According to Yang et al.’s study [39], excessively high acid concentrations do not further enhance fiber separation efficiency; instead, they promote fiber degradation or dissolution. At 100% glycol ether with 0.6 M catalyst (G100-0.6M), the screened yield decreased to 27.21% and shive content increased to 15.24%, despite a total yield comparable to that of G60-0.6M. These results demonstrate that both glycol ether content and catalyst concentration significantly influence pulping yield during bamboo organosolv pulping, and that effective fiber liberation is achieved only under balanced solvent and catalyst conditions.
Table 6.
Effect of glycol ether and catalyst concentration on the pulping yield
| Sample type | Total yields (%) | Screened yields (%) | Shive (%) |
| G0-0.6M | 61.70 | 20.33 | 41.37 |
| G60-0M | 93.14 | 8.15 | 84.98 |
| G60-0.6M | 43.17 | 40.44 | 2.72 |
| G60-1.4M | 41.47 | 35.83 | 5.64 |
| G100-0.6M | 42.45 | 27.21 | 15.24 |
In the subsequent analysis, fiber properties were evaluated excluding the G60-0M condition, where inadequate fibrillation limited reliable assessment. Table 7 presents the changes in fiber length, fiber width, and fines content of bamboo pulp as a function of glycol ether content and catalyst concentration under fixed reaction temperature (110°C) and time (120 min). Under aqueous pulping conditions with acid catalyst and without glycol ether (G0-0.6M), the fiber length was 1.399 mm, and the fines content remained low (2.4%). In contrast, the combination of 60% glycol ether with 0.6 M catalyst (G60-0.6M) resulted in an increase in fiber length to 1.482 mm and a reduction in fiber width to 19.6 µm. The fines content was 5.0%, indicating that, under these conditions, fiber liberation was enhanced without excessive fragmentation. Increasing the catalyst concentration to 1.4 M at the same glycol ether content (G60-1.4M) led to a similar fiber length (1.511 mm), fiber width (21.2 µm), and fines content (4.6%). Increasing acid concentration beyond a certain level does not substantially improve fiber morphology. In contrast, when the glycol ether content was increased to 100% with 0.6 M catalyst (G100-0.6M), the fiber length decreased sharply to 0.749 mm, while the fines content increased markedly to 15.2%, indicating severe fiber fragmentation. The reduced fiber width (15.6 µm) further suggests extensive breakdown of fibrous structures under excessive solvent intensity, resulting in a dominance of fibrillation, short fibers, and fines [40].
Table 7.
Effect of glycol ether and catalyst concentration on fiber characteristics
| Sample type | Fiber length (mm) | Fiber width (µm) |
Fines (%) (< 0.2 mm) |
| G0-0.6M | 1.399 | 22.2 | 2.4 |
| G60-0.6M | 1.482 | 19.6 | 5.0 |
| G60-1.4M | 1.511 | 21.2 | 4.6 |
| G100-0.6M | 0.749 | 15.6 | 15.2 |
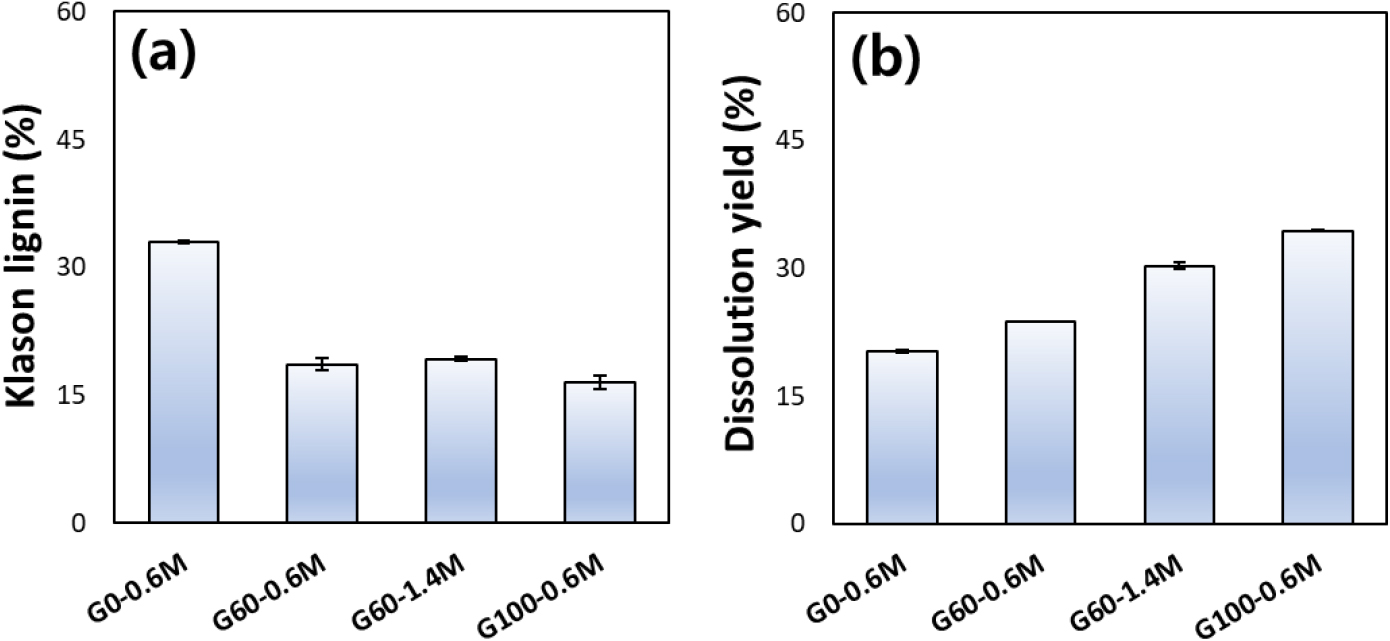
