1. Introduction
2. Materials and Methods
2.1 Materials
2.2 Extraction of tannins
2.3 Analysis of bark extract characteristics
3. Results and Discussion
3.1 The effect of solvent and particle size on extract yield
3.2 Polyphenol content in bark extract
3.3 Analysis of tannin purity and composition
3.4 Ratio of condensed and hydrolyzable tannins
4. Conclusions
1. Introduction
Recent research on climate change mitigation and the establishment of a circular resource society has intensified, making the high-value utilization of underutilized biomass, such as forest by-products, a critical imperative [1,2]. Bark, one underutilized biomass resource, constitutes approximately 10–20% of the total log volume and is generated in large quantities as a by-product during timber sawing and processing [3]. Compared with woody materials, bark has a lower cellulose content and higher levels of lignin, ash, and extractives, negatively affecting pulp and paper processes. Consequently, it is mainly used as a simple fuel or discarded [4]. However, bark contains significant quantities of natural polyphenolic compounds, such as tannins, flavonoids, and triterpenes. These are reported to possess potential for utilization as functional materials, including antioxidants, antibacterial agents, natural dyes, and adhesives [5,6,7].
Tannins are high molecular weight polyphenolic compounds, generally classified as hydrolyzable (gallotannins and ellagitannins) and condensed tannins (proanthocyanidins). Hydrolyzable tannins possess relatively simple structures and are readily soluble in water or alcohol, whereas condensed tannins are polymers of flavonoid units such as catechin and epicatechin. Their stable structure, characterized by excellent heat resistance, high cohesiveness, and water resistance, renders them useful as adhesive raw materials [8]. This structural diversity means that recovery rates and composition vary significantly depending on extraction solvent, temperature, time, and sample particle size [9].
Tannins currently utilized commercially are primarily produced from specific tree species such as chestnut, mimosa, and quebracho, with the majority dependent on imports [10]. Conversely, oak, an economically significant tree species abundantly distributed domestically, accounts for approximately 20% of the total standing timber volume. Its processing into lumber products, such as boards and chips, generates substantial quantities of bark. This bark represents a significant tannin resource with high potential for value-added utilization [11]. Notably, oak bark is reported to contain 10–20% tannins, owing to its high lignin content and the concentration of condensed tannins within its rigid tissue [12]. Despite this potential, systematic research on the extraction, quantification, and compositional analysis of bark tannins has been largely absent domestically.
Tannin extraction is typically performed using reflux or ultrasonic methods with solvents such as benzene, ethanol, methanol, or acetone [8,9,10]. According to Lapornik et al. [13], when ethanol or methanol is used as a solvent, polyphenolic components are extracted at over twice the rate compared with aqueous solvents such as water. Recent studies have focused on environmentally friendly tannin recovery processes employing mixed solvent systems such as ethanol-water or acetone-water. Dedrie et al. [4] reported that a 50% ethanol–water solvent was the most effective for polyphenol recovery from oak bark. They also demonstrated that the solubility of polyphenols and the proportion of condensed tannins vary depending on the solvent composition. Consequently, natural extracts exhibit variations in composition and yield depending on the extraction method employed.
However, previous studies have primarily focused on solvent optimization, and few have provided an integrated analysis of the effects of bark particle size or the characteristics of the bark residue remaining after tannin extraction.
In this study, we systematically compared and analyzed the tannin extraction efficiency, total polyphenol content, tannin purity, and compositional ratio (hydrolyzable/condensed tannin) of oak bark in terms of solvent composition (acetone and ethanol mixture ratio) and particle size.
2. Materials and Methods
2.1 Materials
This study aimed to utilize underutilized forest resources efficiently. Oak bark, a by-product generated during timber sawing and processing, was provided by the Korea Wood Chip Association. Following natural drying, the bark was pulverized using a pulverizing machine (RT-02A, Rong Tsong Precision Technology Co., Taiwan). It was then classified into three grades based on particle size using a standard testing sieve (Chung Gye Sang Sa, Korea): over 40 mesh, 20–40 mesh, and under 20 mesh (Fig. 1). The graded samples were dried at 50°C before use. The solvents used for tannin extraction were acetone (99.5%, Extra pure), ethanol (94.5%, Extra pure), and benzene (99.5%, Extra pure), all purchased from Duksan Pure Chem. Co., Ltd. (Korea).
