1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Methods
3. Results and Discussion
3.1 DS confirmation by 1H-NMR spectroscopy in carboxymethylation pretreatment
3.2 Impact of charge density on CNF morphology
3.3 Impact of passes of high-pressure homogenization on CNF morphology with different DS of carboxymethylation
4. Conclusions
1. Introduction
Cellulose nanofibers (CNFs) have emerged as a promising class of sustainable nanomaterials [1,2] due to their high mechanical properties [3,4,5] biodegradability [6], and renewable nature. Cellulose is the most abundant organic material derived from various cellulose sources, including wood, agricultural residues, and grasses. CNFs are typically produced through combinations of chemical pretreatment [7] and mechanical processes to break down the hierarchical structure of cellulose into nanoscale fibrils. The common approaches involve carboxymethylation pretreatment [8,9,10] or 2,2,6,6-Tetramethylpiperidine-1-oxyl (TEMPO)-oxidation [11,12,13] followed by mechanical fibrillation. Carboxymethylation introduces carboxymethyl groups onto the cellulose chains, enhancing their hydrophilicity and reducing intermolecular hydrogen bonding, which facilitates subsequent disintegration. TEMPO-oxidation oxidizes the primary alcohol in the C6-position of cellulose to carboxylate.
The carboxymethylation reaction has gained significant attention as an effective chemical strategy to make carboxymethylcellulose (CMC) [14]. CMC is used in food [15,16], pharmaceuticals [17,18], cosmetics, and personal care areas [19,20,21]. A lower degree of substitution (DS) through carboxymethylation could bring different properties to cellulose derivatives. Introduction of the carboxymethyl group to cellulose enhances the production of CNFs from natural cellulose sources.
This process involves the introduction of carboxymethyl groups (–CH2COOH) onto the hydroxyl groups of glucose in cellulose chains through etherification, typically using sodium monochloroacetate in an alkaline medium. By modifying the surface chemistry carboxymethylated anion increases the negative charge density and hydrophilicity of the fibers, weakening the strong intermolecular hydrogen bonds that hold the cellulose structure together. This chemical modification reduces the energy required for subsequent mechanical disintegration, facilitating the isolation of nanoscale fibrils with improved uniformity and dispersibility [22,23]. Carboxymethylation pretreatment not only enhances the efficiency of CNF production but also imparts functional properties, such as enhanced water retention [24,25,26] and rheological properties [27,28,29] by carboxymethylated CNFs.
Mechanical fibrillation of cellulose, often achieved through high-pressure homogenization [30,31,32], ultrasonication [33,34], or grinding [35,36,37]. This synergistic process by chemical pretreatment and mechanical fibrillation not only improves the efficiency of nanofiber production but also tailors the surface chemistry and morphology of CNFs. These good-quality CNFs are suitable for diverse applications, including composites, biomedical materials, and eco-friendly packaging.
In this study, we explored the effects of carboxymethylation pretreatment and mechanical treatment on the morphology of CNFs. Proton nuclear magnetic resonance (1H-NMR) spectroscopy was used to determine the degree of pretreatment, and transmission electron microscopy (TEM) was used to determine the size of the CNFs.
2. Materials and Methods
2.1 Materials
The pulp used for carboxymethylation was hardwood bleached kraft pulp (LBKP) provided by Moorim P&P. Sodium hydroxide (96.0%, OCI), acetic acid chloride (98.0% Samjeon Chemical), and ethanol (95%, Samjeon Chemical) were used as reagents used for the carboxymethylation substitution and substitution position, and sulfuric acid (96.0%, Samjeon Chemical) and heavy water (99.9%, Sigma Aldrich) were purchased and used for the analysis of carboxymethylation substitution and substitution location.