The effects of glycol ether content and catalyst concentration on residual lignin content and dissolution behavior of bamboo pulp are shown in Fig. 3, evaluated under fixed reaction temperature (110°C) and time (120 min). Under aqueous pulping conditions with acid catalyst and without glycol ether (G0-0.6M), a relatively high Klason lignin content was observed, indicating limited delignification under water-based pulping conditions (Fig. 3a). G60-0.6M led to a pronounced reduction in Klason lignin content, demonstrating the effectiveness of glycol ether-assisted organosolv pulping in promoting lignin solubilization. An increase in catalyst concentration to 1.4 M at the same glycol ether content (G60-1.4M) resulted in only a marginal additional decrease in Klason lignin content. Excessive acid dosage does not proportionally improve delignification efficiency. At 100% glycol ether with 0.6 M catalyst (G100-0.6M), a Klason lignin content similar to that of glycol ether 60% (G60) was observed. The dissolution yield of pulp is shown in Fig. 3b. Under glycol ether-free conditions with acid catalyst (G0-0.6M), the dissolution yield remained low. This observation is consistent with the findings of Shi et al. [41], who reported that insufficient lignin removal within fibers restricts NaOH penetration and limits dissolution of the cellulose matrix. The incorporation of 60% glycol ether with 0.6 M catalyst (G60-0.6M) resulted in a slight increase in dissolution yield, which can be attributed to improved cellulose accessibility following effective delignification. At higher catalyst concentration (G60-1.4M), the dissolution yield increased by approximately 28% despite only minor changes in Klason lignin content. Increased acid concentration may promote dissolution through structural modification of cellulose rather than through further lignin removal. The highest dissolution yield was observed at 100% glycol ether with 0.6 M catalyst (G100-0.6M). Taken together with the results discussed in Fig. 1b, these findings indicate that pulping conditions involving glycol ether contents of approximately 95% or higher and catalyst concentrations of 0.6 M or above favor the production of bamboo pulp with enhanced dissolution characteristics.
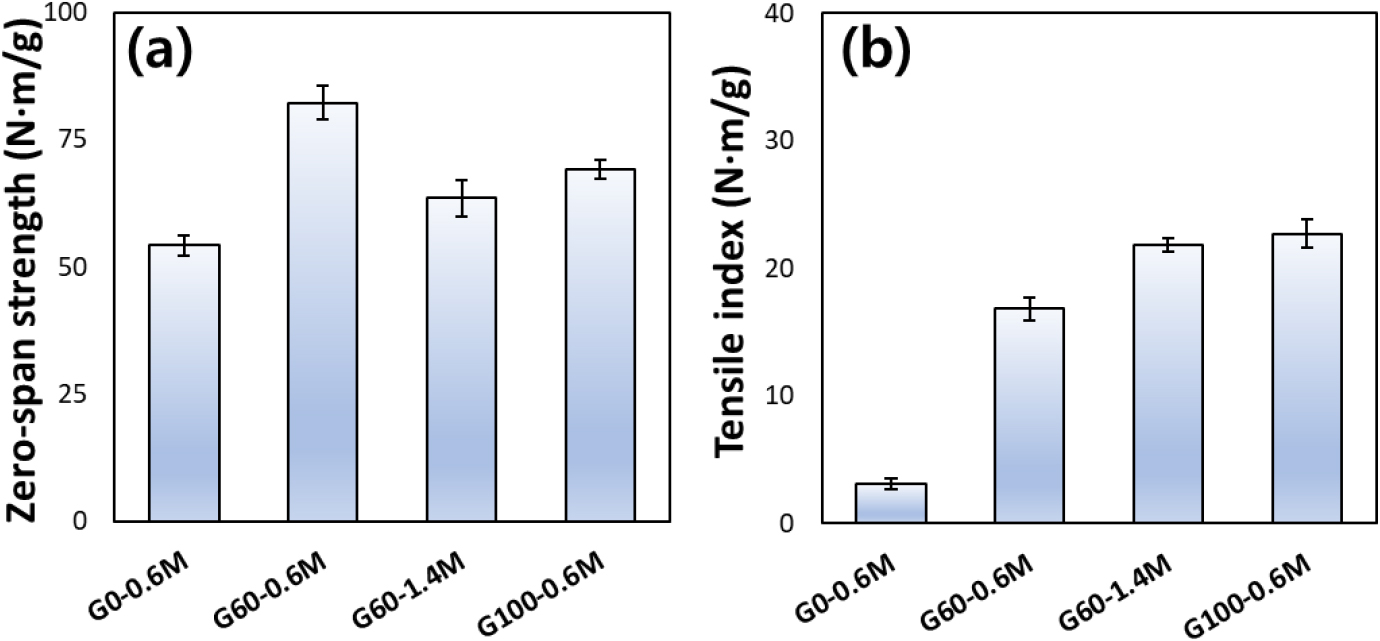
The results of measuring the strength of pulp and handsheet are shown in Fig. 4. In glycol ether-free conditions (G0-0.6M), both zero-span strength and tensile index were relatively low, indicating insufficient development of fiber strength and bonding. The introduction of 60% glycol ether with 0.6 M catalyst (G60-0.6M) resulted in a pronounced increase in zero-span strength. Additionally, the tensile index increased markedly compared to the glycol ether-free condition, indicating enhanced fiber-fiber bonding associated with improved fiber individualization. When the catalyst concentration was increased to 1.4 M at the same glycol ether content (G60-1.4M), the tensile index increased further, while the zero-span strength showed a slight decrease. Higher acid severity can induce fiber deformation, such as curling, and the resulting increase in fiber flexibility expands the fiber-fiber bonding area, thereby enhancing the tensile strength of paper [38]. However, chemical modification of the carbohydrate structure may partially reduce the intrinsic strength of individual fibers [42]. At 100% glycol ether with 0.6 M catalyst (G100-0.6M), the zero-span strength was lower than that observed for the G60-0.6M condition, indicating partial deterioration of intrinsic fiber strength. In contrast, the tensile index of the resulting sheet reached its highest value among the tested conditions. This apparent discrepancy can be explained by the fiber morphology results, which showed a pronounced reduction in fiber length under the G100-0.6M condition. The predominance of shorter fibers increased the number of fiber-to-fiber contact points and enhanced inter-fiber bonding, particularly through hydrogen bonding, thereby improving sheet tensile strength despite reduced single-fiber strength.
Based on these results, it is confirmed that the properties of bamboo pulp can be effectively adjusted by controlling both reaction conditions (time and temperature) and the composition of the pulping liquor. Mild reaction temperatures and times, combined with a balanced glycol ether content and a suitable catalyst concentration, favor the preservation of fiber length and intrinsic fiber strength, making these conditions suitable for paper-grade pulp production with enhanced mechanical properties. Higher solvent and chemical intensity promote lignin removal and dissolution behavior, while inducing fiber shortening and partial degradation of the fibers. Such conditions are therefore more appropriate for producing dissolution-grade pulp. The glycol ether-based organosolv pulping method presented in this study provides a versatile platform for selectively producing paper-grade or dissolution-grade bamboo pulp by controlling pulping conditions to suit the targeted end use. In addition, the use of glycol ethers, characterized by low volatility and high boiling points, offers advantages in reduced solvent loss and improved solvent recyclability compared to conventional organosolv solvents. These features suggest potential benefits for energy efficiency and environmental performance, particularly under mild pulping conditions.
4. Conclusion
This study evaluated the effects of reaction conditions (temperature and time) and pulping liquor composition on the properties of bamboo pulp produced by a glycol ether-based organosolv pulping process. The results showed that increasing thermal severity promoted fiber separation and lignin removal; however, excessively harsh conditions resulted in substantial screened yield loss and increased fines formation. In contrast, mild temperature conditions (100°C) preserved fiber length and intrinsic fiber strength, leading to improved papermaking performance. The dissolution yield increased with increasing reaction temperature, reaction time, and chemical treatment severity, particularly at high glycol ether content (≥95%) and sufficient catalyst concentration (≥0.6 M). Accordingly, harsh reaction conditions and chemical intensity are favorable for producing dissolution-grade bamboo pulp, whereas fiber-preserving conditions are more suitable for papermaking applications. These results suggest a method for producing bamboo pulp suitable for various end-uses via a glycol ether-based organosolv pulping process.