2.2 Extraction of tannins
Acetone and ethanol were mixed with distilled water for tannin extraction and diluted to 90% and 70% (v/v) concentrations, respectively. For each particle size, 5 g of bark was mixed with 100 mL of solvent and reflux-extracted for 5 hours in a Soxhlet extractor (WHM12295, DAIHAN Scientific Co., Ltd., Korea) at 80°C using filter paper (Advantec No. 84, Japan). Following extraction, the solution was processed in a rotary evaporator (Buchi Rotavapor R200, Switzerland) to remove the solvent. After solvent evaporation, the powdered extract (tannin) was recovered. The extraction yield was calculated using the difference in flask weight before and after extraction (Eq. (1)). Furthermore, the bark residue remaining on the filter paper after tannin extraction was washed with distilled water and dried at 105°C for subsequent pulping and chemical composition analysis. For comparative experiments, the TAPPI T204 standard degreasing method using an ethanol-benzene (1:2, v/v) mixed solvent was performed. 2 g of dried bark (> 40 mesh) were mixed with 100 mL of ethanol-benzene solution and extracted for 6 hours using a Soxhlet extractor at 80°C.
2.3 Analysis of bark extract characteristics
The polyphenol content of the bark extract was determined using the Folin–Ciocalteu method [14], and absorbance was measured with a UV–Vis spectrophotometer at a wavelength of 725 nm. Gallic acid monohydrate (Extra pure) was used as the standard material. Calculations were performed by converting the measured values to gallic acid equivalents (GAE) per gram of extract or dry bark based on a calibration curve. The tannin content among the polyphenols was analyzed using the cinchonine sulfate precipitation method [15]. A cinchonine sulfate solution was added to the extract to selectively precipitate the tannin components, and the absorbance of the supernatant was then measured. The difference between the total polyphenol and non-tannin polyphenol content in the supernatant determined tannin purity [16]. Furthermore, the contents of condensed and hydrolyzable tannins within the extracted tannins were quantified separately using the formaldehyde–hydrochloric acid precipitation method [17]. After separating the precipitated condensed tannins, the absorbance of the remaining hydrolyzable tannins in the supernatant was measured to determine the relative ratio of the two tannin types. All experiments were performed in triplicate to ensure reliability.
3. Results and Discussion
3.1 The effect of solvent and particle size on extract yield
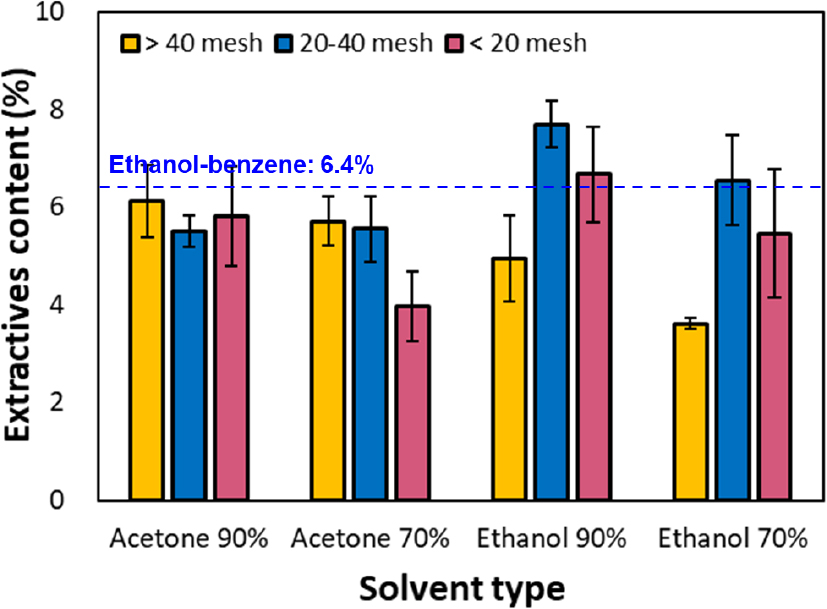
The yield of tannin extract from bark, depending on bark particle size and the solvent used for extraction, is presented in Fig. 2. Generally, particle size is a key factor affecting the solvent’s permeability and diffusion rate in the extraction process [18]. When oak bark was classified into particles larger than 20 mesh, 20–40 mesh, and smaller than 40 mesh for tannin extraction, the yield was 5.5% for the coarse fraction (< 20 mesh). However, the highest average yield of 6.3% was observed for the 20–40 mesh sample. This is likely because finer particles provide a larger surface area per unit mass, enhancing solvent contact and penetration [19]. However, for the finest fraction (> 40 mesh), the average yield decreased to 5.1%. Pătrăuţanu et al. [18] reported that, when extracting tannins from bark, if the bark particle size is extremely fine (0.25–0.31 mm), the particles do not disperse uniformly in the solvent and tend to remain on the sample surface, thereby restricting solvent penetration and reducing extraction efficiency. Similarly, this suggests that the reduced extraction yield was due to the agglomeration of fine particles > 40 mesh (~0.25 mm) on the sample surface, which impeded solvent penetration.