2.2 Methods
2.2.1 Cellulose nanofiber
2.2.1.1 Carboxymethylation pretreatment of pulp
The amount of chemical needed and reaction conditions for carboxymethylation are shown in Table 1. The reaction sequence was to dissociate 10.0 g of pulp in 200 mL of ethanol, dissolved in sodium hydroxide, and leave it at room temperature for 2 hours to produce negative ions in the pulp. After mixing 150 mL of ethanol dissolved in chloride acetic acid in the pulp left at room temperature, it was reacted at 80°C for 2 hours, and the pulp was subjected to carboxymethylation pretreatment. After the reaction was completed, the pulp was neutralized and washed to pH 6–7 using acetic acid and distilled water after removing ethanol, and was used for substitution analysis and preparation of CNFs.
Table 1.
Chemicals and pulp are used for carboxymethylation
| DS | Pulp (g) | NaOH (g) | Chloroactic acid (g) |
| 0.1 | 200 | 7.4 | 11.7 |
| 0.2 | 14.8 | 23.3 | |
| 0.3 | 22.2 | 35.0 | |
| 0.4 | 29.6 | 46.7 | |
| 0.5 | 37.0 | 58.3 | |
| 0.6 | 44.4 | 70.0 |
2.2.1.2 Cellulose nanofiber making
To prepare carboxymethylated pretreated pulp into CNFs, a grinder (Supermasscolloider; MKZA10-15IV, Masuko Sangyo, Japan) and a High Pressure Homogenizer (Panda plus, GEA, Italy) were used. The carboxymethylated pretreated hardwood kraft pulp was diluted to 1.0% (w/w) and then mechanically disintegrated. The disintegrated pretreated pulp was ground with a supermasscollider grinder (MKG-C 120, Masuko Sangyo, Japan) inside, with a rotation speed of 1,000 ± 60 rpm, a disc gap of 200–250 µm, and a pressure of 800 ± 50 bar. Pre-ground pulp was fibrillated with the high-pressure homogenizer at different repeating times. To compare the characteristics of CNFs according to the operating conditions of the manufacturing equipment, it was prepared by passing the grinder a total of 4 times and then passing the high-pressure homogenizer 0–8 times, and samples were collected every 0, 2, 4, 6, and 8 passes and used for subsequent analysis.
2.2.2 Analysis
2.2.2.1 DS in carboxymethylation of hardwood pulp by 1H-NMR spectroscopy
A 1H-NMR spectroscopy analyzer was used to analyze the DS and substitution position of bleached kraft pulp of carboxymethylated hardwoods. After adding 40 mg of the sample, it was hydrolyzed with 0.6 mL of 72.0% sulfuric acid in a pressurized tube in the shaker at 30°C for 1 hour. After the first hydrolysis, the sample in acidic solution was diluted by adding 3.0 mL of deuterium oxide (D2O), and kept in an oven at 100°C for 1 hour. After the reaction, the filtrate was collected and filtered with quantitative filter paper. Sample for 1H-NMR spectroscopy analyzer analysis, 7 drops of concentrated sulfuric acid were added to filtrate. A Bruker AVANCE NMR spectrometer (500 MHz) was used for this analysis and the data were analyzed by integrating the anomeric hydrogen peak and the carboxyl group peak [38]. The integral values were calculated using the following equation to calculate the DS of bleached kraft pulp of carboxymethylated hardwoods [39,40].
2.2.2.2 Transmittance electron microscopy analysis
TEM was used for analysis of the width and length of CNFs [41] The sample was diluted to 0.001% (w/w) and passed through a high-pressure homogenizer at 0–50 bar to ensure that the CNFs were evenly dispersed without agglomeration. The grid used for observation was a copper grid with silicon dioxide as a support film, and the observation grid was made by shade-dyeing CNFs with uranyl acetate. TEM (Libra 120, Carl Zeiss, Germany) was used to observe at 10,000–15,000 magnification with an acceleration voltage of 120 kV [42].
