1. Introduction
In the alkali recovery process, it must be to do that the sodium carbonate (Na2CO3) in green liquor is converted into active sodium hydroxide
Slaking: CaO+H2O→Ca(OH)2
Causticizing: Na2CO3+Ca(OH)2→2NaOH+CaCO3
The causticizing process has two targets, which are obtained from separating the causticized liquor. One is the white liquor containing mainly NaOH. The other is causticizing calcium carbonate (CCC), One is the white liquor containing mainly NaOH. The other is causticizing calcium carbonate (CCC), known as “lime mud” or “lime sludge”. The white liquor can be reused to cooking process of plant. The lime mud, for wood materials, after dewatered, can be calcined in a rotary lime kiln and reused in the causticizing process. But for non-wood materials, this recycled way is limited due to the serious silica problems (1). In order to reach the same calcination performance as wood materials CCC, some technologies such as desilication of green liquor (2) and (3) are researched. But unfortunately, those technologies can not solve the silica problems completely. So the lime mud from the non-wood materials is often discarded as solid waste or filled bury field after dewatering. The secondary pollution occurs due to a small amount of Ca(OH)2 in the lime mud. as a kind of CaCO3, can be used as paper fillers which can bring appreciable environment (4-5). In this way, Nanri and Konno (6-9) had made many efforts to control the morphology of CCC and reduce paper-machine plastic wire abrasion. They found that CCC can be obtained in various forms such as rice-, spindle-, and needle-like particles, some of which demonstrated high opacifying ability and little plasticwire abrasion when used as paper fillers. In china, several lines of CCC filler are completed and the CCC has been used as filler in paper. From the mill trials (10-12), the CCC can be used to replace an amount of PCC, but unfortunately, reducing dosage of CCC had to be adopted because it’s higher influence on sizing efficiency of AKD than that of PCC. Now, many research works have been focused on PCC or GCC for its influence on sizing efficiency of AKD (13-15) but they did not involve the CCC. In the present study, the factors influencing the sizing efficiency of AKD in CCC filled paper were investigated in order to provide technical support for quality improvement of CCC in an alkali recovery.
2. Materials and Methods
2.1 Material
(as shown in Table 1), quicklime, PCC, AKD size (15% solids content), needle bleached kraft pulp (NBKP), laubholz bleached kraft pulp(LBKP) and cationic polyacrylamide(CPAM) were all obtained from a mill in China. Three kinds of mill CCC were obtained from three mills in mills in China. The properties of PCC and CCCs, tested accordance with Chinese National Standards,16) are shown in Table 2.
2.2 Preparation of CCC
Quicklime was added to a flask, which was filled with water and at 300 rpm to achieve slaking. Then the green liquor that was filtered by slow filter paper (Grade 1575) was added to the flask at a constant rate with a transfusion tube, and the causticizing reaction was carried out. The molar ratio of Ca:CO32- was 1.05:1. The reaction temperature was 90℃. The reaction time was 2 h. After these reactions, the CCC was filtered and washed out with water, then deliquated to 20% and injected CO2 until the pH was in the range of 9.5 to 10. After carbonation, the CCC passing 800 meshes was washed out with water and used as filler.
2.3 Determination of physical properties of CCC
The determination of particle size was carried out using a BT-9300H laser particle size analyzer. The determination of BET and BJH was carried out according to an Autosorb-iQ.
2.4 Adsorption of AKD
The experiment designed in two steps. In the first step, using an ultraviolet spectrophotometer, the absorbance of different concentrations of AKD was determined at a wavelength of 238 nm,17) and the standard curve of concentration of AKD and absorbance was drawn. In the second step, AKD was appended into the CCC suspension in a ratio 1:100 (AKD: CCC) and stirred for 5 minutes. Then, the mixture was centrifuged for 10 min in the centrifuge at 3000 rpm. The supernatant liquor was used to determine the absorbance at the wavelength of 238 nm. The content of AKD in supernatant liquor was calculated according to the standard curve. The AKD adsorbed by CCC was equal to the difference between the dosage of AKD and the content of AKD in the supernatant liquor.
2.5 Sheet forming
The 0.2% AKD( to dry stock ) was added to the dispersed stock after 60 s stirring, and 30% PCC (to total paper ) or CCC was added. Then, 0.03% CPAM (to total paper) was added after 30 s stirring, the stock was poured into the sheet form and handsheets were prepared with a basis weight of 80g/m2. The wet paper had pressed for 3 min under the pressure of 4kgf/cm2 and dried 5 min at 95℃.
3. Results and Discussion
3.1 Sizing efficiency of AKD in CCC filled paper
The sizing efficiency of AKD in CCCs or PCC filled paper is shown in Table 3.
Table 3
Sizing efficiency of AKD in filled paper
| Type of filler | PCC | CCC 1# | CCC 2# | CCC 3# |
|---|---|---|---|---|
| Cobb60 (g·m-2) | 24.3 | 70.2 | 86.1 | 123.4 |
Compared with that of PCC filler, the Cobb60 values of paper filled with CCC fillers were higher. That displayed the lower sizing efficiency of AKD. Three kinds of CCC displayed the approximate result. The results showed that there must be differences in the properties of CCC and PCC, which caused the lower sizing efficiency of AKD in the CCC-filled paper.
3.2 The influence of insoluble matter
The green liquor and clarified green liquor were used to prepare the CCC with the same conditions. The basic properties of CCC and the Cobb60 values of paper filled with CCC are shown in Table 4.