Comparing extraction yields according to solvent type and concentration, using 90% acetone and 90% ethanol solvents resulted in average yields of 5.8% and 6.4% respectively, indicating that the ethanol exhibited slightly higher extraction efficiency. This trend is consistent with the findings of Dedrie et al. [4]. Notably, high yields were observed when extracting bark particles larger than 40 mesh with ethanol, whereas the efficiency of ethanol extraction tended to decrease for finer particles (> 40 mesh). Acetone showed less dependence on particle size than ethanol. This may be because acetone more effectively dissolves non-polar extractives, causing solvent–extract interactions to outweigh the surface-area effects of finer particles. Meanwhile, when the solvent concentration was diluted to 70%, acetone and ethanol exhibited yield reductions of approximately 20% and 12%, respectively, compared to the 90% solvent, resulting in extraction yields of 5.1% and 5.2%. Even within the same solvent type, higher extraction rates were observed with higher-concentration solvents. This is likely because the solubility of polyphenolic compounds improves with increasing solvent concentration, thereby enhancing extraction efficiency [20].
Furthermore, the extraction yield of the ethanol/benzene (1:2, v/v) degreasing method, performed as a control, was approximately 6.4%. This was comparable to the 90% acetone solvent extraction at the same particle size. However, the ethanol-benzene extraction method may have yielded a higher extraction rate because it incorporates separate wood extractives in addition to tannins. Conversely, the extraction of 20–40 mesh samples using 90% ethanol solvent resulted in yields more than 20% higher than those of the conventional ethanol/benzene extraction method. These results suggest that extraction processes utilizing environmentally friendly solvents, such as ethanol-water or acetone-water systems, can effectively replace traditional benzene-based solvents.
3.2 Polyphenol content in bark extract
The total polyphenol content in the extract, according to particle size and solvent type, is shown in Table 1 to evaluate the quality of tannins extracted from bark. First, analysis by particle size revealed that the highest polyphenol content was observed in fine particles (> 40 mesh), averaging 919.85 mg GAE/g. As particle size increased, the content decreased to 416.12 mg GAE/g (20–40 mesh) and 302.37 mg GAE/g (> 20 mesh). This was attributed to the increased specific surface area per unit mass of smaller particles, allowing the polyphenolic components to be more readily eluted into the solvent [19]. Specifically, when using fine particles (> 40 mesh) and extracting with 70% and 90% ethanol solvents, polyphenol contents of 959.48 mg GAE/g and 929.02 mg GAE/g, respectively, were obtained. Among these, the 70% ethanol condition yielded slightly higher values, likely because the ethanol–water mixture can partially dissolve both polar and non-polar components, making it more efficient for the extraction of phenolic compounds [9,21]. Acetone showed a similar trend, though it recorded lower values of 883.49 mg GAE/g (90% acetone) and 907.40 mg GAE/g (70% acetone) compared to ethanol. Therefore, combining finely ground samples (> 40 mesh) and ethanol-based solvents represents an optimal condition for high-yield extraction of functional components, such as polyphenols and tannins, from bark.