3. Results and Discussion
3.1 DS confirmation by 1H-NMR spectroscopy in carboxymethylation pretreatment
The DS in carboxymethylation pretreatment was analyzed by nuclear magnetic resonance spectroscopy. NMR peaks were calculated by integrating the peaks of the carboxymethyl group and glucose and then calculating the molar ratio [39]. Even though the NMR method is relatively more expensive than other analysis methods, it was possible to accurately analyze the DS of the carboxymethyl group in the pretreated cellulose. Quantitative and qualitative analysis was performed by integrating anomeric hydrogen peaks and carboxylmethyl group peaks on the spectrum obtained from nuclear magnetic resonance spectroscopy analysis by acid hydrolysis of carboxymethylated cellulose.
During the CNF making process, DS for carboxymethylation of cellulose is an important factor for controlling the quality of CNFs. The results ofcarboxymethylation pretreatment as DS were confirmed by 1H-NMR spectroscopy, are shown in Table 2. The position of carboxymethylation in cellulose was confirmed, that the hydroxyl group in the C6 position is preferred as a substitution in case of DS ranges from 0.1 to 0.3. If DS were higher than 0.4, the hydroxyl group in the C2 position was competed with the C6 position.
In the process of the carboxymethylation reaction, when sodium ethoxide is treated with cellulose, the secondary alcohol produces stable anions, but the primary alcohol at position 6 is easily reacted with physically [43]. In the carboxymethylation substitution reaction with different substitution degrees in Table 2, the primary alcohol at position 6 is displaced first at a substitution degree of 0.4 or less, but the substitution reaction at position 2 is preferred as the substitution degree increases.
Table 2.
DS of carboxymethylated pulp measured by 1H-NMR spectroscopy
3.2 Impact of charge density on CNF morphology
In the case of carboxymethylation pretreatment, as the DS increases, the number of anions in the cellulose increases, resulting in an increase in the electrostatic repulsion between the anions. This tendency is not only for carboxymethylation pretreatment, but also for TEMPO-oxidation pre-treated cellulose. The amount of energy required to manufacture CNFs decreases as the carboxyl group content increases, and the width of CNFs decreases under the same equipment operating conditions. The increase in the carboxymethyl groups was due to the repulsion caused by anions and the reduction of the hydrogen bonds of the cellulose, which made it possible to manufacture cellulosic nanofibers with less energy [44].
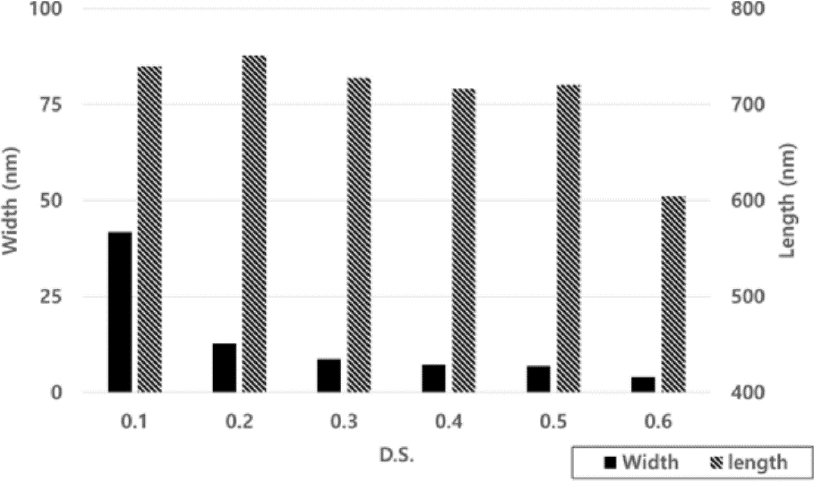
The size of the CNFs produced by different degrees of substitution of a carboxymethyl group in cellulose, and passing it through a grinder 4 times and then a high-pressure homogenizer 6 times, is shown in Fig. 1. As the DS of carboxymethyl substitution increased, the width size decreased to 41.8 nm for DS 0.1, to 4.0 nm for DS 0.6, and the length to 740.2 nm for DS 0.1, and to 604.5 nm for DS 0.6. The aspect ratio was increased with higher DS carboxymethyl substitution.