Table 4
Basic properties of CCCs and the sizing efficiency of AKD
| No. | Source of CO32- | Brightness of CCC (%ISO) | Ave. particle size (um) | Cobb60 (g·m-2) |
|---|---|---|---|---|
| 1 | green liquor | 79.65 | 6.35 | 79.6 |
| 2 | clarified green liquor | 90.07 | 5.87 | 82.2 |
We can know that the insoluble matter in green liquor just affected the brightness of CCC which had a the brightness of paper, but had no obviously influence on the sizing efficiency of AKD when it was used as filler.
3.3 The influence of silicon
The clarified green liquor was treated with sol–gel method treatment and obtained the treated green liquor.18) Two kinds of green liquor were tested the Tyndall effect, as Fig. 1 and Fig. 2. From the Fig. 1, there is colloid in clarified green liquor due to the Tyndall effect showing. When the clarified green liquor was treated with sol–gel method, the Tyndall effect is disappeared (as Fig. 2). The experiment was illuminated that the colloid in clarified green liquor was removed by sol– gel method treatment. Measured the silicon content of two kinds of green liquor, we found, along with the silicon dioxide content of sol–gel treated clarified green liquor was decreased from 6.45 g/L to 1.06 g/L. Meantime, The main component of above sol-gel derived powders was silicon. So the silicon in green liquor had been existed as a colloidal state. The two kinds of green liquor were used to prepare the CCC with the same conditions. When it was used as filler, the Cobb60 values of paper filled CCC are shown as Table 5.
Table 5
Basic properties of CCCs and the sizing efficiency of AKD
| No. | Source of CO32- | Brightness of CCC (%ISO) | Ave. particle size (um) | Cobb60 (g·m-2) |
|---|---|---|---|---|
| 1 | clarified green liquor | 90.07 | 5.87 | 82.2 |
| 2 | sol–gel treated clarified green liquor | 90.34 | 5.19 | 80.1 |
Although the silicon dioxide content contained in two kinds of green liquor were obviously different (6.45 g/L vs. 1.06 g/L), the Cobb60 values of paper filled CCC were similar. The results illuminated that the silicon had no influence on the sizing efficiency of AKD. The reason may be due to the fact that the silicon was existed the inside of CCC, not the surface.
3.4 The influence of other element in clarified green liquor
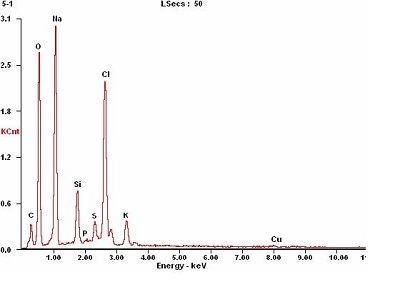
The clarified green liquor was dried at 60℃, and and ground into powder. The powder was tested with EDX as Fig.3.
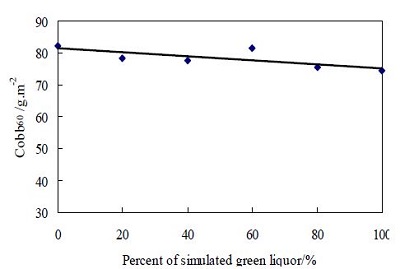
The elements which contained in the clarified green liquor are not only Na, O, C, Si but also Cl, P, S, K, Cu and so on. In order to analysis the influence of other elements on property of CCC which decreased the sizing efficiency of AKD. The Na2CO3 (AR, analytical reagent) and the NaOH (AR) were used to simulate the clarified green liquor according to its content in mill green liquor. The clarified green liquor was mixed with simulated green liquor at different ratio, and the CCCs were prepared with the same conditions. The Cobb60 values of paper filled with CCC are shown in Fig. 4.
There are no elements of Cl, P, S, K, Cu in Na2CO3 (AR) or NaOH (AR), sothe mixture which mixed the simulated green liquor with clarified green liquor is equivalent to decrease those of elements. From the Fig.4, the Cobb60 values of paper filled CCC are no obviously different, so we thought that the other elements had been no influence on the sizing efficiency of AKD.
3.5 The influence of CCC
By precisely controlling the causticizing reaction as Yasunori19) and Wang’s research,20) the needle-like CCCs were prepared as Fig. 5. When it was used as filler, we surprised to find that the Cobb60 value of paper filled CCC was approximately that of PCC, as shown in Table 6.
Table 6
the properties of fillers and the Cobb60 values of filled paper
| No. | Type of filler | Ave. particle size (um) | Cobb60 (g·m-2) |
|---|---|---|---|
| 1 | PCC | 4.03 | 24.3 |
| 2 | Needle-like CCC | 4.42 | 20.2 |
There is no obviously different ave. particle size between PCC and needle-like CCC. But the sizing efficiency of AKD in the CCC-filled paper was enhanced significantly. The results showed that there must be some changes occurred in the properties of CCC with the change of its micromorphology. The physical properties of fillers are shown in Table 7.
From the Table 7, the ave. particle size of mill CCC 2# or irregular CCC is a little bigger than that of PCC or needle-like CCC, but, compared with PCC or needle-like CCC, both its BET and BJH pore volume are increased to about 3 times. The higher BET of filler, the more AKD is adsorbed. The AKD that is adsorbed by the filler will become spreading AKD molecules and migrate into the void structure of the pore of CCC in drying process. Meanwhile, a portion of the AKD molecules in contact with surface of CCC can also migrate into the void structure in the drying process. Those AKD are then unavailable for sizing.21) That provides an explanation why there has lower sizing efficiency when conventional CCC is used to fill the paper. So controlling the BET and the BJH pore volume of the CCC are effective way to improve the sizing efficiency of the CCC filled paper.
4. Conclusions
In this study, the results showed as following;
- The materials in green liquid, such as insoluble matter in green liquid, silicon and metal ions, were a little influence on the sizing efficiency of AKD.
- The higher BET and BJH pore volume of the CCC were the main reason for lower sizing efficiency of AKD when it was used as filler.