Table 1.
Polyphenol yield per gram of bark extract (mg GAE/g extract) according to particle size and solvent composition
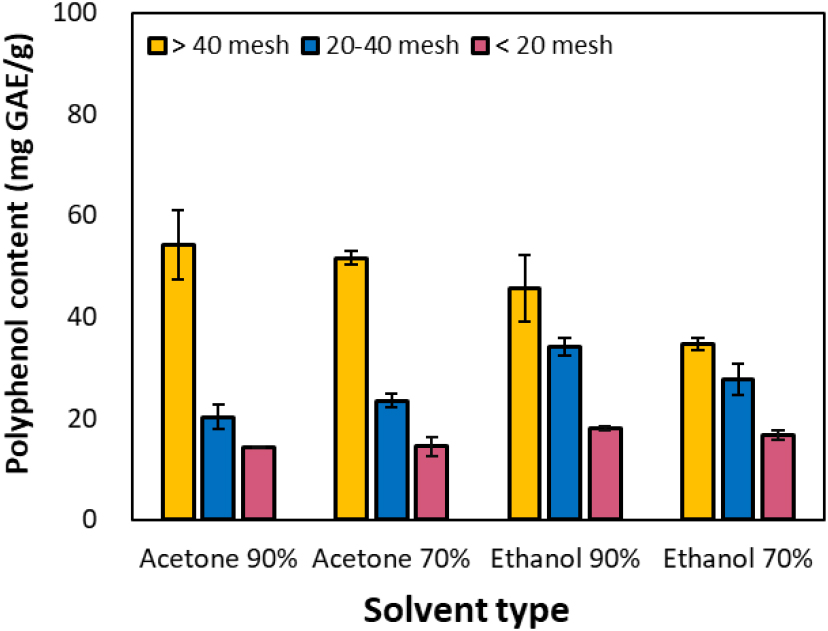
The results of analyzing the total polyphenol content extractable per gram of dry bark (mg GAE/g) are presented in Fig. 3. This analysis aimed to quantitatively compare not only the simple yield but also the effect of solvent conditions and particle size variation on the actual active ingredient (polyphenol) content. Overall, the extractable polyphenol content per gram of bark increased as particle size decreased. Notably, under the condition of applying acetone to fine particles, a high polyphenol yield of approximately ≥ 54 mg GAE/g was achieved. Under identical solvent conditions, extraction efficiency improved with increasing solvent concentration, enhancing the polyphenol content per unit mass.
3.3 Analysis of tannin purity and composition
Table 2 compares the purity of the extracted tannins. Regardless of solvent conditions, the finer fraction (> 40 mesh) exhibited relatively high tannin purity (508.82 mg GAE/g), which is 4.2 times and 3.3 times higher than that of the 20–40 mesh and < 20 mesh fractions, respectively. Depending on the solvent, the 90% ethanol showed a relatively high purity, averaging 336.36 mg GAE/g. Notably, the highest purity value of 647.31 mg GAE/g was under fine-particle and 90% ethanol conditions. This suggests that the high-concentration polar solvent acted favorably in selectively extracting polyphenolic components within the fine particles [20]. Consequently, the optimal conditions for high-quality tannin extraction were determined to combine fine particles of > 40 mesh and high-concentration ethanol (90%).
Table 2.