The surface charge density has a great influence on the manufacture of CNFs. The carboxymethylation pretreatment applied in this study partially substitutes the hydroxyl group of cellulose with a carboxymethyl group and introduces negative ions on the surface, thereby increasing the surface charge density. This increase in surface charge density acts as a repulsive force between cellulose molecules and interferes with hydrogen bonding, making CNFs easier to fabricate.
When bleached softwood kraft pulp (BNKP) and unbleached softwood pulp (UBNKP) were prepared using a grinder without pretreatment, CNFs from unbleached softwood pulp had a higher surface charge density than bleached softwood kraft pulp [45]. In addition, to check the degree of nanofibrilization according to the hemicellulose content, the degree of nanofibrilization was confirmed by the preparation of CNFs with holocellulose and alkaline-treated holocellulose, with hemicellulose residual, which is advantageous for nanofibrillation [46]. These results indicate that uronic acids, such as gluconic acid, are present in hemicellulose, and then the nanofiber proceeds well due to the presence of hemicellulose. It has been confirmed that it has a repulsive effect due to the negative ions caused by uronic acid, which is advantageous for nanofibrillation.
In addition to the anion contained in the raw material, the carboxyl group introduced through pre-treatment also affects the manufacture of CNFs. In particular, the cellulose surface charge density increases as more ions from the pretreatment process, and the width and length of CNFs have been decreased [47]. In the study on the change in the size of nanofibers according to the degree of pretreatment, the oxidized cellulose with charge density of 380, 820, 990, and 1,360 µmol/g respectively by applying TEMPO-oxidation pretreatment to the bleached chemical pulp and nanofibers with homogenizers As a result of the increase in charge density, the width size is between 2.1–2.6 nm, but the length is 614, 683, 583, 419 nm, and the size of the CNFs decreased as the charge density increased [48].
In addition, bleached kraft pulp with charge densities of 0.37, 0.49, and 0.51 mmol/g for softwoods and 0.46, 0.55, and 0.67 mmol/g for hardwoods was prepared by TEMPO-oxidation pretreatment on softwood and hardwood bleached kraft pulp, respectively, and the width of CNFs were 18.4–22.0 nm for softwoods, 15.1–17.5 nm for hardwoods, and the length of CNFs were 815–992 nm for softwoods and 652–850 nm for hardwoods, which meant the width and length of CNFs decreased as the charge density increased [49]. In addition, when the surface charge density of cellulose increases by the charge density, of cellulose increases by the charge density, the length of CNFs decreases. Results have been reported [50]. In carboxymethylation pretreatment, Im et al. [8] showed that the size of CNFs decreased when the charge density was increased by the pretreatment process. Similar to TEMPO-oxidation, in the case of carboxymethyl substitution pretreatment, an anion is introduced to the surface of cellulose, and the charge density increases as the degree of pretreatment increases. As for the increase in the introduction of carboxymethyl groups, there are studies that show that the mechanical processing step decreases as the carboxyl group content increases by pretreatment, and the width and length decrease [51]. It was confirmed that the anions introduced in pretreatment steps increase the charge density and decrease the size of CNFs during the nanofibrillation process. With increasing anions on the surface of the cellulose and the surface charge density due to higher DS, it has been confirmed that the size of the CNFs decreases.