Tannin yield per gram of bark extract (mg GAE/g extract) according to particle size and solvent composition
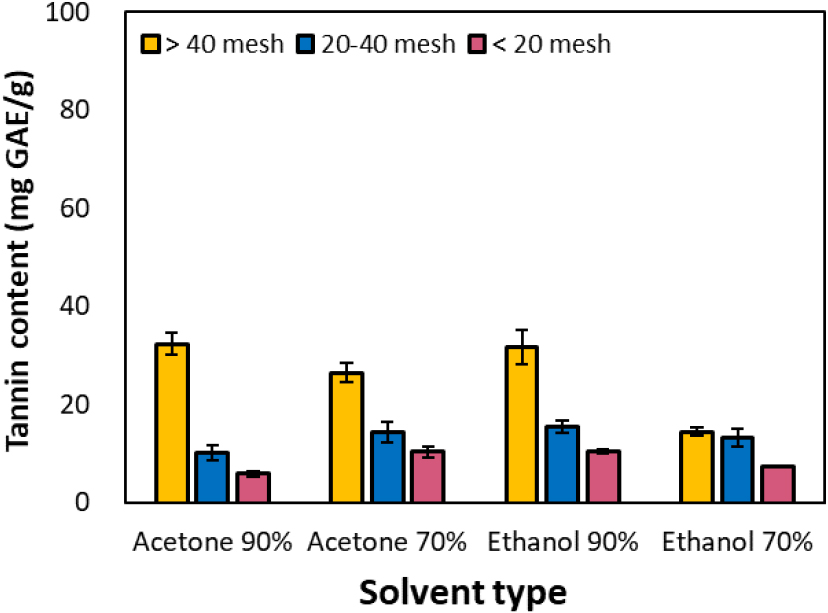
Considering the extraction yield, the actual tannin content extractable per gram of dry bark was highest in fine particles (> 40 mesh), as shown in Fig. 4. In particular, when extracted from samples finer than 40 mesh using 90% acetone and ethanol, a high value of approximately 32 mg GAE/g was obtained. The 90% solvent condition demonstrated superior tannin extraction efficiency compared to the 70% condition, and acetone generally exhibited higher purity tannin extraction efficiency than ethanol. Specifically, under the 70% acetone condition, the extraction tannin yield at > 40 mesh was approximately 26.5 mg GAE/g, representing about 81.8% of the yield observed at the same particle size (32.4 mg GAE/g) under 90% ethanol conditions.
Conversely, for the ethanol/benzene extracted extract (Table 3), the total polyphenol content was relatively high at 391.6 mg GAE/g, whereas the actual tannin content was only 37.5 mg GAE/g, indicating low tannin purity. This was attributed to increased leaching efficiency of lipophilic components (resins, waxes, fatty acids) when benzene was used, resulting in a lower proportion of pure tannins. These results suggest that particle size, solvent polarity, and solvent concentration are key factors in maximizing extraction efficiency and establishing optimal conditions for the extraction process. Specifically, the combination of 90% ethanol and fine particles (> 40 mesh) was identified as a high-efficiency and high-purity extraction condition.
Table 3.
Polyphenol and tannin content of bark extract obtained by the ethanol-benzene extraction method
| GAE (mg) / bark extraction (g) | GAE (mg)/ dried bark before extraction (g) | |
| Polyphenol | 391.55 ± 4.12 | 23.96 ± 0.25 |
| Tannin | 37.53 ± 1.06 | 2.30 ± 0.06 |
3.4 Ratio of condensed and hydrolyzable tannins
Table 4 compares the hydrolyzable and condensed tannin content per gram of dry bark. Generally, hydrolyzable tannins were measured at the highest levels in the fine fraction (< 40 mesh). Specifically, in samples > 40 mesh using a 70% acetone solvent, hydrolyzable tannins showed the maximum value of 104.7 mg GAE/g. In coarser fractions (> 20 mesh), the extraction yield of tannins decreased markedly under all conditions. Furthermore, a tendency was observed where the extraction yield of condensed tannins increased with decreasing particle size. The influence of solvent concentration and type was also distinctly observed. When using 90% acetone and ethanol with > 40 mesh particles, the hydrolyzable tannin decreased compared with the lower 70% solvent, while the condensed tannin content slightly increased. This was attributed to the characteristic behavior of condensed tannins, which generally possess polymeric structures with strong hydrogen bonds and intermolecular cohesive forces, thereby increasing their solubility when water is retained in the solvent system. This suggests that, since tannins are distributed deeply within the bark tissue, higher recovery rates can be achieved when solvent penetration and cell wall disruption are more effectively facilitated [8]. Conversely, hydrolyzable tannins, being relatively low-molecular-weight compounds present in the bark surface layer, exhibited a comparatively smaller change in extraction yield with increasing particle size and solvent concentration than condensed tannins.
Table 4.