3.3 Impact of passes of high-pressure homogenization on CNF morphology with different DS of carboxymethylation
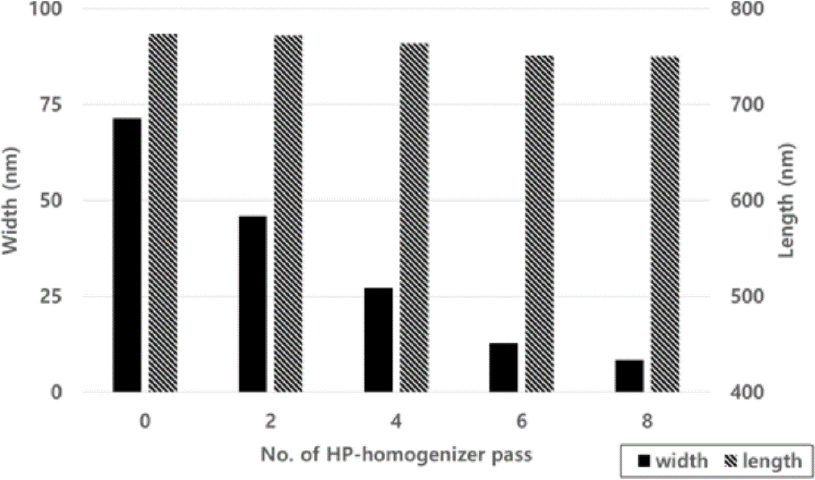
The effect of repeated high-pressure homogenizer treatment on the width and length of CNFs was investigated with DS at 0.2, 0.4, and 0.6, respectively, and the size of CNFs was measured. To investigate the impact of repeated high-pressure homogenization effect, crude CNF was made by 4 passes of supermasscolloider grindings with different DS of pulps. The high-pressure homogenizer was passed 0–8 times at a pressure of 800 ± 50 bar repeatedly, and samples were collected and analyzed every 2 passes.
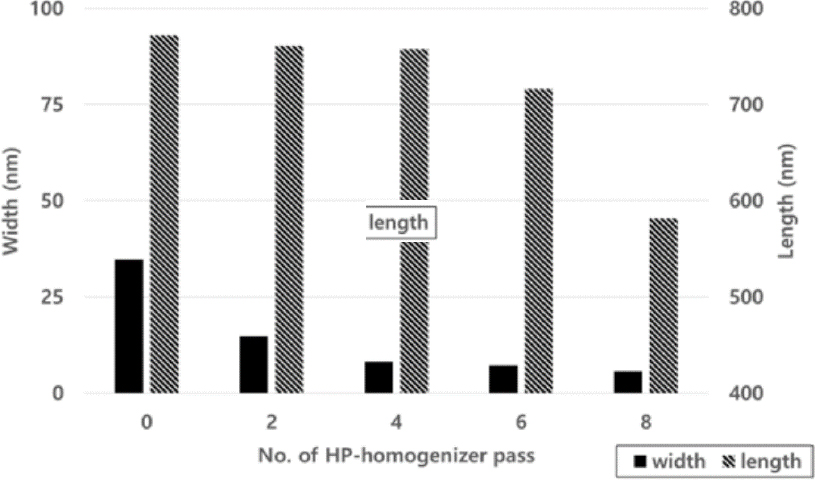
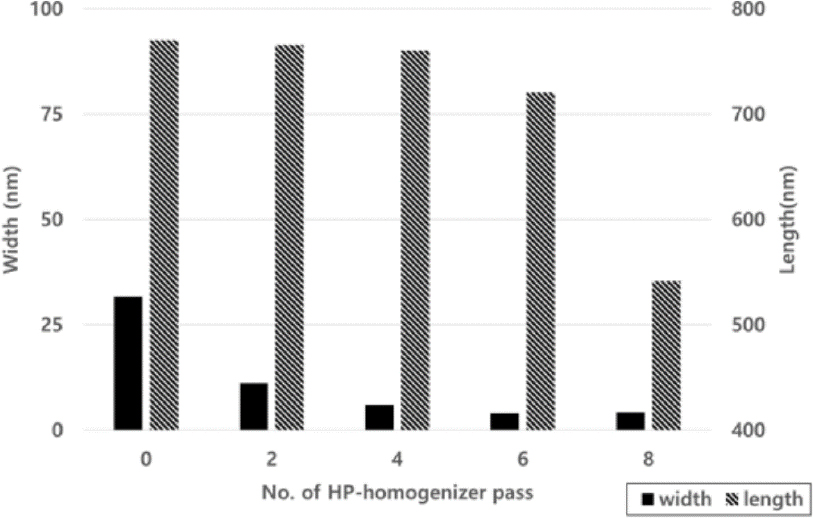
When the carboxymethyl group substitution was 0.2, the width and length were measured in Fig. 2 as the number of passes through the manufacturing equipment increased, and the width decreased from 71.4 nm to 8.4 nm, and the length did not decrease much from 749.8 to 773.5 nm as the number of times the equipment was operated. When the width and length measurements were made at 0.4 substitution degrees in Fig. 3, the width decreased from 34.7 nm to 5.8 nm, and the length remained unchanged from 716.5 to 772.1 nm after six passes, but decreased to 581.6 nm after 8 passes. When the width and length of the substitution degree of 0.6 close to the substitution degree of conventional commercial CMC were confirmed in Fig. 4, the width decreased from 31.7 nm to 4.2 nm, and the length tended to be similar to the substitution degree 0.4, showing no significant difference between 720.6 and 770.2 nm after 6 passes, but decreased to 541.2 nm after 8 passes.
This phenomenon was similar to the carboxymethyl pretreatment performed in this study, as well as other anion pretreatments. In a previous study, when CNFs were prepared using a high-pressure homogenizer after TEMPO-oxidation pretreatment on eucalyptus bleached kraft pulp, the particle size was confirmed by laser diffraction analysis, and the particle size was found to be smaller as the number of passes through the autoclave increased. In addition, the analysis of dynamic light scattering confirmed that the particle size was from 5 nm to 1,000 nm after 5 passes, but after 15 passes, the particle size was confirmed to be about 55 nm, which confirms that the size of the cellulose decreases as the equipment is operated for more times [51].