Content (mg GAE) of hydrolyzable and condensed tannins per gram of tannin
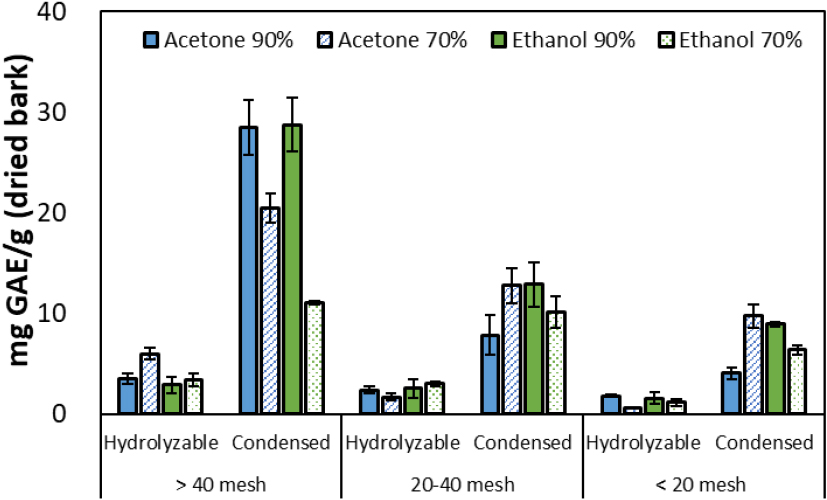
Considering the extraction yield, the results of analyzing the extractable tannin content per gram of dry bark (including both hydrolyzable and condensed types) according to solvent type, concentration, and bark particle size are presented in Fig. 5. For hydrolyzable tannins, a low recovery rate of 1–6 mg GAE/g was observed, irrespective of solvent type or particle size. Conversely, condensed tannin content ranged from 4–29 mg GAE/g, exhibiting significantly higher values than hydrolysable tannins. These results align with previous reports indicating proanthocyanidins (condensed tannins) as the primary polyphenolic constituents in oak bark, suggesting the abundant presence of high-molecular-weight condensed tannins within the bark [8,22].
Notably, approximately 28 mg GAE/g of condensed tannins were extracted from fine particles > 40 mesh under conditions using 90% acetone and 90% ethanol, respectively. Furthermore, despite the relatively low solvent purity of the 70% acetone solvent, over 21 mg GAE/g of condensed tannins could be obtained. This is due to acetone maintaining its solubility for phenolic compounds while inducing relatively rapid penetration and diffusion. Consequently, the combination of fine particles (> 40 mesh) and a high-concentration (≥ 70%) solvent was confirmed as the most suitable condition for the efficient recovery of condensed tannins.
4. Conclusions
This study comprehensively evaluated the extraction yield, polyphenol content, and tannin purity variations based on bark particle size, solvent type, and concentration, utilizing oak bark—an underutilized biomass resource—for tannin extraction. Analysis by particle size revealed that samples with a mesh size of 20–40 exhibited the highest extraction efficiency, with an average yield of 6.3%. Conversely, fine particles with a mesh size of > 40 showed a relatively low average yield of 5.1%. However, considering the polyphenol content and tannin purity within the extract, smaller particles demonstrated a higher tannin recovery efficiency. Comparing extraction efficiency by solvent type, 90% ethanol yielded the best results with an average of 6.4%, followed by 90% acetone at 5.8%, and 70% ethanol and acetone at 5.2% and 5.1% respectively. A trend was observed where higher solvent concentrations increased the yield of the extract. Notably, when using 90% ethanol, adjusting the particle size of the bark enabled yields equivalent to or higher than those achieved with the conventional extraction method using a mixed solvent of ethanol/benzene (1:2, v/v). Furthermore, when evaluated based on polyphenol content and tannin purity within the extract, acetone extraction demonstrated higher efficiency than ethanol. It was confirmed that relatively high tannin content could be obtained even under low-concentration conditions, such as 70% acetone. Meanwhile, the extraction of fine bark particles (> 40 mesh) using 90% ethanol yielded the highest efficiency in terms of condensed tannin recovery. These results suggest that particle size and the type of extraction solvent significantly affect the quality of the extracted tannin.