In another study, eucalyptus bleached kraft pulp was nanofibrized with a grinder for 11 hours without pretreatment, and the width of the CNFs was reduced by 3–5 nm, accompanied by a decrease in length. In addition, when CNFs were made into films, it was found that the width and length became smaller as the grinder processing time increased, increasing transparency [52]. Eucalyptus kraft pulp was nanofiberized by repeatedly passing it through a grinder device for 25 times, and it was found that the width size decreased from 40 nm to 4 nm as the number of grinds increased [53]. After pre-treatment with endo-glucanase enzyme in softwood bleached kraft pulp, nanofiberization using a grinding device for a total of 2.5 hours reduced the width size from 54.8 nm to 35.8 nm. It has been found that the width size distribution becomes more uniform as the grinder processing time increases [54]. When the microcrystalline cellulose was passed through a high-pressure homogenizer at a pressure of 20,000 psi up to 20 times, and then the width size was measured using a scanning electron microscope, it was confirmed that the number of passes through the hyperbaric homogenizer decreased from 100 nm to 28 nm as the number of passes increased [24]. When comparing the results of the previous study with the results of the CNF size measurement in this study, it was found that the increase in the usage time or number of passes of the CNF manufacturing device was associated with a decrease in the size of the CNFs.
4. Conclusions
This study demonstrates that carboxymethylation DS and mechanical fibrillation conditions precisely control CNF morphology. At DS ≤ 0.3, width reduction dominates due to moderate anionic repulsion, while at DS ≥ 0.5, both width and length decrease significantly due to enhanced charge density. The DS range of 0.3–0.5 marks a critical threshold where length reduction initiates, as confirmed by 1H-NMR spectroscopy and TEM analyses. Hemicellulose’s uronic acids further enhance nanofibrillation, complementing carboxymethylation effects. These findings provide a robust framework for tailoring CNF dimensions, advancing their use in sustainable applications like composites, biomedical materials, and eco-friendly packaging.